Procedural learning may be defined as the development of skills in which the strategy of execution cannot be explicitly described (learning by doing), such as bicycling or hand writing. Cognitive or motor procedural skills are learned progressively with practice until the optimal performance is automatized.
1 This type of learning is usually assessed with tasks requiring unfamiliar motor or cognitive skills. For instance, a subject may be asked to learn and replicate an arbitrarily defined visuospatial sequence or to track a moving target along a predictable path. Results on such tests were first reported to be abnormal in neurological illnesses involving the striatum in diseases such as Huntington's disease (HD) or Parkinson’s disease (PD).
2,3,4 The striatum was then suggested to be a critical structure involved in procedural learning. Other cerebral structures such as the cerebellum, the prefrontal cortex, and the supplementary motor area have also been found to be involved, although their specific contributions vary considerably depending on the different learning stages examined and on the specific cognitive or motor processes required in the task.
5,6 For instance, PET studies performed on normal subjects have shown that frontal structures are activated during the initial or adaptation stages of a sensorimotor procedural acquisition, while striatal and cerebellar structures are active throughout the learning process.
5Previous studies have concluded that patients with schizophrenia are able to learn new cognitive or motor procedures,
7,8,9,10,11 although, qualitatively, the learning profile in these patients appears to differ from that of normal subjects. In particular, low learning rates,
7,12 and large inter-trial fluctuations
13,14,15 characterize the procedural learning of patients with schizophrenia. These abnormal procedural learning profiles have been suggested to result from a neuroleptic-induced dysfunction of the striatum
8,12,13,14 or of the frontal cortex. In normal subjects, acute administration of classical neuroleptics such as chlorpromazine has been found to induce procedural learning deficits.
16,17 Although a dopamine-blocking effect is suspected in these studies, there was no direct evidence of such a mechanism, and the specific contribution of the frontal cortex and the striatum remains unresolved.
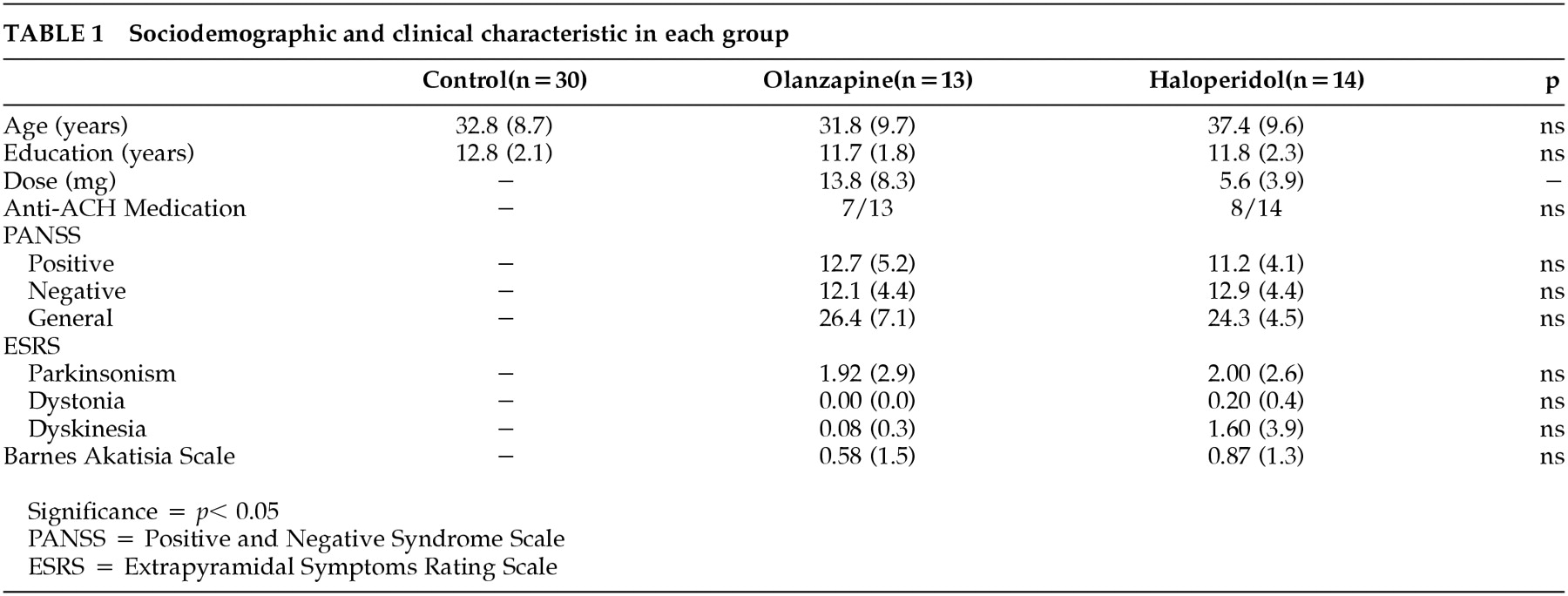
Patients with schizophrenia have previously been found to exhibit different learning profiles, depending on whether they were treated with a classical or an atypical neuroleptic.
