Participants and recruitment
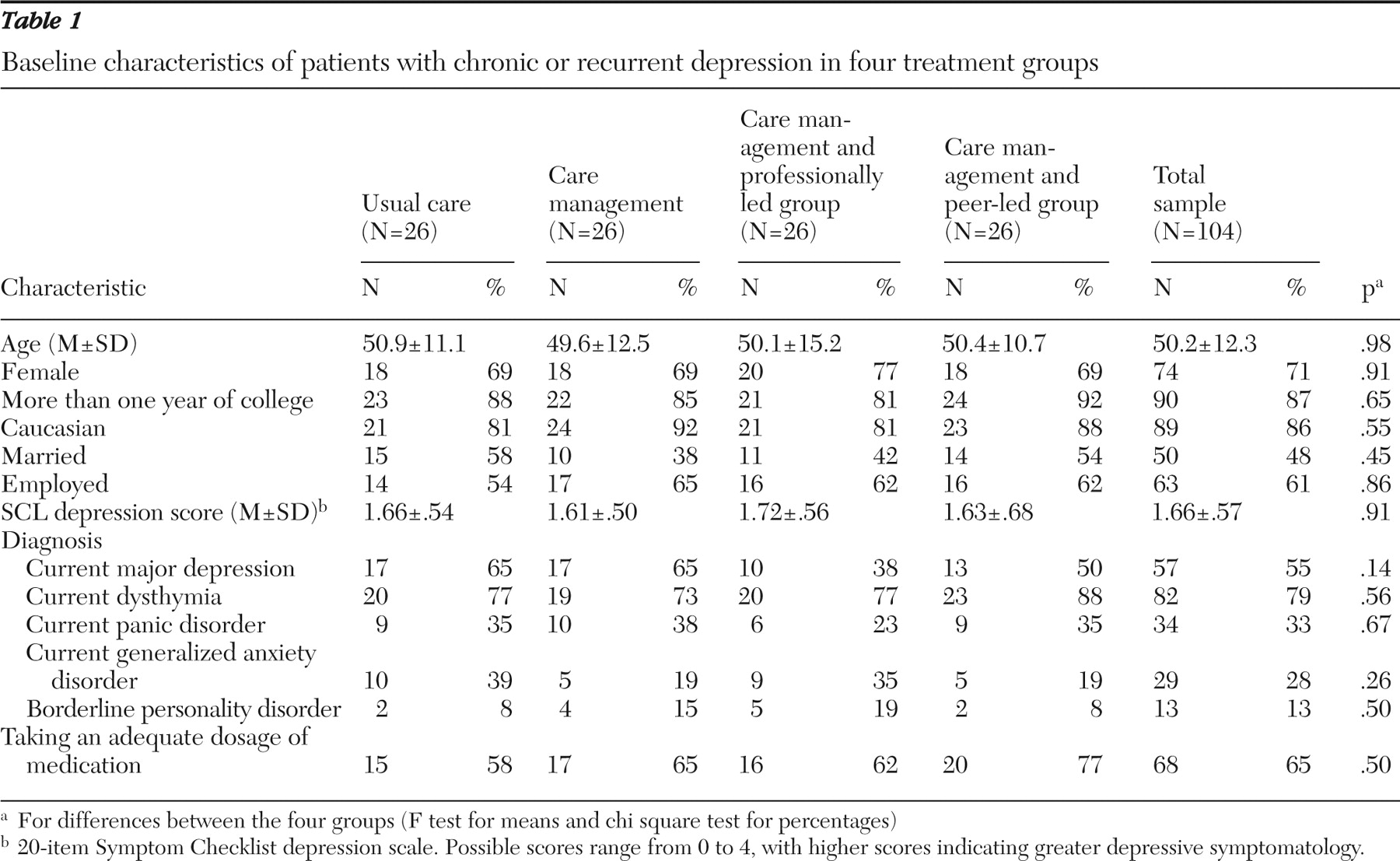
Study participants were enrolled between December 2003 and May 2004. Follow-up data were collected until July of 2005. Participants were recruited from the Central Behavioral Health Clinic of Group Health Cooperative (GHC), a health maintenance organization that serves approximately 500,000 individuals in Washington State. The clinic serves about 8,500 patients per year, of whom approximately 1,700 (20%) are treated for chronic or persistent depression.
