It is generally acknowledged that schizophrenia has a multifactorial etiology, with multiple susceptibility genes interacting with environmental insults to yield a range of phenotypes in the schizophrenia spectrum. The field has moved to parse the underlying etiological components of schizophrenia using, for example, candidate gene, imaging, and cognitive science strategies that increasingly employ populations other than those with schizophrenia itself, including siblings or relatives of patients with schizophrenia, healthy volunteers, and subjects who are prodromal for schizophrenia and schizophrenia-related personality disorders. The cross-sectional study of the naturalistic variability of the schizophrenia spectrum, including schizotypal personality disorder, offers one unique and understudied vantage point from which to understand the schizophrenia disorders. Indeed, a strategy of investigating the similarities and differences between chronic schizophrenia and milder spectrum disorders can facilitate the dissociation of the pathophysiological mechanisms that are associated with the core cognitive and social impairments of this spectrum of disorders from those associated with recurrent or chronic psychosis and the extreme cognitive and social deficits of chronic schizophrenia. In this overview, we present studies that used comparable research paradigms to examine subjects with chronic schizophrenia, that have been reviewed more extensively in other articles, including meta-analyses, but are summarized here. We also discuss studies of subjects with schizotypal personality disorder, which while much more limited in number and sample size, serve to illustrate the potential power of a strategy that has received only limited attention to date in the field. We focus on studies of subjects selected to meet DSM criteria for schizotypal personality disorders rather than studies of relatives of schizophrenia probands or college student volunteers selected on the basis of self-report scales of schizotypy. While we acknowledge the limitations of data available from subjects with schizotypal personality disorder, we propose a theoretical model generating testable hypotheses to stimulate further investigation using this paradigm.
People with schizotypal personality disorder share common phenomenological, genetic, biological, outcome, and treatment response characteristics with more severely ill patients with chronic schizophrenia. However, at the same time, they are freer from the multiple artifacts that potentially confound research in schizophrenia, including the effects of long-term and usually ongoing medication treatment, multiple hospitalizations or institutionalization, and prolonged functional impairment secondary to chronic psychosis and social deterioration. Patients with schizotypal personality disorder share with patients with schizophrenia their persistent asociality and cognitive impairment, albeit to a milder degree, which presumably emerge from common spectrum-related risk factors that are both genetic and environmental
(1). However, while chronic schizophrenia as the “end-stage” disease of the schizophrenia continuum, or spectrum, is characterized by severe, generalized deterioration across a variety of domains, including cognitive and social function, the deficits of patients with schizotypal personality disorder are more circumscribed and selective. An identification of those factors, such as altered brain regional structure and function, associated with the severe deficits and cognitive deterioration of chronic schizophrenia in contrast to those associated with the the milder impairment of schizotypal personality disorder might enhance the possibilities of preventing or ameliorating the social and cognitive dysfunction of schizophrenia. Identification of the factors that mitigate the emergence of psychosis and serious cognitive deterioration in patients with schizotypal personality disorder (or alternatively, extra liability factors in patients with schizophrenia that confer a greater susceptibility to psychosis and cognitive impairment) is critical in formulating interventions to reduce the morbidity of psychotic exacerbations in patients with schizophrenia. Thus, we posit that there are several partially separable but interactive pathophysiological processes in schizophrenia and that the study of subjects with schizotypal personality disorder offers a unique opportunity to tease apart these processes.
In this article, we selectively review the phenomenological, genetic, psychophysiological, cognitive, imaging, and neurochemical studies that might inform the hypotheses of the pathophysiology of the schizophrenia spectrum, followed by outlines of a proposed physiological model of the schizophrenia spectrum that incorporates these findings. Such a model generates research strategies, questions that can help realize a more specific understanding of the neurodevelopmental, cellular, and ultimately, molecular basis of the schizophrenia disorders as well as having implications for genetic, prodromal, and treatment studies.