13,14 Clozapine- or risperidone-treated patients show normal progressive learning from trial to trial, while patients treated with haloperidol have significant learning difficulties, characterized by a poor improvement and many fluctuations from trial to trial. These results were attributed to the differential pharmacological effects of these drugs on the striatum, presumably the different dopamine (D
2) receptor affinities, although there are actually no direct results supporting such a view. The present study was an attempt to further investigate this question by comparing the effects of a classical neuroleptic (haloperidol) and an atypical neuroleptic (olanzapine), on both a procedural learning task and on the striatal D
2 receptor occupancy. The two hypotheses were: 1) given its effect on the striatum, the conventional neuroleptic should be more deleterious on the procedural learning than the atypical neuroleptic and 2) this deleterious effect should be related to the striatal D
2 receptor occupancy, as assessed by Single Photon Emission Computed Tomography (SPECT) with
123I-IBZM.
DISCUSSION
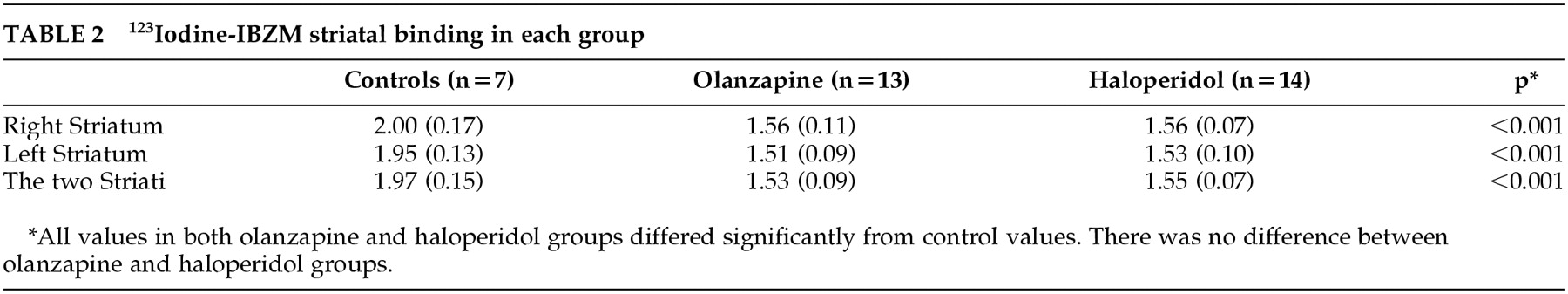
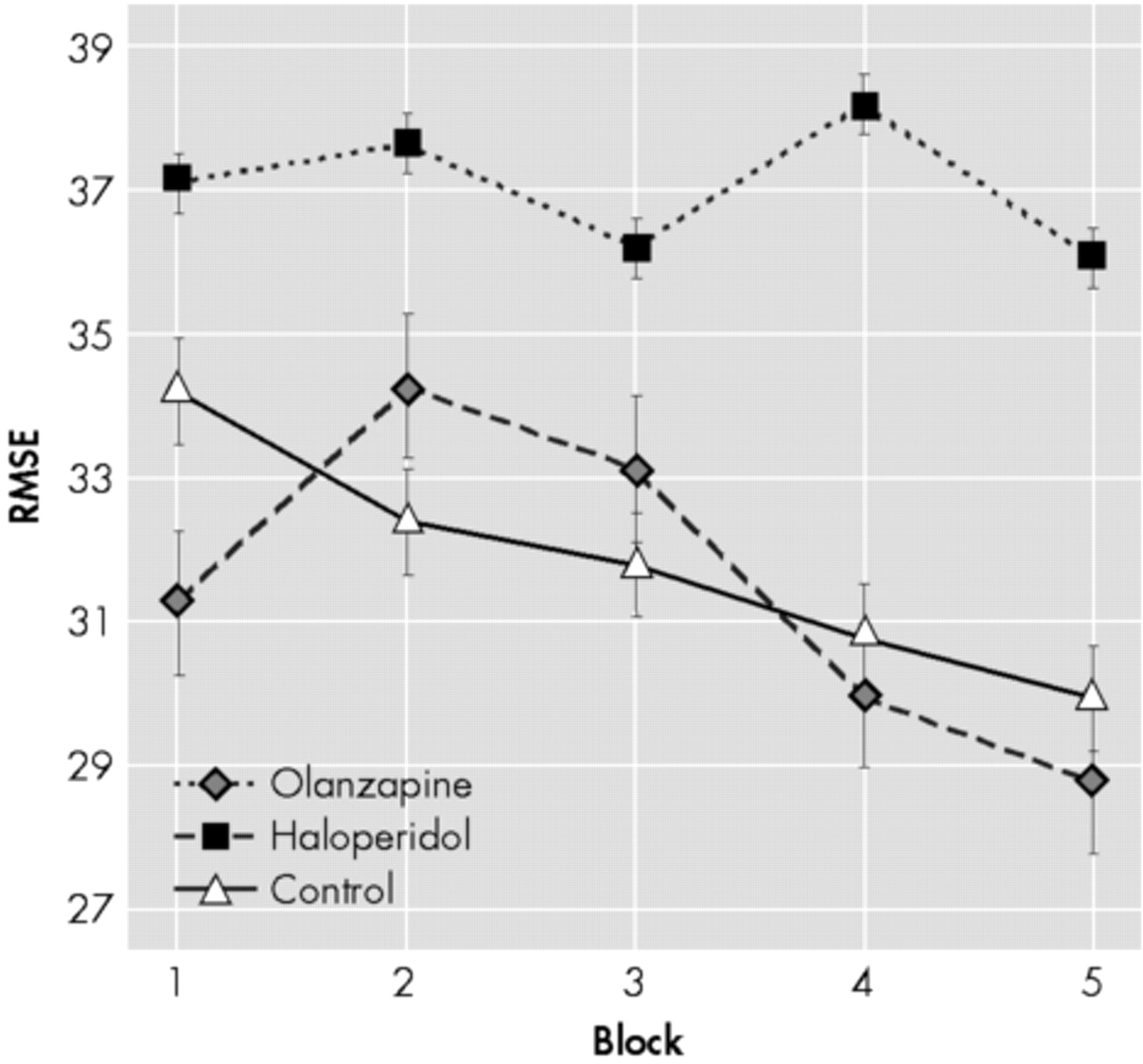
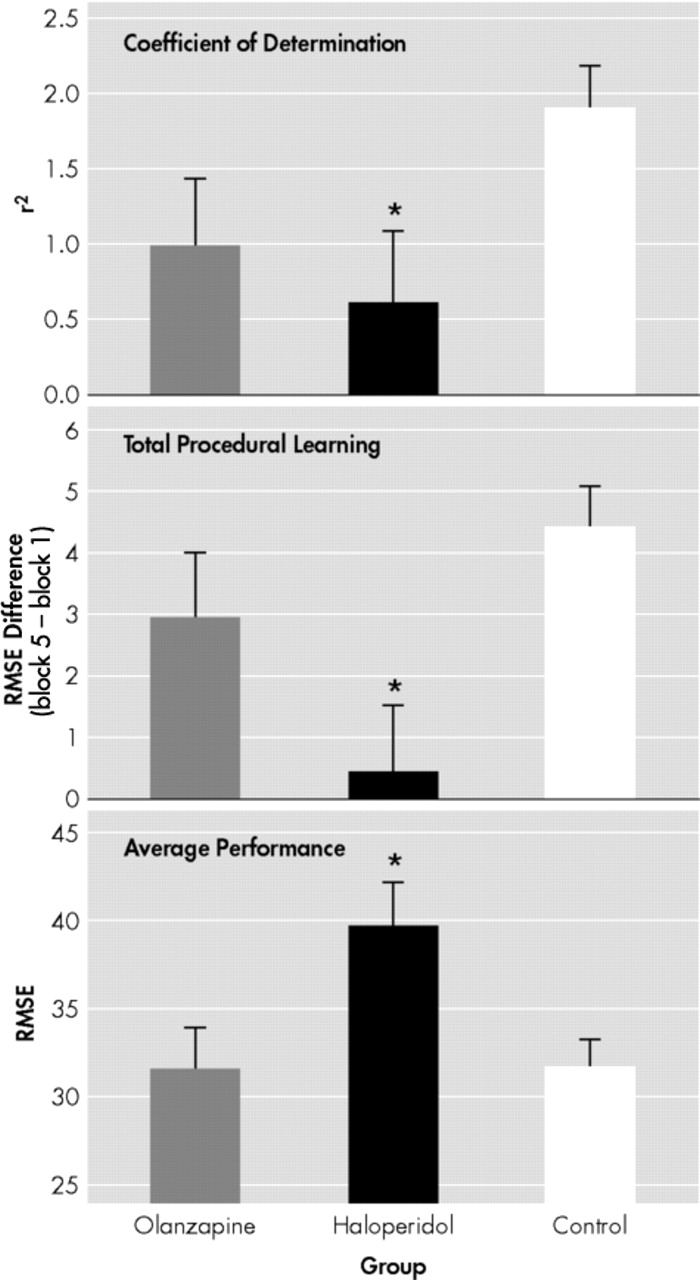
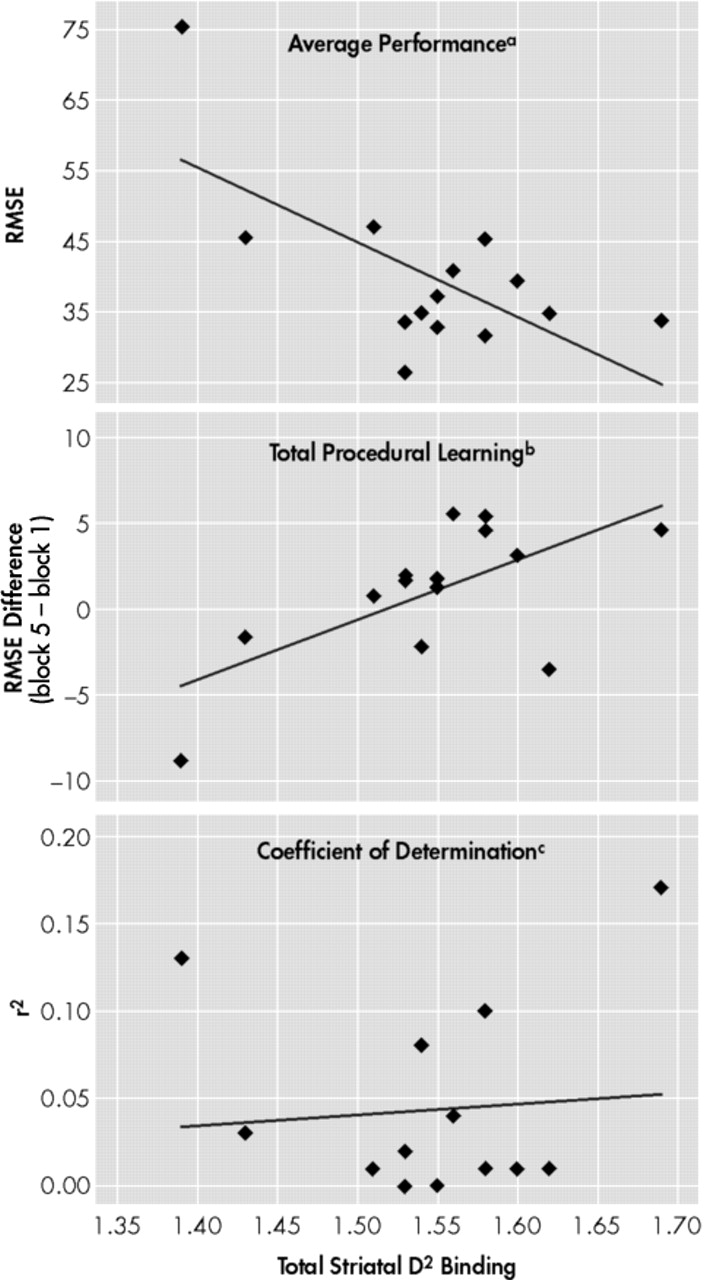
The results obtained in this study indicate that patients with schizophrenia may show procedural learning disturbances, depending on their neuroleptic medication. Patients treated with olanzapine performed as well as normal controls, while those treated with haloperidol showed procedural learning deficits affecting both quantitative (total learning, average performance) and qualitative (learning smoothness) aspects of learning. The procedural learning deficits observed in the haloperidol treated patients were related to both the dose and the level of D2 receptor occupancy in the striatum. Furthermore, the mediator analyses revealed that the striatal D2 receptor occupancy is probably the mechanism by which haloperidol induces procedural learning deficits. No correlation was found between the procedural learning deficits and the social or clinical variables, including age, education, disease duration, or any of the clinical symptom scales. These social and clinical variables were not found to differ between the olanzapine and haloperidol groups. Therefore, it appears that haloperidol, but not olanzapine, induces procedural learning deficits in patients with schizophrenia, and that this effect is related to the dopaminergic blockade in the striatum.