We identified patients who had persistent symptoms of depression despite at least six months of antidepressant treatment prescribed in specialty care. Computerized data systems were used to identify GHC members aged 18 and older who had initiated antidepressant treatment at least 180 days previously, had a visit diagnosis of major depressive disorder at the time of initial antidepressant prescription, were continuously enrolled in GHC for at least the previous 180 days, and had no diagnosis of bipolar disorder or psychotic disorder or prescription for a mood stabilizer or antipsychotic medication in the past two years.
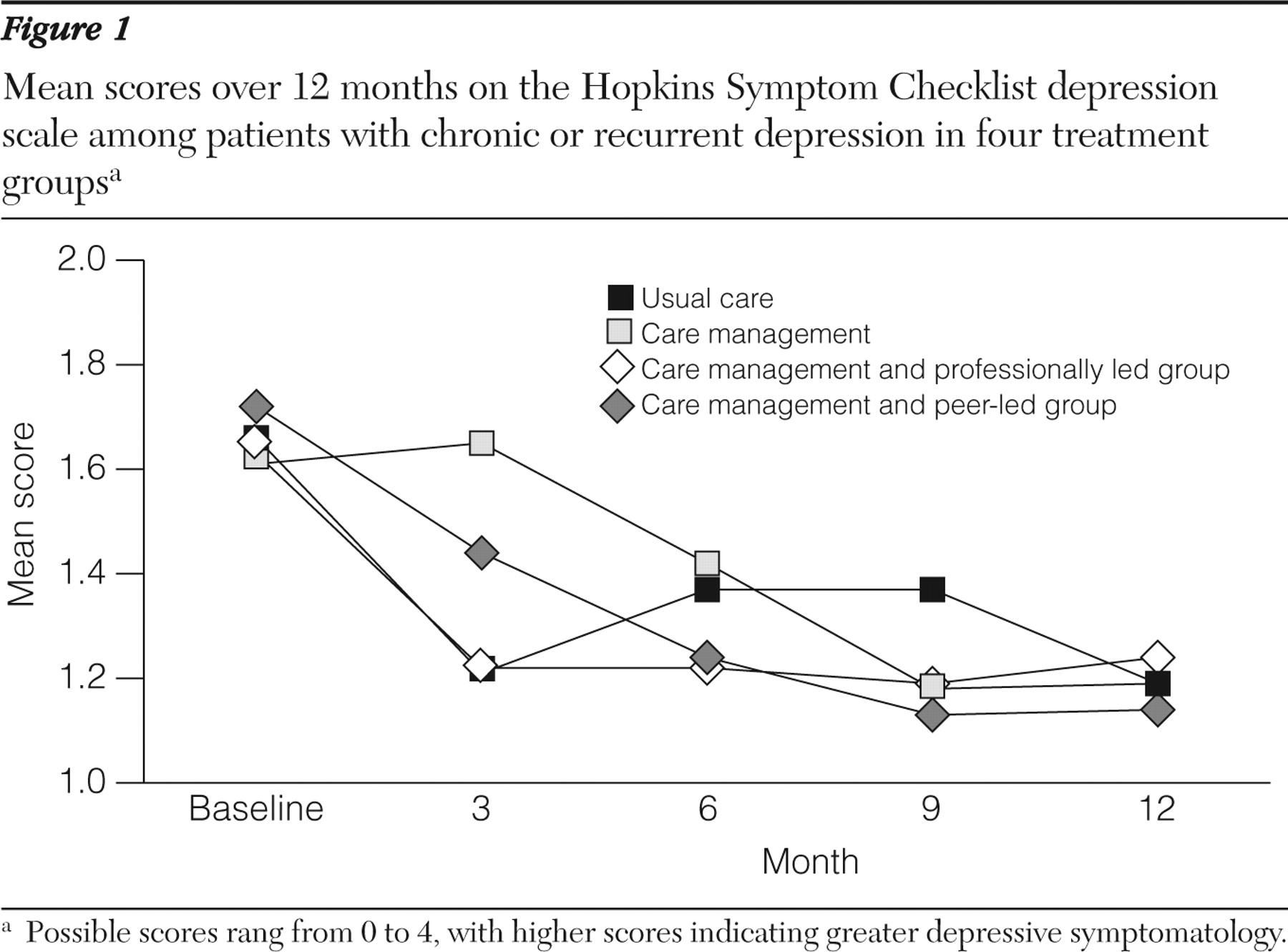
All eligible patients received an invitation letter that included a detailed study description and an option to decline further contact. A telephone call approximately seven days later included a 20-item depression scale extracted from the 90-item Hopkins Symptom Checklist (SCL-90) (
39 ). Patients who scored above .75 on the SCL-90, indicating significant current symptoms, were invited to complete an in-person baseline assessment. All invited participants were offered $20 compensation for completing the baseline assessment.