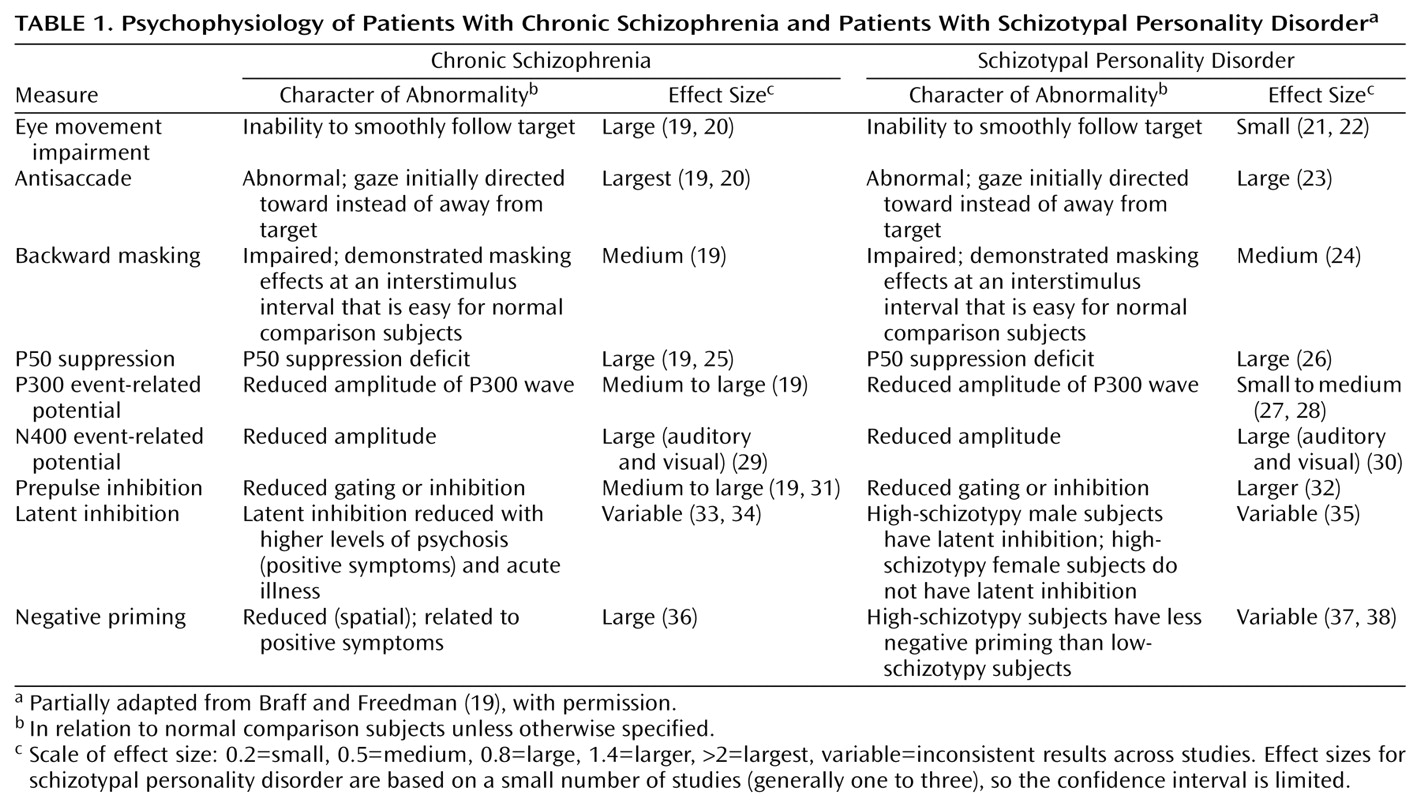
P50 suppression abnormalities are heritable, have also been identified in patients with schizotypal personality disorder as well as in clinically unaffected relatives of schizophrenia subjects, and are modulated by nicotinic receptors in the hippocampus
(25,
26). Indeed, use of the reduced P50 evoked potential as an endophenotype in a linkage study of families of schizophrenia subjects resulted in the demonstration of linkage of this phenotype to a variant of the alpha-7 nicotine receptor gene
(39). Prepulse inhibition in a blink startle paradigm also has been identified in patients with schizophrenia, schizotypal individuals, and relatives of patients with schizophrenia and is modulated by cortical-striatal-pallidal-thalamic circuitry
(31,
32). Reduced N400-evoked potentials, critically modulated by ventral temporal regions, are reduced throughout the spectrum and may reflect a failure of recurrent inhibition
(29,
30). Evoked potential abnormalities, particularly reduced P300 amplitude, have been reported in schizotypal subjects and have been associated with smaller volumes of the left posterior superior temporal gyrus in patients with schizophrenia
(27,
28).
Patients with schizophrenia not only demonstrate qualitative and quantitative impairment in smooth-pursuit tracking but also perform less accurately in antisaccade and motion detection tasks
(20,
23). Smooth-pursuit eye movements are mediated by the frontal and temporal cortex as well as the brainstem, while motion detection is mediated by the inferior temporal cortex
(40). Subjects with schizotypal personality disorder show impairment in smooth-pursuit tracking measured qualitatively and by indices such as tracking gain and saccadic intrusions, as do patients with schizophrenia. These abnormalities are particularly correlated with the social deficits and interpersonal isolation of these patients
(22). Schizotypal subjects, like patients with schizophrenia, whether identified in samples of patients, volunteers, or relatives of patients with schizophrenia, also show deficits in d′, a measure of accuracy in performance on the Continuous Performance Test, an attentional task that depends on frontostriatal circuitry
(41–
43). Of interest, schizotypal individuals perform relatively normally under conditions with low processing demand, such as nondegraded stimulus presentation, but perform more poorly than normal comparison subjects and other personality disorder comparison groups when stimuli were degraded or a more challenging identical-pair Continuous Performance Test paradigm or a dual-task Continuous Performance Test was used
(42,
44). Other psychophysiological or information-processing abnormalities shared between subjects with schizotypal personality disorder and schizophrenia include latent inhibition
(33–
35), negative priming
(36–
38), and interference
(45).