The view that procedural learning deficits are related to striatal dopaminergic process is in accordance with other studies. Using healthy volunteers, Kumari et al.
28 showed that procedural learning, as measured by a target-tracking task, may improve or worsen, depending on whether the subjects have received a dopamine agonist (such as amphetamine) or antagonist (such as haloperidol), respectively. In animals (rats), it has also been shown that the firing of the dopaminergic nigrostriatal fibers is specifically increased during the learning of a T-maze procedural task.
29 Furthermore, previous studies performed in patients with schizophrenia have found that conventional neuroleptics, such as haloperidol, are more deleterious on procedural learning than atypical neuroleptics, such as clozapine.
13,30 In general, the deleterious effect was hypothesized to be related to the higher striatal D
2 receptor occupancy of the conventional neuroleptics, compared with the atypical neuroleptics.
19,31,32 However, this view may not explain the result of the present study which shows no procedural learning deficit in the olanzapine-treated patients, in spite of a D
2 receptor occupancy as high as in the haloperidol-treated patients.
The fact that the striatal D
2 receptor occupancy, as measured by
123I-IBZM, did not differ between groups may appear surprising given that previous studies have shown that haloperidol possesses much greater affinity for D
2 receptors than does olanzapine.
32,34,35 Using
123I-IBZM single photon emission tomography (SPET), Pilowsky et al.
34 reported that the striatal D
2 occupancy of olanzapine is similar to that of clozapine, and much lower than that of typical neuroleptics, including haloperidol. However, this study had several limitations, including the small sample size of olanzapine-treated patients (
n = 6), non-randomized dose assignment, and the different duration of olanzapine treatment among participants before SPECT scanning. Furthermore, the study found no relationship between the dose administered and D
2 receptor occupancy, which is inconsistent with the literature on olanzapine,
36,37,38,39 and leads one to question the internal consistency of the results.
31 In another study, Tauscher and his group
35 have shown that, at therapeutic doses, striatal D
2 receptor occupancy is lower for olanzapine-treated patients (75%) than for haloperidol-treated patients (84%). However, despite significant group differences for striatal D
2 receptor occupancy, these authors argue that the observed difference might have been confounded by the high daily dose of haloperidol used in their study (mean value of 13 mg for haloperidol and 18 mg for olanzapine). Kapur and Seeman
40 also criticize the fact that clinical trials generally use high doses of typical antipsychotics (i.e., 10–20 mg/day of haloperidol), which give rise to D
2 occupancy of greater than 90%; this compares to doses of atypical antipsychotics that give rise to D
2 receptor occupancy of less than 80%. In fact, clinical studies with haloperidol do not suggest any advantage for dosages exceeding 5 mg/day.
41 Low doses (1–5 mg/day) of haloperidol induce 60%–80% dopamine D
2 receptor occupancy, which seems to be adequate to produce a clinically efficient neuroleptic response.
42 As for olanzapine, Kapur and his group
31 argued that at clinical doses this neuroleptic occupies more than 60% of D
2 receptors and, if used at doses above 20 mg/day, occupies more than 80% of dopamine D
2 receptors. They concluded that D
2 receptor occupancy with olanzapine is greater than that of clozapine, and comparable to that of typical neuroleptics.
31 This is consistent with our results. In our study, doses of haloperidol (5.6 mg/day) remain within the low therapeutic range, whereas olanzapine doses (13.8 mg/day) were slightly over the recommended range for that age group (10–12mg/day). This may explain why the haloperidol- and olanzapine-treated patients had equivalent striatal D
2 receptor occupancy.
Even if doses and binding considerations might explain the absence of difference between the two drugs D
2 receptors occupancy, this does not explain why patients treated with olanzapine had no procedural learning disturbances, while those treated with haloperidol had noticeable deficits. Olanzapine may have some protective properties against its D
2 affinity. One possibility is that the anticholinergic properties of olanzapine may modulate its own D
2-blocking effect within the striatum.
