Introduction
Time limited psychodynamic psychotherapy is garnering support as an empirically supported treatment for depression (
de Maat et al., 2008;
Driessen et al., 2010;
Gerber et al., 2011;
Luyten & Blatt, 2012). However, the efficacy of psychodynamic interventions has never been evaluated in a medical care context as an adjunct to medical treatment for cancer. The median point prevalence rate of major depression in cancer patients ranges from 22%-29%, with depression prevalence in women with breast cancer estimated at approximately 30% (
Hotopf, Chidgey, Addington-Hall, & Ly, 2002;
Massie, 2004). Relative to nondepressed cancer patients, depressed cancer patients present with more severe psychosocial and psychiatric problems (
Parker, Baile, de Moor, & Cohen, 2003) as well as deteriorated physical health and quality of life (
Hopko al., 2008;
Mystakidou et al., 2007;
Spiegel & Giese-Davis, 2003;
Stommel, Given, & Given, 2002;
Weitzner, Meyers, Stuebing, & Saleeba, 1997). This case study examined the efficacy of pragmatic psychodynamic psychotherapy ([PPP]
Summers & Barber, 2010) in treating a depressed breast cancer patient in the context of an oncology clinic.
Pragmatic psychodynamic psychotherapy is a semistructured approach and is derived from pre-existing case formulations and psychodynamic treatment techniques. This treatment approach is predicated on a developmental and conflict model of psychopathology, entails clearly defining goals at the onset of treatment, and is more focused than traditional psychoanalytic approaches. This time-limited approach is also explicit in promoting change in behaviors, thoughts, and affect, with treatment goals that are ideographically modified and consist of decreasing vulnerability to abandonment and decreasing harsh self-criticism (
Summers & Barber, 2010). Therapy initially focuses on creating a supportive environment, depression education, psychosocial history assessment, and collaborative efforts to identify problems and develop treatment goals. This approach also involves identifying key relationship themes, with the intention of developing a collaborative understanding of the patient’s Core Conflictual Relational Theme ([CCRT]
Luborsky, 1977). This CCRT facilitates communication and a collaborative conceptualization of the patient’s adaptive and maladaptive intra- and inter-personal style of relatedness. Change subsequently evolves through increased insight about maladaptive relational patterns and by working to become more flexible and open to novel styles of relatedness. The final treatment phase entails the patient and therapist working to consolidate insights gained about depressive affect, practicing new relational patterns, and processing any encountered conflict(s).
In general, there is empirical evidence to support the efficacy of psychodynamic psychotherapy, with reported effect sizes being comparable to other treatments that have been labeled “empirically supported” (see
Shedler, 2010 for review). The efficacy of psychodynamic therapy has been tested via randomized controlled trials and has garnered support for treating various psychological problems including depression, anxiety, panic, somatoform, and eating, substance-related, and personality disorders (
Leichsenring, 2005;
Milrod et al., 2007). Specific to depression, the few studies that have directly compared cognitive behavioral treatment (CBT) to short-term psychodynamic therapy for depression have not distinguished either treatment as being more efficacious than the another (
Cuijpers, van Straten, van Oppen, & Andersson, 2008;
Leichsenring, 2001).manifested as decreased energy Additionally, studies comparing eight sessions of psychodynamic therapy to 16 sessions of CBT for depression have found comparable outcomes (
Barkham et al., 1996;
Shapiro et al., 1994). Continued investigation into psychodynamic treatments for depression is warranted.
This case study involved the treatment of an elderly woman named “Isabella” who developed depression secondary to her breast cancer diagnosis. Our objective was to evaluate whether PPP could be efficaciously administered in a specialized oncology clinic. Blood oxygenation level dependence (BOLD) was assessed with functional magnetic resonance imaging (fMRI) at pre- and post-treatment to examine brain activity prior to and following the intervention. This secondary aim was to pilot a recently developed scanner probe aimed at assessing brain activity thought to underscore benign response to pleasure and reward prior to and following PPP. Specifically, the question of interest was whether PPP would be associated with modified brain activity related to pleasure and reward. This probe was adapted from neuroimaging studies that listening to music elicits activity in brain regions thought to underscore pleasure (
Menon & Levitin, 2005;
Osuch et al., 2009).
