Cost-Effectiveness of Preventing Depression Among At-Risk Youths: Postintervention and 2-Year Follow-Up
Abstract
Objective:
Methods:
Results:
Conclusions:
HIGHLIGHTS
Methods
Participants
Clinical Outcomes
Costs
| Any use (%) | Frequency of use | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 9 months | 33 months | 9 months | 33 months | |||||||||
| CBP | Usual care | CBP | Usual care | |||||||||
| Service | CBP | Usual care | CBP | Usual care | M | SD | M | SD | M | SD | M | SD |
| Inpatient mental health days | 1.4 | 1.3 | 3.4 | 3.4 | 46.5 | 58.7 | 11.0 | 9.9 | 6.6 | 76.6 | 15.2 | 15.0 |
| Inpatient alcohol or drug days | 1.4 | 0 | .7 | 0 | 24.0 | 28.3 | na | na | 25.5 | — | na | na |
| Counseling or medication management visits | 30.7 | 27.4 | 47.3 | 43.8 | 12.0 | 17.9 | 9.1 | 14.3 | 16.0 | 22.9 | 15.2 | 22.0 |
| Day hospital days | 0 | 0 | .7 | 2.1 | na | na | na | na | 107.0 | — | 11.7 | 9.5 |
| Alcohol or drug treatment visits | .7 | .6 | 4.1 | 2.1 | 13.0 | — | 33.0 | — | 8.0 | 5.1 | 31.0 | 26.1 |
| Crisis services | 2.1 | .6 | 2.7 | 2.7 | 31.7 | 37.9 | 2.0 | — | 24.3 | 34.3 | 4.3 | 4.0 |
| Medical doctor visits | 7.1 | 11.5 | 15.8 | 15.8 | 2.1 | 1.5 | 1.8 | 1.1 | 2.6 | 1.9 | 2.8 | 4.8 |
| Emergency department visits | 2.1 | 1.3 | 3.4 | 2.1 | 1.0 | 0 | 2.5 | 2.1 | 1.0 | 0 | 2.0 | 1.7 |
| Days of antidepressant medication | 5.1 | 5.1 | 15.8 | 12.7 | 120.9 | 77.8 | 126.0 | 86.5 | 145.0 | 194.5 | 168.8 | 214.4 |
| Days of stimulant medication | 3.2 | 1.9 | 8.9 | 6.4 | 105.6 | 74.2 | 61.0 | 30.0 | 202.1 | 257.0 | 535.9 | 806.6 |
| Days of other psychotropic medication | 0 | 1.3 | .6 | 1.9 | na | na | 92.0 | 86.3 | 121.0 | — | 223.7 | 236.1 |
| Any school services | 22.1 | 22.9 | 32.9 | 34.2 | 23.4 | 52.5 | 44.9 | 105.2 | 33.5 | 91.9 | 38.6 | 99.2 |
| Juvenile correction contact | .7 | 3.2 | 4.1 | 11.6 | 1.0 | — | 5.2 | 7.3 | 6.2 | 4.5 | 7.6 | 9.3 |
