There are several reports regarding the use of vitamin B
6 in the treatment of the patients suffering from neuroleptic-induced movement disorders
(1–
4). With one exception
(1), the dose of vitamin B
6 was relatively low (100–500 mg/day), and the subjects experienced some benefit. The current study is the first report to our knowledge of a double-blind trial of vitamin B
6 in the treatment of tardive dyskinesia in patients with schizophrenia or schizoaffective disorder.
Method
Fifteen inpatients with schizophrenia or schizoaffective disorder (four men and 11 women) who had been receiving a stable psychotropic regimen for at least 1 month and who fulfilled diagnostic criteria for tardive dyskinesia were included in this study. The patients’ age range was 28–71 years. All were free of any concurrent medical or neurological disorders as well as evidence of substance or alcohol abuse. None had received vitamin treatment. Written informed consent and institutional review board ethics committee approval were obtained. All patients received the regular balanced hospital diet.
All patients received traditional or atypical neuroleptics; 11 received oral preparations, and four received injectable long-acting neuroleptics (mean dose=490 mg/day chlorpromazine equivalents). Twelve patients received combination therapy.
The study design was double-blind, with crossover and placebo control. Vitamin B
6 or placebo were added to the patients’ ongoing treatment for 4 weeks each and then crossed over after a 1-week washout period to allow for return to normal levels of vitamin B
6 (5).
Doses of all psychotropic medication were kept unchanged throughout the study. The dose of vitamin B6 was increased by 100 mg/week from 100 mg/day to 400 mg/day in twice-daily divided doses.