Cognitive Function
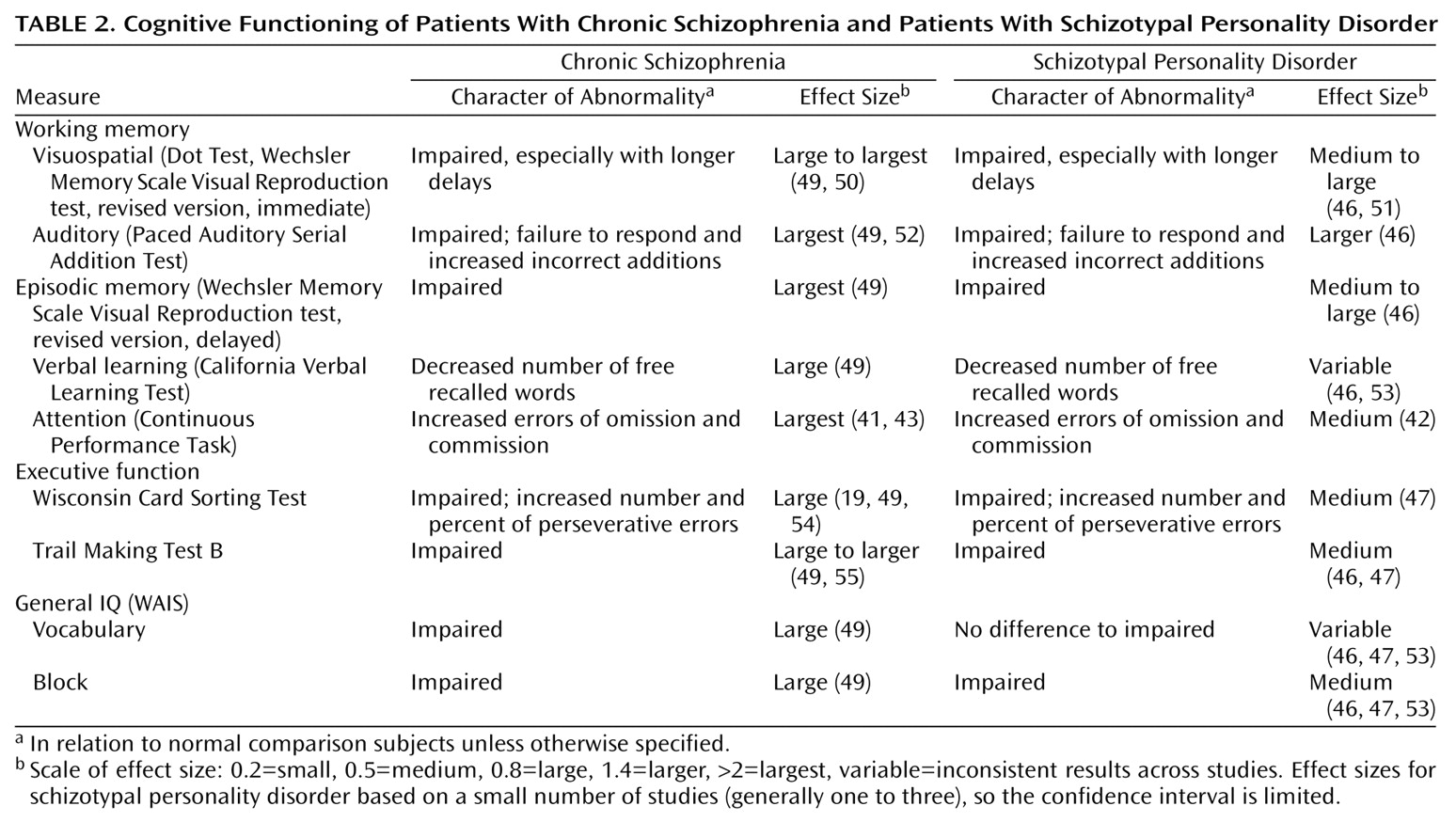
Schizotypal patients display selective deficits in cognitive processing similar to those observed in patients with schizophrenia
(46–
48) (
Table 2), reflecting domains of cognitive performance impaired in schizophrenia for which data is available for subjects with schizotypal personality disorder as well. While overall IQ seems to be preserved in patients with schizotypal personality disorder, schizotypal individuals show deficits in working memory and verbal learning as well as attentional deficits. Initial studies using a broad survey of cognitive tasks suggest impaired performance on the Wisconsin Card Sorting Test
(19,
47,
48) in schizotypal patients as well as in schizotypal relatives of patients with schizophrenia. Schizotypal individuals also demonstrated less accurate responses on the Stroop Color Word Interference Test and on the Trail Making Test B
(46,
47) as well as on other tests of executive function and abstraction
(48). These data suggest deficits in executive function that are often compatible with prefrontal cortical impairment.
It has been suggested that impairment of these executive function tasks may be, in large part, a function of impaired working memory
(46,
49,
52,
56). While the Wisconsin Card Sorting Test involves a component of spatial working memory, more direct tests of visuospatial working memory involving computerized or paper-and-pencil paradigms have demonstrated working memory impairment without visual processing defects in schizotypal subjects
(51,
53) and in relatives of patients with schizophrenia
(57). Verbal learning and episodic memory deficits, reflected in initial word list learning and multitrial memory learning, also have been reported in schizotypal individuals
(46,
48,
49,
53,
58). Schizotypal individuals, on the other hand, appear to show no deficits in perceptual processing tasks, such as in the Benton Line Orientation Test, in motor capabilities, such as in the Purdue Peg Board Test, or general intelligence
(47; Siever, unpublished data).
Thus, schizotypal individuals manifest deficits in executive function, working memory, verbal learning or episodic memory, and attention but not generalized intellectual, perceptual, and motor deficits. These impairments may point to the involvement of specific brain systems. For example, the prefrontal cortex, particularly the dorsolateral prefrontal cortex, has been implicated in both animal and human studies of executive function, such as the Wisconsin Card Sorting Test and visuospatial delayed response or working memory
(51,
59), while temporal cortical regions have been implicated in verbal learning. Frontostriatal circuits play a central role in modulating sustained attention. These studies also suggest that circuitry involving critical cortical areas, such as the frontal and temporal cortex, may be compromised in subjects with schizotypal personality disorder.