Statistical Analyses
Sensitivity Analyses
Missing Data
Results
Clinical Outcomes
Service Use and Costs
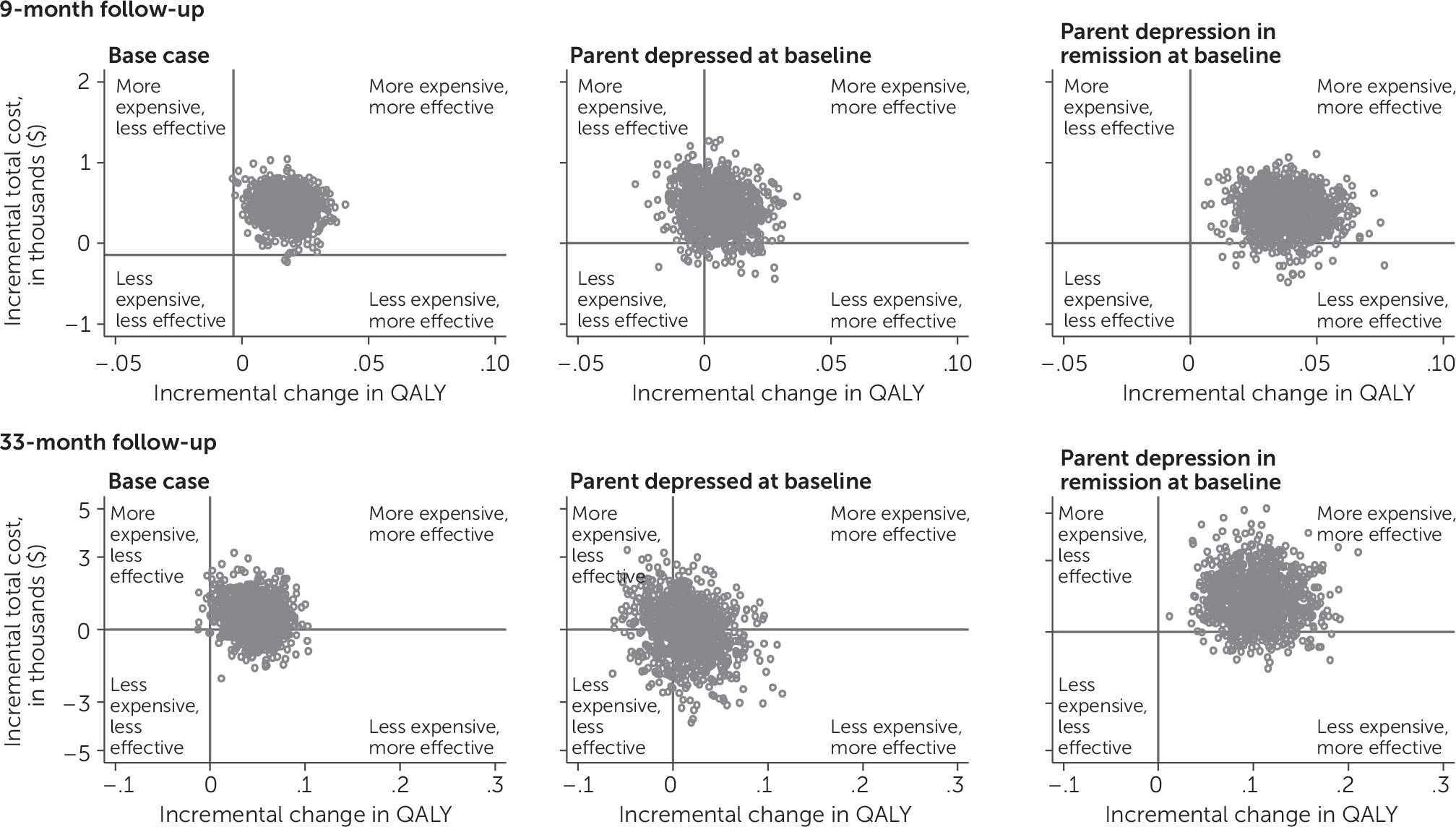
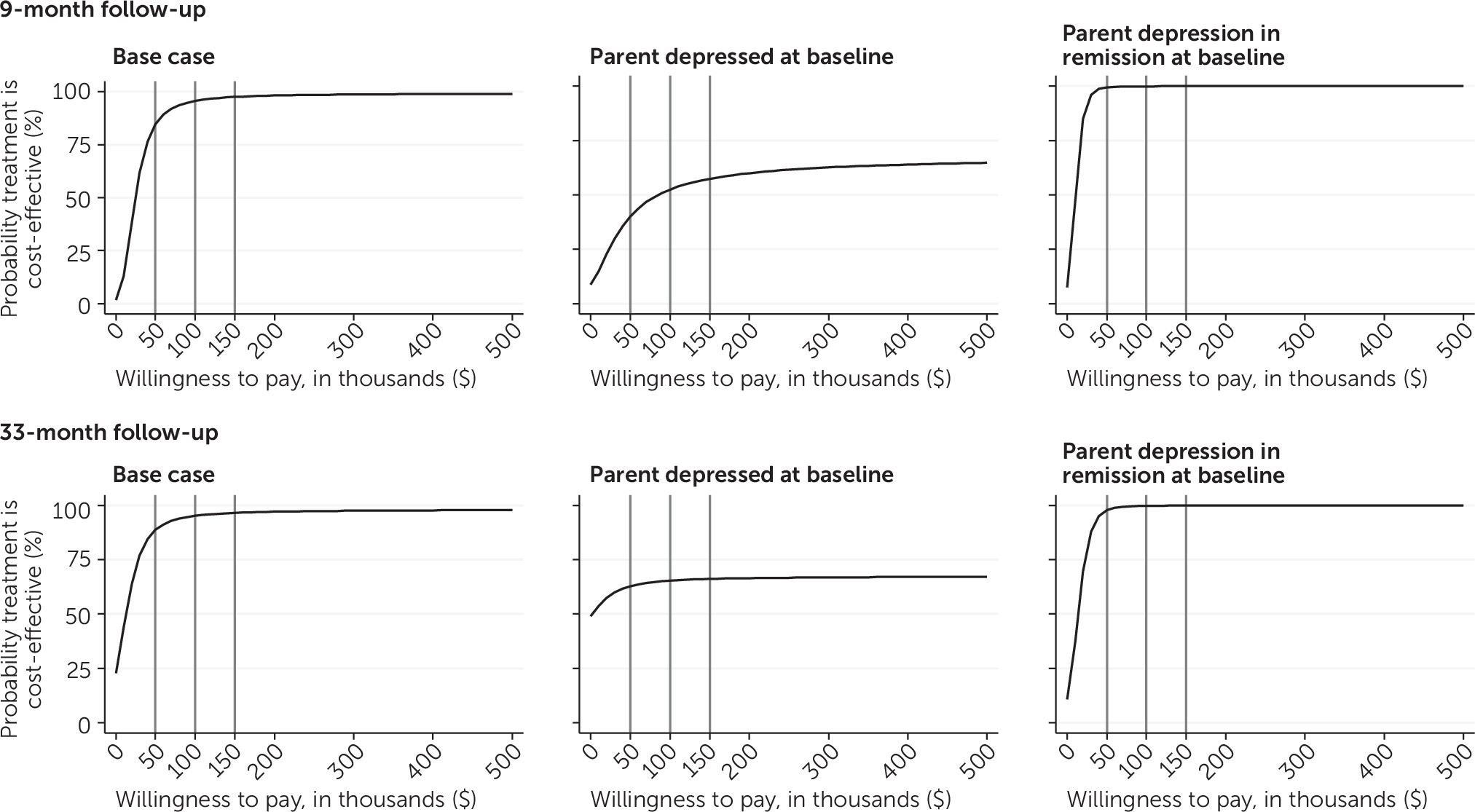
Cost-Effectiveness Analysis
| Outcome | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Clinical outcome | ||||||||||
| Cost ($) | DFD | QALY | Cost per DFD ($) | Cost per QALY ($) | ||||||
| Follow-up point and variable | M | SEb | M | SEb | M | SEb | M | 95% CIc | M | 95% CIc |
| 9 months | ||||||||||
| Base-case analysis (N=315) | ||||||||||
| CBP | 1,189 | 177 | 231.1 | 4.3 | .853 | .006 | ||||
| Usual care | 753 | 174 | 218.8 | 4.3 | .835 | .006 | ||||
| Difference (CBP–usual care)d | 436* | 184 | 12.3* | 4.9 | .018* | .007 | ||||
| ICER | 35 | 1 to 154 | 24,558 | 663 to 106,839 | ||||||
| Subgroup analysis | ||||||||||
| Parental depression, current | ||||||||||
| CBP | 1,326 | 281 | 229.6 | 6.4 | .850 | .009 | ||||
| Usual care | 892 | 271 | 226.1 | 6.6 | .845 | .009 | ||||
| Difference (CBP–usual care)d | 433 | 266 | 3.5 | 6.5 | .005 | .009 | ||||
| ICER | —e | —e | —e | —e | ||||||
| Parental depression, not current | ||||||||||
| CBP | 1,008 | 232 | 235.4 | 6.0 | .859 | .009 | ||||
| Usual care | 611 | 232 | 209.2 | 5.9 | .821 | .009 | ||||
| Difference (CBP–usual care) | 397 | 244 | 26.2** | 7.5 | .038** | .011 | ||||
| ICER | 15 | –6 to 43 | 10,498 | –3,793 to 29,965 | ||||||
| 33 months | ||||||||||
| Base-case analysis (N=315) | ||||||||||
| CBP | 3,879 | 667 | 887.6 | 17.8 | 2.586 | .020 | ||||
| Usual care | 3,305 | 667 | 846.7 | 17.6 | 2.541 | .020 | ||||
| Difference (CBP–usual care)d | 574 | 738 | 40.4* | 17.9 | .045* | .020 | ||||
| ICER | 14 | –28 to 133 | 12,787 | –24,991 to 119,958 | ||||||
| Subgroup analysis | ||||||||||
| Parental depression, current | ||||||||||