43 It is well known that substances with anticholinergic properties may have a protective effect against D
2-antagonist-induced striatal dysfunction. This constitutes the theoretical premise for administering anticholinergic substances to reduce neuroleptic-induced extrapyramidal symptoms in schizophrenia. On the other hand, patients treated with haloperidol in the present study received concurrent anticholinergic medication, and this has not prevented their procedural learning deficits. In addition, clinical scales of extrapyramidal symptoms were not different between the haloperidol- and olanzapine-treated patients, suggesting that the anticholinergic treatment in the haloperidol group was sufficient to compensate for the extrapyramidal symptoms but not the procedural learning disturbances. In other words, the procedural learning tests may be more sensitive than the extrapyramidal symptom scale at detecting neuroleptic-induced striatal dysfunction.
Another explanation may be related to the relative 5-HT
2/D
2 receptor affinity of the medications. Greater serotonin 5-HT
2 than dopamine D
2 activity is a characteristic shared by atypical neuroleptics.
32,44,45,46,47 Olanzapine is classified as an atypical neuroleptic and possesses greater in vitro 5-HT
2 than D
2 receptor affinity.
32 Haloperidol is a highly selective dopaminergic D
2 receptor antagonist, and is devoid of 5-HT
2 receptor activities.
48 Higher 5-HT
2 than D
2 receptor affinity of neuroleptics has been found to be associated with a reduced incidence of extrapyramidal symptoms,
21 presumably due to a compensating effect of the 5-HT
2 blocking activity over the D
2 blocking activity in the basal ganglia. Although the mechanism underlying this effect is not fully understood, it may involve a 5-HT
2 blockade of the inhibiting serotoninergic terminals arising from the raphe and projecting to the dopaminergic terminals in the striatum.
19,31,46,49 This 5-HT
2 receptor blockade would increase the dopaminergic firing, thereby compensating for the D
2 receptor blockade. It has been suggested previously
18 that, at therapeutic doses, atypical neuroleptics do not reach a critical receptor occupancy in the nigrostriatal system (presumably associated with extrapyramidal symptoms and procedural learning deficits), while at these doses these substances may occupy significant 5-HT
2 receptors in the mesolimbic and mesocortical systems (presumably associated with schizophrenic symptoms). This would protect against striatal dysfunction when treating for psychotic symptoms. However, our finding of similar striatal binding of
123I-IBZM in the haloperidol and olanzapine groups does not favor this hypothesis.
It has also been suggested that the protective effect of olanzapine in the striatum is related to its high 5-HT
1A receptor agonist activity.
50 These somatodendritic serotonin autoreceptors have been found in high numbers in the raphe nuclei, where they modulate the firing of serotoninergic neurons. 5-HT
1A agonists like olanzapine inhibit the firing of serotoninergic neurons, once again increasing the level of dopamine in the striatum, which would have a compensating effect on its D
2 receptor occupancy. The absence of any such compensating effect in the haloperidol group would, in contrast, favor striatal dysfunctions and procedural learning disturbances.
Finally, differences in procedural learning in our two groups of patients could also be explained by social and clinical factors that influence the choice of an antipsychotic. Factors such as history of treatment response, history of recent relapse, or occupational functioning and history of success in the community may all influence the choice of a conventional or atypical antipsychotic in a given patient. This would indicate different clinical profiles that may not necessarily be detected with clinical scales (as those used in the present study), but that may indirectly contribute to the differences in procedural learning between groups.
CONCLUSION
The neuroleptics examined in this study have been shown to differently affect procedural learning. Olanzapine does not disturb procedural learning in patients with schizophrenia, while haloperidol produces a generalized procedural learning deficit. These results also show a primary role of the dopaminergic processes in procedural learning, as significant correlations between procedural learning deficits and striatal D2 receptor occupancy of 123I-IBZM have been observed. The high 5-HT2 relative to D2 receptor affinity of olanzapine may be responsible for its protective effect against the neuroleptic-induced striatal dysfunction. The anticholinergic and 5-HT1A agonist activity of olanzapine may also contribute to this differential effect. Given that procedural learning plays a crucial role in many daily activities, one should take the pharmacological profile of a neuroleptic into account before using it to treat schizophrenia, in order to avoid any skill learning deficiency.