Structural Imaging
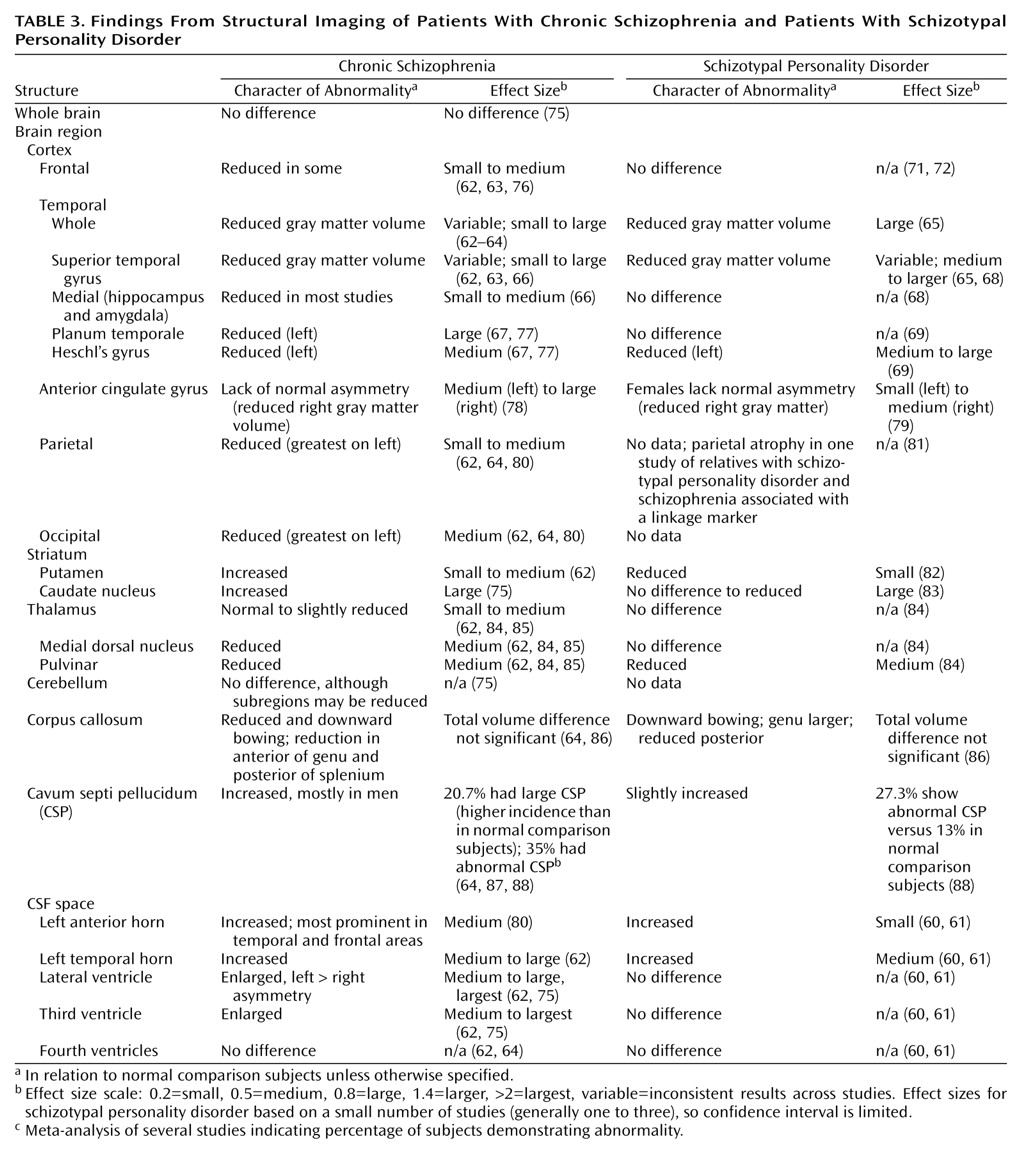
The temporal cortex, the frontal cortex, the striatum, and the thalamus have been particularly implicated as regions of interest in the schizophrenia disorders (
Table 3). CSF volumes are generally increased and cortical volumes reduced in schizotypal personality disorder
(60,
61). Volume reductions in the temporal cortex, particularly the superior temporal gyrus, have been among the most consistent structural alterations reported in chronic schizophrenia
(62–
67). The reduced size of the temporal cortex also now has been reported in schizotypal personality disorder, both in the superior temporal gyrus
(68) and Heschl’s gyrus
(69), as well as in the inferior and middle temporal gyri
(65). These reductions have been associated with schizophrenia-related psychopathology
(65). Studies of patients with schizophrenia and relatives of patients with schizophrenia also suggest reductions in medial temporal regions, including the amygdala and/or hippocampal complex, but they have not been observed in schizotypal subjects
(66,
68,
70). Thus, these findings are consistent with a model of common temporal abnormalities across the schizophrenia spectrum. Frontal cortical volume, on the other hand, appears to be relatively preserved in initial studies of schizotypal personality disorder
(71,
72), while reductions in frontal volume have been found in many but not all studies of patients with schizophrenia. However, relative reductions in frontal volume are correlated with the deficit-like symptoms of schizotypal personality disorder in healthy volunteers, implying that patients with lower frontal volume will be more likely to display traits such as asociality
(73,
74). While a number of factors extrinsic to the illness itself, including sustained neuroleptic treatment, alcohol abuse (rare in schizotypal personality disorder), and chronic psychosis, might contribute to the differences between schizotypal personality disorder/normal comparison subjects and schizophrenia/normal comparisons of frontal cortical volumes, these factors could not easily explain the fact that temporal regions are comparably reduced in both schizophrenia and schizotypal personality disorder. The finding of normal frontal volume, however, in schizotypal personality disorder requires replication.