In neuroimaging studies in which depressed individuals exhibited diminished response in brain regions involved in pleasure and reward processing (
Bardo, 1998;
Epstein et al., 2006;
Keedwell, Andrew, Williams, Brammer, & Phillips, 2005;
Pfaus, Damsma, Wenkstern, & Fibiger, 1995;
Schaefer, Putnam, Benca, & Davidson, 2006;
Schilström, Svensson, Svensson, & Nomikos, 1998;
Carelli, Ijames, & Crumling, 2000, Osuch, et al., 2009), common behavioral features of depression, such as increased avoidance and diminished experience and anticipation for reward and pleasure in daily activities (Dimidjian, Barrera, Martell, Munoz, & Lewinsohn, 2011;
Jacobson, Martell, Dimidjian, 2001;
Kasch, Rottenberg, Arnow & Gotlib, 2002) were also observed. Treatment outcome studies have begun to use functional neuroimaging to assess brain activity prior to and following treatment for depression (
Brody et al., 2001;
Fu et al., 2008;
Goldapple et al., 2004;
Kennedy et al., 2007;
Martin, Martin, Rai, Richardson, & Royall, 2001). Recently, Buchheim and colleagues (2012) assessed pre- and post-treatment changes in brain activity after 15 months of long-term psychodynamic psychotherapy. Their research design used images from the Adult Attachment Projective Picture System ([APPS]
George & West, 2001) during fMRI procedures to assess relational attachment patterns and observe functional brain changes that corresponded with treatment outcome. This study was highly innovative in that most prior clinical trials examining functional brain changes following psychotherapy have been criticized for not including functional tasks to probe brain activity (
Frewen, Dozois, & Lanius, 2008). Incorporating functional tasks during scanning is thought to elicit brain activity that underscores phenomenological experiences associated with psychiatric disorders and may bear relevance to understanding whether psychological interventions affect theoretically relevant brain regions. Among individuals with depression, for example, it would be especially fruitful to investigate whether psychotherapy affects brain regions typically associated with environmental reward, pleasure, and depressive affect. (e.g., cingulate cortex, nucleus accumbens, caudate, orbital, medial, and dorsolateral prefrontal regions, and ventral striatum;
Mayberg 2003,
2006;
Osuch et al., 2009). Consequently, we hypothesized that following successful PPP we would observe increased brain activation in response to a pleasurable music listening paradigm in regions such as the ventral striatum (nucleus accumbens), medial orbital (moPFC) and dorsolateral prefrontal cortex (dlPFC). We also anticipated that, following treatment, reduced activity would be observed in the subgenual cingulate cortex and expected all changes in activation to correspond to reduction in depression.
Background Information
The patient in this study, hereafter referred to as “Isabella.” was in her 60s, right-handed, married, and Caucasian, with four years of graduate-level education. She was diagnosed with breast cancer two years prior to her pretreatment evaluation. She received cancer treatment in the form of a left radical mastectomy (followed by six months of chemotherapy) approximately two years prior to PPP. She also received hormone treatment (i.e., tamoxifen) for breast cancer from one year prior to PPP through midway through psychotherapy. Her medication regimen for cholesterol, hypertension, edema, allergies, asthma, and breast cancer was consistent throughout treatment with the exception of tamoxifen. Her depression emerged in conjunction with her cancer diagnosis and her husband suffering a stroke, approximately two years prior to PPP treatment. The depression was manifested as decreased energy, fatigue, listlessness, agitation, and “feeling like a failure”. At the time of her pretreatment evaluation she was diagnosed with major depressive disorder using the Anxiety Disorders Interview Schedule-IV ([ADIS-IV]
Brown, Di Nardo, & Barlow, 1994). All sessions were provided on an outpatient basis at a cancer institute within a university medical center. An advanced male clinical psychology doctoral student with extensive training in psychodynamic psychotherapy conducted treatment. Informed consent was obtained, as approved by the University Institutional Review Board.
Baseline Assessment
Isabella was recruited in the context of a clinical trial at a university-based medical center cancer institute, which was examining the efficacy of behavioral activation and problem-solving therapy for patients with breast cancer who were also depressed (
Hopko et al., 2011). Pragmatic psychodynamic psychotherapy treatment and fMRI were used with this patient in a pilot study examining feasibility of treatment implementation and efficacy of PPP for major depression. As indicated by the ADIS-IV assessment, major depressive disorder was her primary diagnosis. Prior to (and throughout) treatment, Isabella completed the Beck Depression Inventory-II [(BDI-II]
Beck, Steer, & Brown, 1996). Isabella’s pretreatment BDI-II score was 31, indicative of severe depression. Her pretreatment Beck Anxiety Inventory ([BAI];
Beck & Steer, 1993) was 5, reflecting minimal somatic anxiety. The Environmental Reward Observation Scale ([EROS];
Armento & Hopko, 2007) assesses environmental reward (range = 0–40), with Isabella reporting moderate (18) environmental reward at pretreatment. This measure also was completed following each therapy session. Isabella also completed the Multidimensional Scale of Perceived Social Support ([MSPSS] range = 12–84; Zimet, Dahlem, Zimet, & Farley, 1988), with higher scores reflecting poorer social support from family, friends, and significant others. She had an MSPSS baseline score of 41, which reflected low to moderate perceived social support. The Hamilton Rating Scale for Depression ([HRSD]
Hamilton, 1960) was also completed at baseline by an advanced clinical psychology doctoral student. Isabella had a baseline HRSD score of 21, reflecting moderate depression.