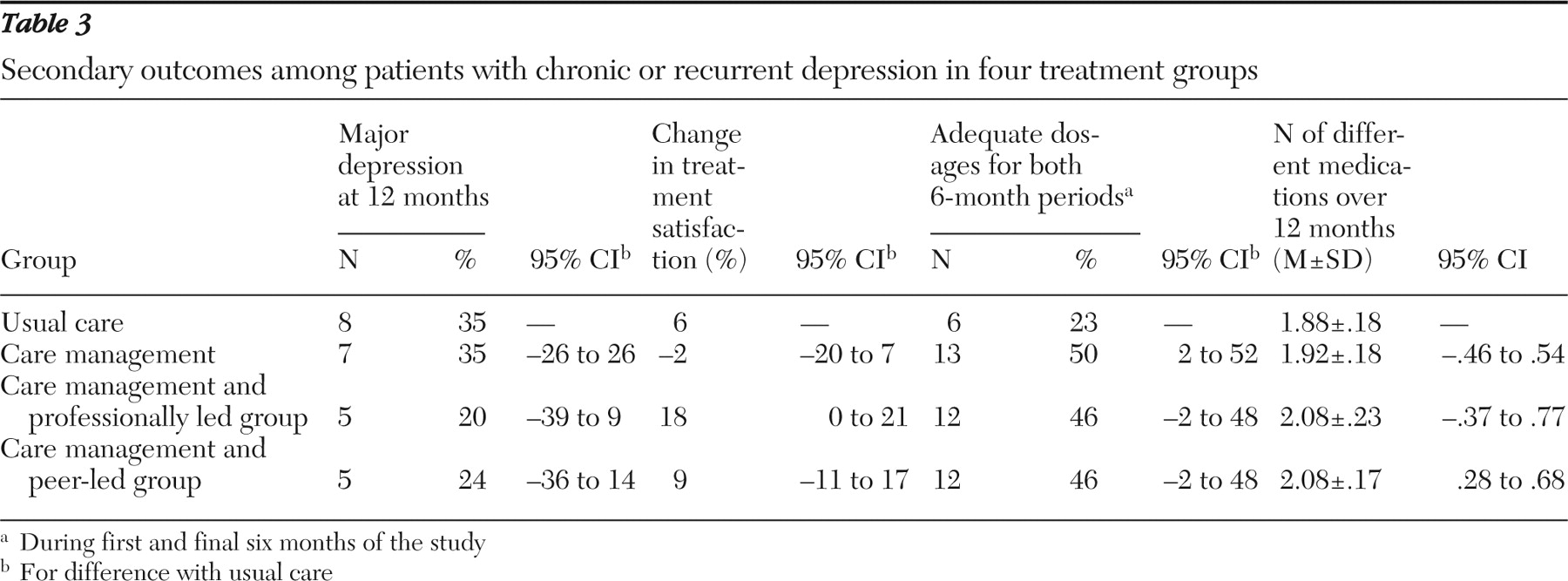
Inclusion criteria assessed at the baseline interview (see below) required at least one major depressive episode in the past two years as diagnosed by a structured interview and a history of either recurrent major depression (more than three episodes in the past five years) or dysthymia. In other words, all patients met criteria for recurrent major depression or dysthymia, but (consistent with our effectiveness design) patients were heterogeneous with respect to current mood state (dysthymia, chronic major depression, partial remission, relapse, or recurrence) and current antidepressant treatment. Exclusion criteria included history of mania or hypomania, cognitive impairment, near-terminal medical illness, intent to disenroll from GHC within the next 12 months, and emergent clinical needs (for example, risk of harm to self or others). Alcohol or drug use disorders were not exclusion criteria.
After a full description of study procedures, risks, and benefits, all participants provided written consent before the baseline assessment and again before enrollment in the randomized trial. Participants were advised that some would be offered additional treatment services, but willingness to accept any intervention was not a requirement for participation. All study procedures were reviewed and approved by GHC's Institutional Review Board.