The thalamus is a critical nodal link that integrates diverse circuits in the brain, including incoming sensory information, with higher cortical regions involved in planning response strategies. The thalamus as part of the circuitry, including the cortex and the cerebellum, has been hypothesized to play a central role in the pathophysiology of schizophrenia, and this hypothesis is supported by postmortem
(89–
91) and imaging
(62,
84,
85) studies. The thalamus encompasses a number of distinct nuclei that have partially different patterns of connectivity to other brain regions. For example, the pulvinar, which has close connections with temporal lobe structures, is reduced in subjects with schizotypal personality disorder, as it is in patients with schizophrenia, in relation to normal comparison subjects. However, the volume of the medial dorsal nucleus, associated with the prefrontal cortex, is not reduced in schizotypal patients in relation to normal comparison subjects, in contrast to the reductions observed in patients with schizophrenia
(84). Thus, reductions in the subcortical nuclei relaying from the thalamus to cortex seem to parallel reductions in associated cortical regions in schizotypal personality disorder—i.e., temporal but not frontal volume reductions.
The striatum and its connections to the cortex also have been implicated in schizophrenia, in part, because dopamine is a key neurotransmitter in these regions and striatal structures are a major target of the neuroleptic medication’s dopamine D
2 antagonism. Compensatory increases in dopaminergic dendritic structures after long-term neuroleptic administration are believed to account largely for striatal volume increases that have been rather consistently reported in patients with schizophrenia
(62,
75,
82,
92). These considerations suggest that under circumstances of altered dopaminergic activity, changes in striatal volume may partially reflect dopaminergic activity. In never-medicated patients with schizophrenia, striatal volumes may be normal or even slightly reduced. Striatal volumes have been found to be reduced in schizotypal patients in relation to normal comparison subjects and patients with schizophrenia. While these differences could, in principal, be due to differences in degree of past neuroleptic use, even never-medicated schizotypal patients have demonstrated significantly reduced striatal volume in relation to both never-medicated patients with schizophrenia and normal comparison subjects
(82) (
Table 3). In another study, reduced caudate volume was found in subjects with schizotypal personality disorder in relation to comparison subjects
(83). While these studies cannot directly address dopaminergic function, neuroleptic-induced striatal volume increases appear likely to be due to proliferation of dopaminergic dendrites or mitochondria
(92). Thus, the reduced striatal volume in schizotypal patients might be compatible with reduced dopaminergic activity in schizotypal patients compared to patients with schizophrenia.
Functional Imaging
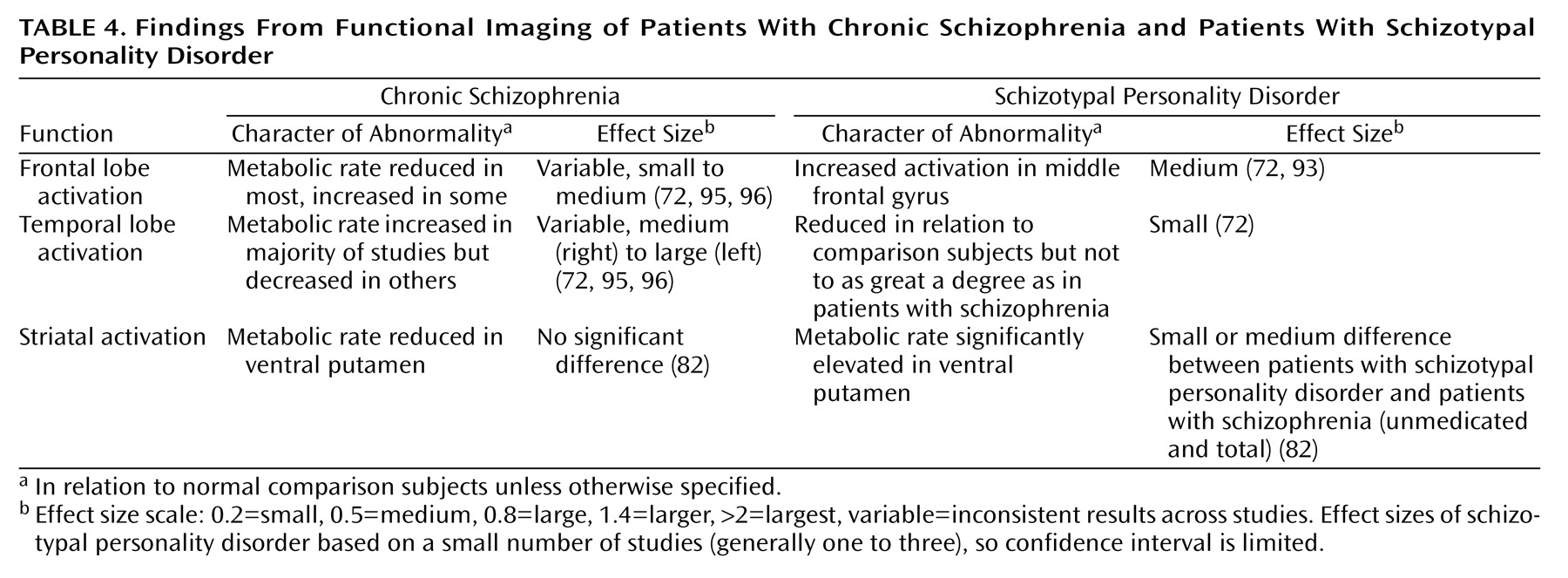
Numerous imaging studies in schizophrenia suggest reduced and/or anomalous activation of the cortex, primarily the frontal cortex. In a single photon emission computed tomography (SPECT) study measuring blood flow conducted in our laboratory, schizotypal patients performing the Wisconsin Card Sorting Test showed lower activation in the left middle frontal gyrus but greater activation in other regions of the brain, particularly the right prefrontal cortex, than in comparison subjects. These results suggested that patients with schizotypal personality disorder were less effective in activating prefrontal regions to accomplish the task efficiently than normal subjects. However, the schizotypal subjects did activate other prefrontal brain regions, such as right prefrontal cortex, which was not activated in healthy volunteers, possibly as a compensatory mechanism to offset reduced efficiency in the left prefrontal cortex
(93).