In addition to self-report measures, Isabella participated in functional brain imaging scans one week prior to therapy and one week following completion of therapy on a 1.5-T Siemens MRI scanner located at the University Medical Center’s Department of Radiology. All data preprocessing and analyses were done with Statistical Parametric Mapping (SPM8; Wellcome Department of Cognitive Neurology, London, United Kingdom) with parameters, methodology, and correction for multiple comparisons outlined in
Gawrysiak et al. (2012). During her scans, Isabella listened to alternating passages of music, which were selected based on Isabella’s preference and identified as “preferred” and “neutral” ([based on a previously established coding system]
Gawrysiak et al., 2012;
Osuch et al., 2009). Pretreatment brain response was assessed by comparing the images acquired as Isabella listened to preferred and neutral music conditions. No differences were found in any brain region (
p > 0.05 uncorrected), suggesting that brain response did not respond differentially to preferred or neutral music in any way. Brain response—to confirm activation in the auditory cortex, as would be expected during music listening task—was also examined while the patient listened to music or was at rest. This contrast demonstrated bilateral activations in auditory cortex, suggesting that our music condition elicited brain response that reflected auditory stimulation in manner that supported our paradigm.
Evaluating Outcome
Isabella achieved full remission of her depression and exhibited clinically relevant reductions in symptoms following treatment, which was consistent with reliable change indices (
Jacobson & Truax, 1991) established through treatment outcome research with depressed breast cancer patients (
Hopko et al., 2011). Her BDI-II was reduced from 31 to 3 and her HRSD from 21 to 3, both measures reflecting a reduction from moderate-severe to minimal depression. Consistent with behavioral models of depression that highlight increased environmental reward as inversely associated with depression severity (
Hopko, Lejuez, Ruggiero, & Eifert, 2003;
Lewinsohn, 1974;
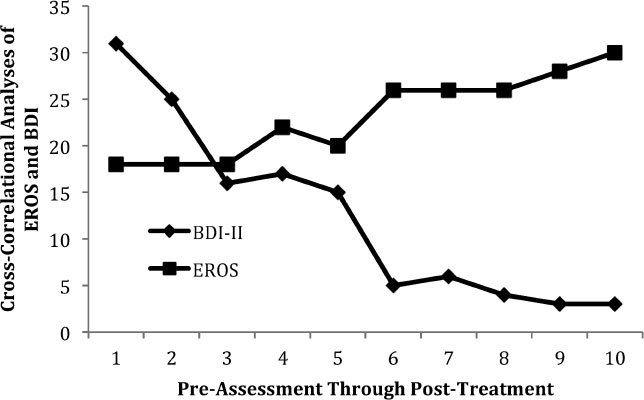
Martell, Addis, & Jacobson, 2001), Isabella’s EROS increased from 18 to 30. Her BAI decreased from 5 to 2, reflecting minor reductions in anxiety, and her MSPSS decreased from 41 to 34, reflecting increased social support. To more finely assess the relation of decreased depression and increased environmental reward assessed weekly throughout therapy, a cross-correlation analysis (CCA) was conducted using the Simulation Modeling Analysis software ([SMA]
Borckardt, 2008). CCA determines the degree that two measures relate to one another at a specified interval of time. For Isabella, the two measures most highly correlated at lag 0 (
r =–0.90,
p = 0.001; see
Figure 1), indicating that BDI-II scores were most strongly related to EROS scores on a session-by-session basis. Accordingly, self-reported environmental reward systematically increased as depression attenuated.
There were no differences in BOLD response to preferred versus neutral music passages pre-treatment (
p > .05). The BOLD response to preferred music relative to neutral was compared between pre- and post-treatment scans, restricted to our regions of interest (cingulate cortex, nucleus accumbens, caudate, orbital, medial, and dorsolateral prefrontal regions, and ventral striatum). This contrast indicated significant differences in regions of the ventral-medial prefrontal cortex ([vmPFC]
p = .023, cluster-level uncorrected) and near significant differences in the left orbital prefrontal cortex ([oPFC]
p = .091, cluster-level uncorrected). Posttreatment, vmPFC and left oPFC BOLD response was higher for preferred relative to neutral music (
p = .000 and
p = .036 respectively, corrected for multiple comparisons at the cluster level;
Figure 2).
Indeed, the vmPFC and oPFC have been implicated in conscious regulation of emotional states (
Phillips, Drevets, Rauch, & Lane, 2003), have been shown to correlate with pleasure ratings of music, and have been observed to distinguish depressed and healthy individuals during music listening tasks (Elliott, Agnew & Deakin, 2008;
Osuch et al., 2009). These regions have also been implicated in relative, rather than absolute, reward (Elliott, Agnew & Deakin, 2008) and are frequently included in neurobiological models of depression (
Mayberg, 2003,
2006). Consequently, these posttreatment fMRI findings are conceptualized as a plausible neurophysiological reflection of Isabella’s adaptive development and increased capacity for reward and pleasure. However, given the single subject design, these findings must be interpreted with caution.