Interventions
The chronic care model (
32,
33 ) guided the overall design of the intervention programs. Important components of this model include information systems to monitor treatment quality and treatment adherence, decision support through treatment algorithms and appropriate specialty consultation, practice redesign to ensure appropriate follow-up care, and patient education and activation to promote effective self-management. The telephone care management system was directed at the first three elements, and the two group programs were directed at the fourth. Patients in all groups were also free to use any nonstudy services normally available.
Telephone monitoring and care management. A computerized care manager decision support system supported systematic tracking of patient contacts, scripted clinical assessments, automatic application of treatment algorithms, and generation of feedback reports. The care manager (a master's-level counselor) contacted each patient at specified intervals—at least monthly during the first three months, then at intervals that varied according to symptoms, medication adherence, and side effects. During the first session the care manager helped each participant create a written care plan. Each contact included a structured five- to ten-minute assessment of depressive symptoms, medication use, and side effects. Following computer-assisted scripts, the care manager provided education about medication adherence and management of side effects and incorporated motivational enhancement strategies to address ambivalence about medication use when patients had discontinued medication treatment or were taking dosages lower than those prescribed. Information collected about symptoms, side effects, and current medication dosage generated specific recommendations to the patient's usual-care treating provider.
After each contact, the care manager sent the treating provider a report of current symptoms, medication use, side effects, prior treatment, and algorithm-based recommendations. In the case of moderate or severe symptoms (or if algorithm-generated recommendations suggested urgent intervention), the care manager communicated with the treating provider by telephone within 24 hours. The care manager also provided any needed outreach and care coordination, including facilitation of follow-up care.
Care management training included four hours of didactic training, four hours of role-play, and direct observation of two care management contacts before certification. The care manager received weekly supervision by the study psychologist and psychiatrist. Cases reviewed at supervision meetings included all patients overdue for monitoring calls, all patients with moderate or greater levels of depressive symptoms, and any cases requested for review by the care manager.
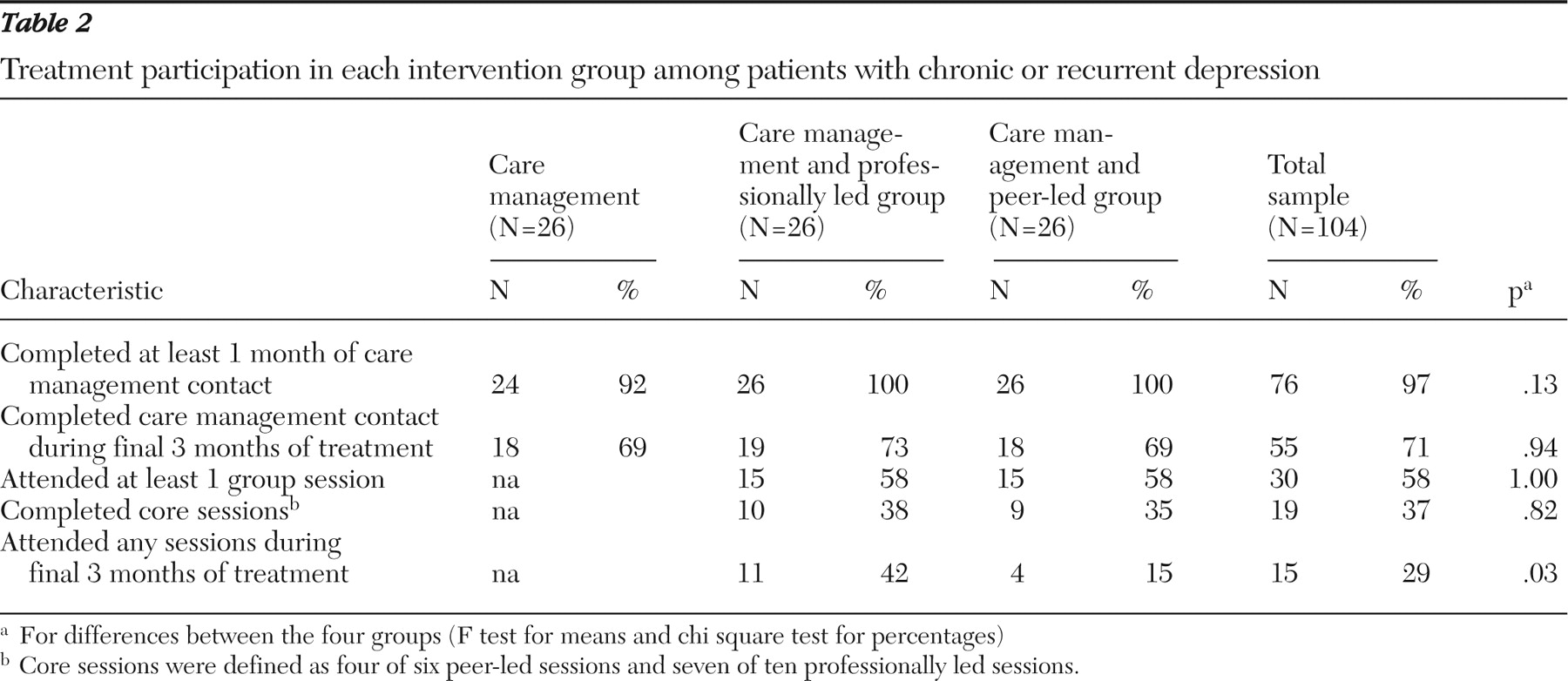
Peer-led chronic-disease self-management program. The peer-led program is an evidence-based program shown to relieve symptoms (such as pain and depression), reduce use of health services (
38,
43,
44,
45,
46,
47,
48 ), and reduce activity limitations (
49 ) across a range of chronic conditions. The six-week workshop includes several core components: disease-related goal setting and problem solving, cognitive symptom management (relaxation, distraction, self-talk, and visualization), communication skills, medication management, development of a patient-physician partnership, and use of community resources.
The program incorporates strategies that are based on self-efficacy theory (