In a [
18F]fluorodeoxyglucose (FDG) positron emission tomography (PET) paradigm measuring brain glucose metabolism, schizotypal patients demonstrated a pattern of altered temporal lobe use in laterality similar in character but not as severe as that observed in patients with schizophrenia
(72). New blood-oxygen-level-dependent (BOLD) functional magnetic resonance imaging (fMRI) data from our laboratory
(94) also suggest that patients with schizotypal personality disorder do not activate the dorsolateral prefrontal cortex to the degree that normal comparison subjects do, while they do activate Brodmann’s area 10 to a greater degree than comparison subjects. These results suggest compensatory activation of prefrontal regions in schizotypal patients other than those employed by normal comparison subjects. They are consistent with the possibility that patients with schizotypal personality disorder are better able than patients with schizophrenia to use frontal reserves to compensate for inefficiency of task performance.
The FDG PET study performed in our center also focused on striatal structures. Significantly increased metabolic activity was found in the ventral putamen, an area rich in D
2 receptors, in subjects with schizotypal personality disorder in relation to patients with schizophrenia and normal comparison subjects
(82). As these D
2 receptors mediate dopaminergic inhibition of putamen activity, these results raised the possibility of reduced dopaminergic modulation of the putamen. This increased activation also was seen in never-medicated schizotypal patients compared to never-medicated patients with schizophrenia, suggesting that the difference between subjects with schizotypal personality disorder and schizophrenia was not an artifact of different medication histories. Greater activation was associated with less psychotic-like schizotypal personality disorder symptoms, while reduced activation (possibly reflecting greater dopaminergic inhibition) was associated with greater psychotic-like symptoms
(82).
In summary, functional imaging studies suggest that activation patterns in the cortex in subjects with schizotypal personality disorder display similar anomalies to those observed in patients with schizophrenia but to a lesser degree (
Table 4). Furthermore, the presence of compensatory activity in the frontal lobe may diminish the functional impact of reduced activation in temporal or other cortical regions. Metabolic activity appears increased in the ventral striatum, possibly associated with reduced dopamine inhibition.
Neurochemistry
Due to the profound influence of the dopamine hypothesis of schizophrenia, based largely on the efficacy of neuroleptic medication, neurochemical studies of schizotypal individuals initially focused on dopamine and its metabolites in CSF and plasma. Decreased levels of dopamine metabolites have been reported in patients with schizophrenia with poor prognosis and moderately severe social impairment. Increased levels of dopamine metabolites have been noted in patients with greater psychotic symptoms
(97,
98). In the first report from our laboratory, CSF homovanillic acid (HVA) was found to be increased in schizotypal patients in relation to normal comparison subjects. However, this increase was entirely accounted for by the psychotic-like symptoms of the disorder, and covarying for these symptoms abolished the difference. Furthermore, significant correlations were found between CSF HVA activity and these psychotic-like symptoms of schizotypal personality disorder
(99).
Similar results were obtained in studies in our center using plasma HVA as an index of dopaminergic activity. Plasma HVA concentrations were increased in schizotypal patients in relation to normal comparison subjects or patients with other personality disorders. These increases were correlated with the number of psychotic-like symptoms but not other deficit-related schizotypal symptoms. Covarying for these symptoms similarly nullified the differences between groups
(100).
Plasma HVA concentrations also were determined in relatives of patients with schizophrenia
(101). These relatives were diagnosed either with schizotypal personality disorder or other non-schizophrenia-related personality disorder or had no diagnosis. The schizotypal relatives of the patients with schizophrenia had reduced plasma HVA concentrations compared to the relatives with other personality disorder diagnoses or no diagnosis. In this case, the deficit-like symptoms, which are more prominent in the relatives of patients with schizophrenia than the psychotic-like symptoms
(5), accounted for the findings of reduced HVA concentrations. Covarying for negative deficit-like symptoms rendered any differences insignificant; significant correlations were found between the extent of deficit-like symptoms and reduced HVA concentrations. After residualizing the variance of negative or deficit-like symptoms, the positive correlation between HVA concentrations and psychotic-like symptoms emerged just as in the clinical patients. While plasma concentrations derive from both peripheral and central sources, under usual conditions, the peripheral component is correlated with the central component
(102), suggesting that these differences in correlations may reflect, in part, central differences. Furthermore, in this personality disorder cohort, CSF and plasma HVA were positively correlated
(73) (
Table 5).