We used the parkinsonism, dystonia, and dyskinetic movement subscales of the Extrapyramidal Symptom Rating Scale
(6) to assess tardive dyskinesia. Assessments were taken at baseline and repeated every week by the same investigator at the same time of day in order to rule out any influence of diurnal fluctuation of the tardive dyskinesia symptoms
(7). The interrater reliability (kappa) in Extrapyramidal Symptom Rating Scale scores from baseline to week 4 was good (intraclass correlation coefficient=0.92). A 20% reduction in Extrapyramidal Symptom Rating Scale scores from baseline to week 4 was taken to represent no response, 21%–40% as minimal improvement, 41%–60% as moderate improvement, and more than 61% as marked improvement.
Plasma levels of vitamin B
6 were assessed at baseline and every other week by radioenzymatic assay of plasma pyridoxal-5′-phosphate (normal range=20–120 nmol/liter)
(8). The raters were kept blind to the results.
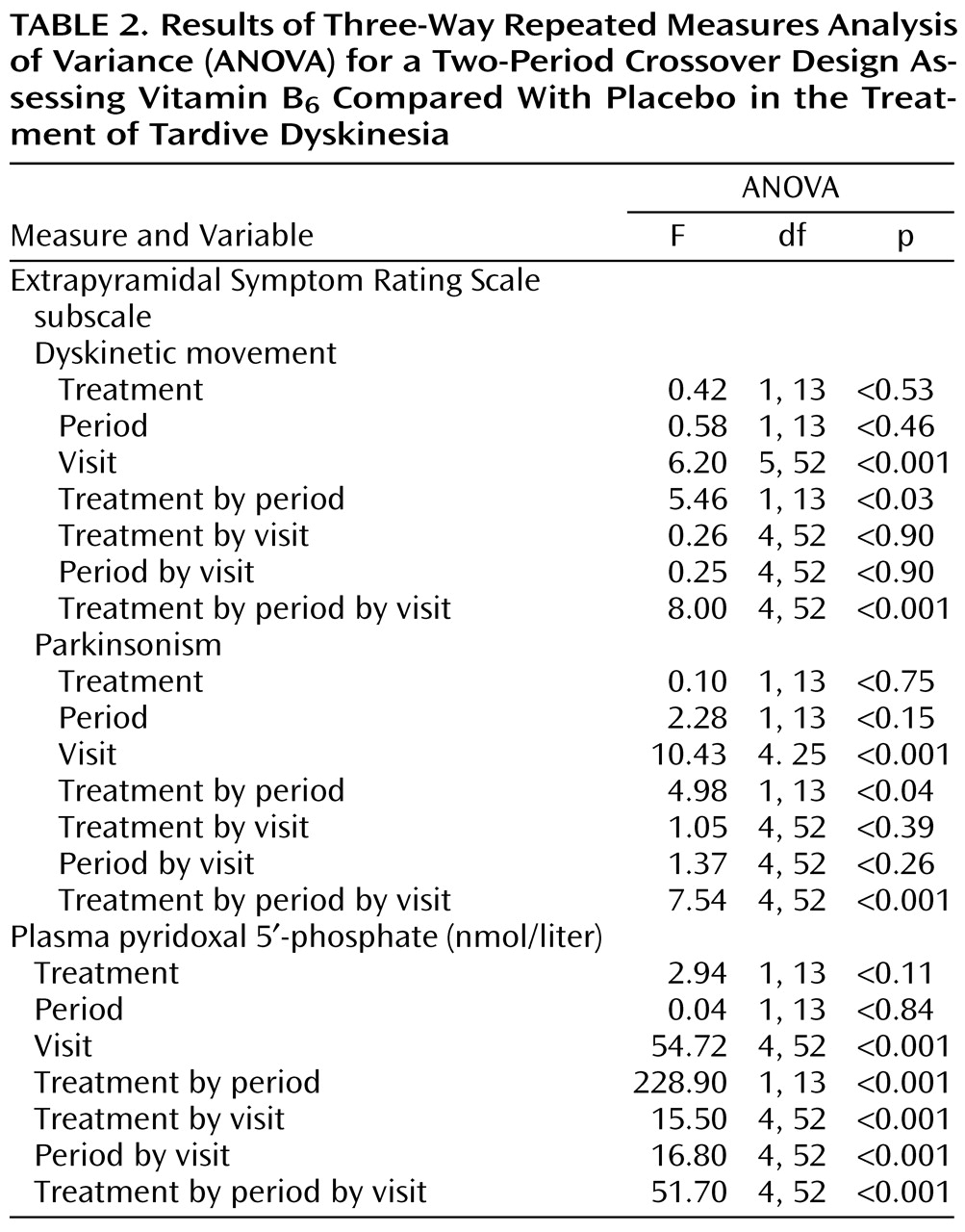
To compare effects in groups, in periods, and across all visits, variables were analyzed by three-way repeated measures analysis of variance (ANOVA) for a two-period crossover design. Post hoc comparisons employed the Scheffé post hoc test.
Results
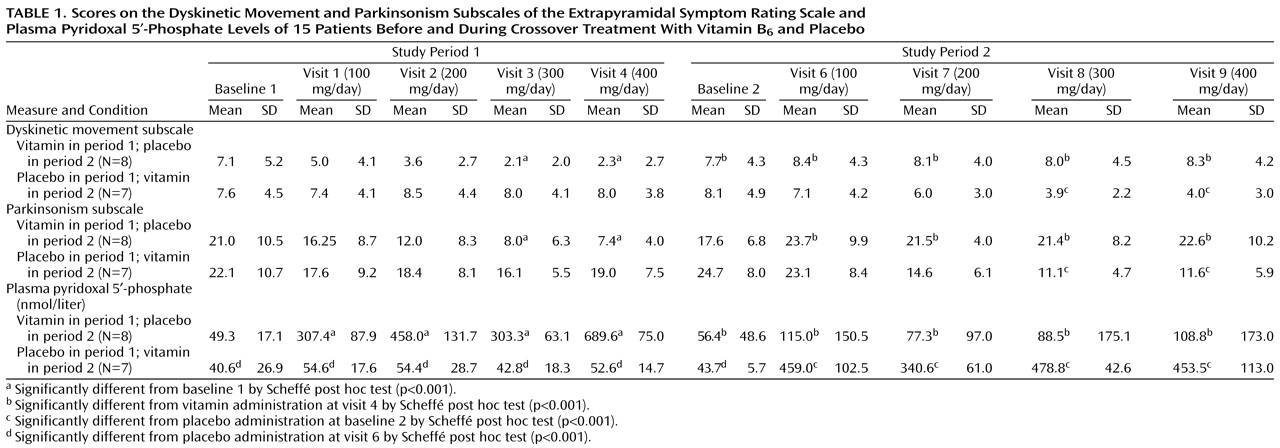
All patients received the maximal dose of 400 mg/day of vitamin B
6 without adverse effects. At both baselines, there were no significant differences between groups receiving placebo or the vitamin in dyskinetic movement subscale scores, parkinsonism subscale scores, or pyridoxal-5′-phosphate levels (
Table 1 and
Table 2).