| CBP | 3,894 | 989 | 872.6 | 25.3 | 2.570 | .028 | ||||
| Usual care | 3,867 | 992 | 858.9 | 26.2 | 2.554 | .029 | ||||
| Difference (CBP–usual care) | 28 | 1,109 | 13.7 | 25.5 | .015 | .028 | ||||
| ICER | —e | —e | —e | —e | ||||||
| Parental depression, not current | ||||||||||
| CBP | 4,037 | 951 | 912.3 | 24.1 | 2.614 | .027 | ||||
| Usual care | 2,656 | 933 | 821.0 | 22.5 | 2.512 | .025 | ||||
| Difference (CBP–usual care) | 1,381 | 1,060 | 91.3** | 27.2 | .101** | .030 | ||||
| ICER | 15 | –5 to 53 | 13,620 | –4,111 to 47,563 | ||||||
Discussion
Conclusions
Acknowledgments
Supplementary Material
- View/Download
- 28.71 KB
References
Information & Authors
Information
Published In
History
Keywords
Authors
Funding Information
Metrics & Citations
Metrics
Citations
Export Citations
If you have the appropriate software installed, you can download article citation data to the citation manager of your choice. Simply select your manager software from the list below and click Download.
For more information or tips please see 'Downloading to a citation manager' in the Help menu.
View Options
View options
PDF/EPUB
View PDF/EPUBLogin options
Already a subscriber? Access your subscription through your login credentials or your institution for full access to this article.
Personal login Institutional Login Open Athens loginNot a subscriber?
PsychiatryOnline subscription options offer access to the DSM-5-TR® library, books, journals, CME, and patient resources. This all-in-one virtual library provides psychiatrists and mental health professionals with key resources for diagnosis, treatment, research, and professional development.
Need more help? PsychiatryOnline Customer Service may be reached by emailing [email protected] or by calling 800-368-5777 (in the U.S.) or 703-907-7322 (outside the U.S.).