The convergence of the CSF and plasma HVA studies suggest that higher dopaminergic activity may be associated with greater psychotic-like symptoms and reduced dopaminergic activity may be associated with greater deficit-like symptoms in patients with schizotypal personality disorder. Group differences may emerge when one or the other symptom domains predominates, but the correlations between symptom clusters and HVA concentrations are the most consistent finding in these studies of HVA. Indeed, these results are consistent with studies of patients with schizophrenia, suggesting that reductions of dopaminergic activity in the prefrontal cortex are associated with the deficit symptoms and cognitive impairment of schizophrenia and that increased activity in the subcortex is associated with the psychotic symptoms of schizophrenia
(97). Studies of schizotypal personality disorder may permit a clearer dissociation of these two relationship roles in schizotypal personality disorder than is easily possible in chronic schizophrenia, where deficit and psychotic symptoms may be more highly correlated as a function of severity of illness.
Neurochemical Imaging
More direct assessment of neurotransmitter functions, such as dopamine, in specific brain regions is possible with neurochemical imaging paradigms utilizing SPECT or PET. For example, dopamine release from the striatum can be evaluated following the administration of a single dose of amphetamine and measuring displacement of radiolabeled [
123I]IBZM, which labels D
2 receptors in the striatum. The degree of IBZM displacement or percent reduction in binding can then be used as a measure of dopamine release stimulated by amphetamine. A similar paradigm using the D
2 ligand raclopride’s displacement by dopamine release in PET scans has also been applied to patients with schizophrenia
(104). Amphetamine induces greater dopamine release in patients with schizophrenia than in normal comparison subjects in several independent studies using either a SPECT-IBZM protocol
(105) or a PET-raclopride protocol
(104). The degree of dopamine release following amphetamine administration is associated with worsening of psychotic symptoms in response to the amphetamine in these studies. These results suggest that in patients with acute schizophrenia, stimulated dopamine release is greater than in normal comparison subjects, and this may be associated with their acute psychosis. More recent results measuring basal dopaminergic activity by using alpha-methylparatyrosine to deplete available dopamine and measuring increases in raclopride after administration of alpha-methylparatyrosine suggest that basal dopaminergic activity is similarly increased in patients with schizophrenia and is indeed correlated with the extent of amphetamine-stimulated release.
In contrast, in a study of patients with schizotypal personality disorder using the IBZM SPECT paradigm, IBZM displacement after amphetamine administration in schizotypal personality disorder was significantly less than that observed in patients with schizophrenia, although it was modestly but significantly higher than that of normal comparison subjects
(106). No worsening of psychotic-like symptoms was seen in any of the schizotypal patients studied after the amphetamine administration, and improvement in negative symptoms was observed in the total cohort as well as improvement in positive symptoms in some patients (
Table 5). While the plasma and CSF measures of dopamine metabolism in the initial studies were assessed in groups of patients with schizotypal personality disorder with more prominent psychotic-like symptoms and were correlated with psychotic-like symptoms, the patients in this study had a range of symptoms, but deficit symptoms were more prominent. These studies cumulatively suggest that there may be variable dopaminergic activity in schizotypal personality disorder that may be directly associated with the extent of psychotic-like symptoms and inversely with deficit-like symptoms.
Stressors, such as the administration of 2-deoxyglucose, a glycopyruvic stressor, result in increases in dopamine release in the subcortex reflected in increases in plasma HVA and as well as increases in ACTH and cortisol. Patients with schizophrenia show increased plasma HVA and ACTH responses to 2-deoxyglucose compared to normal comparison subjects
(107,
108). In contrast, patients with schizotypal personality disorder show unchanged plasma HVA concentrations in response to 2-deoxyglucose relative to that of normal comparison subjects and reductions in plasma cortisol in relation to normal comparison subjects in a 2-deoxyglucose protocol similar to that used in the study of patients with schizophrenia
(109). These results suggest that while patients with schizophrenia may have a greater susceptibility to subcortical dopaminergic release under stress, schizotypal individuals do not show this increased response, suggesting better buffering of the subcortical dopamine system in this disorder. Thus, dopaminergic indices do not appear to be consistently increased in prototypical patients with schizotypal personality disorder who have not exhibited frank psychotic symptom profiles, in contrast to patients with schizophrenia, in which direct measures of release correlate with psychotic exacerbation.
A Pathophysiological Model of the Spectrum Disorders
The results of the various studies reviewed, coming from diverse investigative perspectives, suggest not only that there may be partially distinct susceptibilities to the schizophrenia spectrum and psychosis but also hint at the beginnings of an understanding of the underlying pathophysiological processes underlying each. Thus, we propose a model based on the data that can generate testable hypotheses for future research. In this model, schizotypal and schizophrenic individuals are hypothesized to share a common genetic anomaly that renders the temporal cortex particularly vulnerable to environmental insults such as hypoxia. However, genetic factors independent of the vulnerability to the schizophrenia spectrum per se and/or more favorable environmental influences would leave the schizotypal individual better buffered with regard to frontal volume and function as well as stabilization of subcortical dopaminergic activity. Thus, phenotype determining genetic and environmental factors that are partially distinct from those factors directly associated with the susceptibility to schizophrenia, e.g., frontal reserve capacity or, at a cognitive level, general intelligence, serve as mitigating factors in schizotypal personality disorder, diminishing the impact of the genetic susceptibility genes to schizophrenia. These modifying factors may play an important role in determining whether a susceptible individual develops chronic schizophrenia or a milder spectrum condition, with potentially important implications for early intervention and treatment.