Three-way repeated measures ANOVA for the two-period crossover design revealed significant differences between vitamin B6 and placebo groups in scores on the dyskinetic movement subscale in period 1 and period 2.
The improvements in dyskinetic movement subscale scores after 3 and 4 weeks of treatment with vitamin B6 in period 1 were 69.2% (SD=14.4%) and 68.6% (SD=23.0%). In period 2 the improvements were 38.2% (SD=46.1%) and 32.8% (SD=57.0%), respectively. (The percentage of improvement was calculated on the basis of individual scores rather than means for the entire group.)
In period 1, treatment with vitamin B6 caused a significant improvement in parkinsonism subscale scores, first observed in the third week of the treatment (dose of vitamin B6 was 300 mg/day) and continuing till the end of this phase of treatment. In period 2, vitamin B6 also caused a significant improvement in parkinsonism scores with vitamin B6 doses of 300 mg/day. The improvements on the parkinsonism subscale after 3 and 4 weeks of treatment with vitamin B6 in period 1 were 51.6% (SD=43.8%) and 57.1% (SD=30.1%) and in period 2 were 52.0% (SD=20.8%) and 51.6% (SD=22.8%), respectively.
After the washout period following vitamin treatment, the dyskinetic movement and parkinsonism subscale scores on the Extrapyramidal Symptom Rating Scale returned to baseline levels.
Baseline plasma pyridoxal-5′-phosphate levels were in the normal range in 11 patients (20–120 nmol/liter). In one patient, a higher pyridoxal-5′-phosphate level (163.5 nmol/liter) was found, and in four patients pyridoxal-5′-phosphate levels were below normal (5.0–16.2 nmol/liter). In both experiment periods, levels of vitamin B6 were significantly higher after the first week of treatment and continued till the end of the phase. However, we could not find a direct relationship between therapeutic response and pyridoxal-5′-phosphate level or changes in tardive dyskinesia.
Discussion
The results of this small study suggest that vitamin B6 is effective in treating tardive dyskinesia at doses from 300 mg/day. Vitamin B6 was effective during the treatment period only.
There have been several uncontrolled controversial reports about treatment of tardive dyskinesia with vitamin B
6 (1–
4,
9). Our results are consistent with previous reports suggesting the beneficial effect of pyridoxine on movement disorders
(1–
4).
We were not able to establish a correlation between pyridoxal-5′-phosphate levels and changes in two subscales of the Extrapyramidal Symptom Rating Scale. We assume, therefore, that serum pyridoxal-5′-phosphate is not directly associated with tardive dyskinesia and that serum levels of vitamin B6 do not correlate directly with pyridoxine-dependent activity of biogenic amines.
The mechanisms by which pyridoxine attenuates the symptoms of tardive dyskinesia are not completely understood. Pyridoxyl-5-PO4, derived from dietary pyridoxine, serves as a cofactor in the enzymatic decarboxylation of dopa to dopamine
(10) and other metabolic transformations, including γ-aminobutyric acid, serotonin, and melatonin
(11,
12).
One possible explanation for the effects observed in this study is that they are due to the antioxidant and free radical scavenger activities of vitamin B
6 (2,
13). Free radicals have been implicated in a variety of neuropsychiatric conditions, many of which are marked by the gradual development of psychopathologic symptoms and movement disorders. There is evidence that radical-induced damage may be important in tardive dyskinesia and, possibly, in schizophrenia as well
(14). Because vitamin B
6 takes part in almost all the possible mechanisms proposed to underlie tardive dyskinesia, it appears to make sense that it could alleviate symptoms of tardive dyskinesia.
To our knowledge, this is the first double-blind study examining the treatment of tardive dyskinesia with vitamin B
6. The potential usefulness of vitamin B
6 in treating tardive dyskinesia could be of clinical importance because it has no side effects in small doses
(15). Furthermore, to date there is no other effective treatment for this troublesome and sometimes incapacitating condition.
Our study group was rather small, and further studies should involve larger numbers of patients and a long-term study design. In retrospect, we feel that it would have been possible to limit the study to a single period, without the crossover phase, since the results from period 1 were statistically significant. Moreover, it is necessary to examine the effect of concomitant use of vitamin B6 and risperidone or olanzapine in patients with tardive dyskinesia.