Discussion
This study assessed changes in depression symptom severity following eight sessions of PPP (
Summers & Barber, 2010) and utilized functional neuroimaging as a proposed method to gauge treatment outcome and associated mechanisms of change. Findings suggest that PPP not only reduced depression, but may also promote improved quality of life and greater capacity to experience pleasure in daily activities and behaviors. The patient demonstrated favorable responses to treatment as reflected by clinically significant changes on clinician and self-report measures of depression, and she was deemed to have achieved full depression remission. This pilot case study provides preliminary support for the efficacy of PPP as a treatment for depression, and suggests feasibility and patient tolerability of PPP as a treatment in a medical facility context. Given that PPP is a structured intervention allowing for ideographic case conceptualization, it may represent a treatment approach capable of addressing a broad spectrum of depression manifestations triggered in part by coexistent medical problems. This hypothesis warrants future investigation. In this case self-reported depression and environmental reward exhibited a direct inverse relationship. The CCA indicated depression attenuation was associated with increased environmental reward reflected in session-by-session measurements. Accordingly, these data suggest that more frequent experiences of pleasure and reward in daily experiences correspond with reductions of depression. While it is highly speculative, this observation may reflect Isabella’s increased capacity to draw upon inner strength and resources that allowed her to live life with a greater sense of freedom, optimism, and purpose (
Shedler, 2010).
Although this case study is restricted in the generalizability of observations of functional brain activity, important implications are noteworthy. First, few examinations of functional brain activity both prior to and after treatment for depression have incorporated a functional task during scanning. Future randomized clinical trials may benefit from using similar music-listening probes to examine brain activity associated with reward responsiveness and pleasure as it relates to psychotherapy and depression. Second, this study demonstrates a novel model for evaluating meaningful life change that is not arbitrary. A criticism of the process of establishing empirical support for psychotherapies is that measures of symptom change may be arbitrary in terms of evaluating the extent to which therapy contributes to meaningful life improvement (
Westen, Novotny, Thompson-Brenner, 2004;
Kazdin, 2006). Simple measures of symptom reduction provide little information to guide clinicians in best practices as most therapies tend to reduce symptoms (i.e., the “dodo bird effect”:
Lurborsky, 1975). Assessing benign neurophysiological responses to pleasure, in conjunction with self-report indices of reward and depression, may serve as plausible and more valid means of evaluating treatment response as these approaches of measuring symptom change reliably reflect capacity for pleasure and happiness.
Important study limitations are noteworthy. First, treatment was not independently evaluated to assess therapist competence or general adherence to the PPP model. A systematic strategy of assessing treatment adherence may be difficult to develop due to the flexible and adaptive nature of PPP. Special consideration may be required by researchers examining the efficacy of PPP in larger randomized clinical trials. Second, although treatment was associated with reduced depression, data may be confounded by Isabella’s discontinuation of tamoxifen, a hormone treatment for breast cancer that has depression listed as a adverse effect for 15% of women (
Demissie, Silliman, & Lash, 2001). Third, while the vmPFC and oPFC were noteworthy functional brain activations in this patient at posttreatment, future investigations with larger sample sizes and more heterogeneous patient samples are needed to replicate this finding. Despite these limitations, preliminary results support that time-limited psychodynamic therapy may be efficacious for depressed breast cancer patients in an oncology clinic. Controlled clinical trials for patients who are depressed and have breast cancer are limited and the efficacy of psychodynamic approaches for this population is virtually unstudied (
Fann et al., 2008;
Hopko et al., 2011). Such work is clearly warranted. Finally, addressing potential problems associated with arbitrary metrics in treatment outcome research (
Kazdin, 2006), the combined approach of synthesizing weekly assessment measures, pre-post treatment clinician and patient measures, and corresponding functional neuroimaging assessment may yield data that is highly innovative, more integrative, and potentially more valid then traditional assessment strategies.
Acknowledgments
Acknowledgement:
The authors would also like to express their great gratitude to the Cole family, the Cole Neuroscience Center, and the Cole Gift Fund for their generous financial support. This paper was prepared with the support of the VISN 4 Mental Illness Research, Education, and Clinical Center, Philadelphia Veterans Affairs Medical Center, Philadelphia, PA. The paper does not necessarily represent the position or opinions of the Department of Veterans Affairs, and those opinions expressed are the sole responsibility of the authors. The authors would like to acknowledge and thank Stevie N. Grassetti for her helpful comments and contributions towards this article.