50 ) and evidence that positive role models (that is, lay leaders with experience) increase patients' confidence for disease management. The chronic-disease self-management program aims to enhance self-efficacy for disease management by promoting self-directed application of newly acquired skills, reinterpreting symptoms as multidetermined and modifiable, modeling coping behaviors, and using guided rehearsal and social persuasion. The program follows a highly structured and detailed protocol that includes a structured method for group problem solving and weekly action planning. We supplemented the six-week workshop with ongoing bimonthly groups focused on continued goal setting and problem solving in order to reinforce skill mastery and problem-solving abilities. Each group had two leaders, and at least one leader had prior experience teaching the course. Peer leaders referred all clinical concerns to treating providers or the care manager.
Group leaders each attended a four-day training workshop that used an explicit training manual developed by the Stanford Patient Education Research Center (
51 ). The study psychologist provided ongoing biweekly supervision during the first six weeks of the group and as-needed supervision thereafter to the senior peer leader. The first six sessions of the program were audiotaped, and a continuation session was directly observed for quality assurance and treatment fidelity.
Professionally led group program. A psychologist delivered the manualized group intervention over ten consecutive weeks, followed by six months of twice-monthly "booster" sessions. Selection of specific intervention elements was informed by Jacobson and colleagues' (
52,
53 ) randomized trial demonstrating that therapy limited to behavioral activation and identification and interruption of negative automatic thoughts was as effective as "complete" cognitive-behavioral treatment (that is, treatment that includes exploration and modification of core schema).
Traditional acute-phase cognitive-behavioral therapy components were adapted to address persistent depression (
54 ) and emphasized setting reasonable goals, implementing lifestyle changes (such as regular aerobic exercise), enhancing medication adherence, increasing positive reinforcement (such as developing daily activity schedules), managing cognitive distortions, and using other behavioral strategies (such as social skills rehearsal). Session content explicitly addressed self-management, encouraging participants to identify and evaluate current and potential coping strategies (
55 ). For example, substance use and suicide attempts were discussed as potential but maladaptive strategies for coping with depression. Booster sessions emphasized self-directed goal setting and problem solving and attention to long-term self-care planning and provided sustained social support.
Continuation cognitive-behavioral therapy of this type delivered in an individual format has been shown to improve outcomes for patients with recurrent depression and incomplete recovery between episodes (
13 ). In contrast to some cognitive-behavioral therapy interventions designed as stand-alone interventions, the group program explicitly addressed medication adherence and effective collaboration with prescribing providers.
A psychologist (DM), who was trained in cognitive-behavioral therapy and who had eight years of experience treating depression, led the groups. The study psychologist provided ongoing weekly or biweekly supervision during the first ten weeks of the group program and as-needed supervision thereafter. All sessions were audiotaped for assessment of protocol adherence and quality assurance.
Usual care. Participants assigned to usual care were free to use any primary care or specialty services normally available inside or outside GHC. No additional services were provided, but no services normally available were withheld.