This model of the pathophysiology of the schizophrenia spectrum disorders posits that the social deficits and cognitive impairment characteristic of the entire spectrum of schizophrenia-related disorders is the result of an underlying genetic diathesis that, in conjunction with modifying environmental factors, adversely affects cortical structures such as the temporal and prefrontal cortex. These structures may be particularly sensitive to developmental aberrations such as altered migration of neurons, aberrant dendritic growth patterns, or alterations in the compaction and maintenance of the myelin sheaths of axons, with subsequent disconnection between critical brain regions
(110). Temporal and related lateral cortical regions that may be impaired across the schizophrenia spectrum disorders represent logical sites to investigate, for reasons to be elucidated.
As cited in this overview, temporal volume reductions and functional abnormalities are among the most consistently observed findings in schizophrenia
(64,
66). These abnormalities are hypothesized to constitute a primary spectrum abnormality (i.e., present across the entire spectrum of schizophrenia-related disorders) and are associated with the core symptom profile and cognitive impairment of the schizophrenia disorders. This pathology might emerge from genetic susceptibilities interacting with adverse environmental events, such as hypoxia from birth complications, that represent a risk factor for the schizophrenia-related disorders
(111). An animal model of schizophrenia suggests that neonatal lesions in the ventral hippocampus, which represents a medial temporal structure, can cause a number of abnormalities in the adult rat that resemble the clinical signs and symptoms of schizophrenia
(112). Animals with these lesions demonstrate locomotor hyperactivity after pharmacological stimulation with agents such as amphetamine or
N-methyl-
d-aspartic acid antagonists and after stress paradigms associated with altered subcortical dopaminergic responsiveness as well as social deficits. These effects appear to be mediated by prefrontal cortical neurons, and the vulnerability to these behavioral consequences depends in part on genetic factors
(113). Along these lines, in twins discordant for schizophrenia, hippocampal volume reductions are significantly correlated with cognitive dysfunction in the Wisconsin Card Sorting Test that is mediated substantially by the frontal lobe
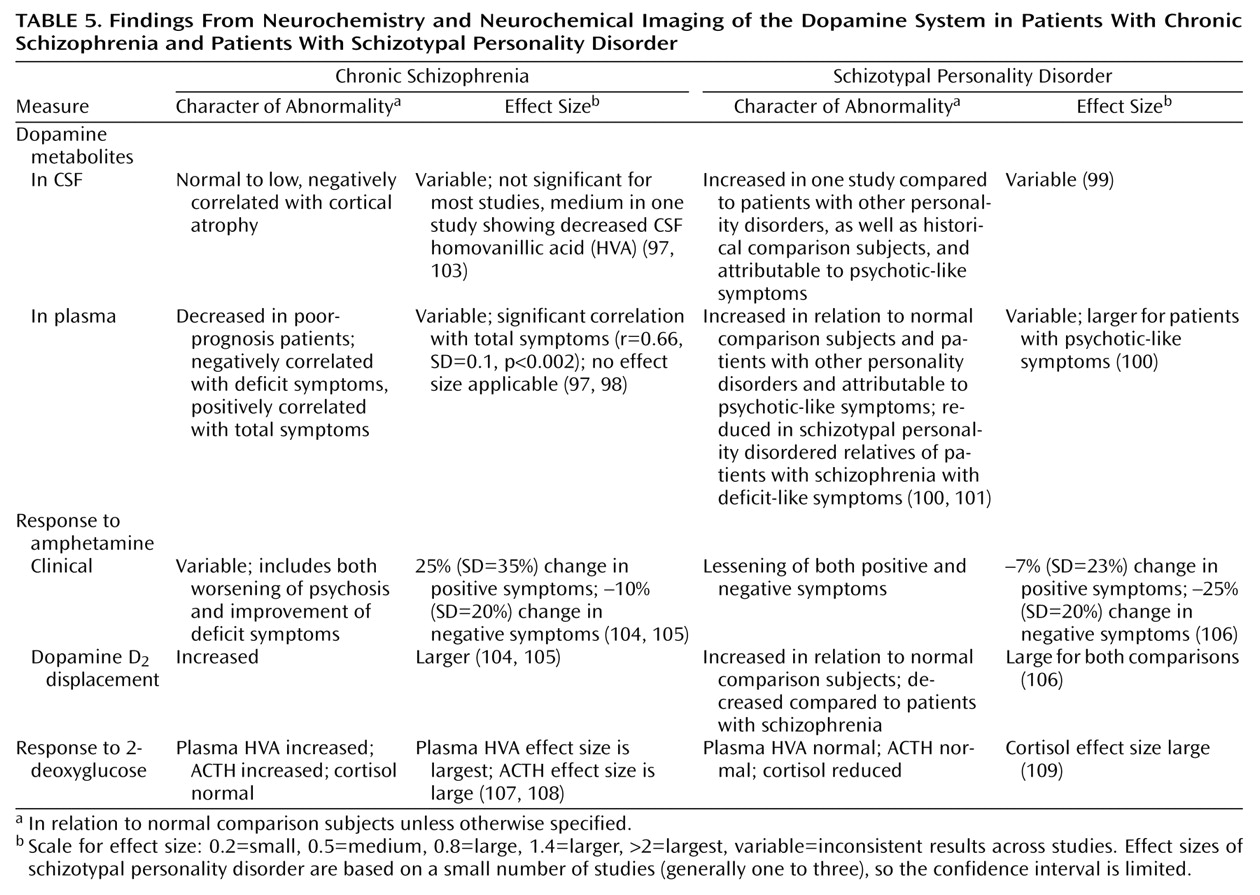
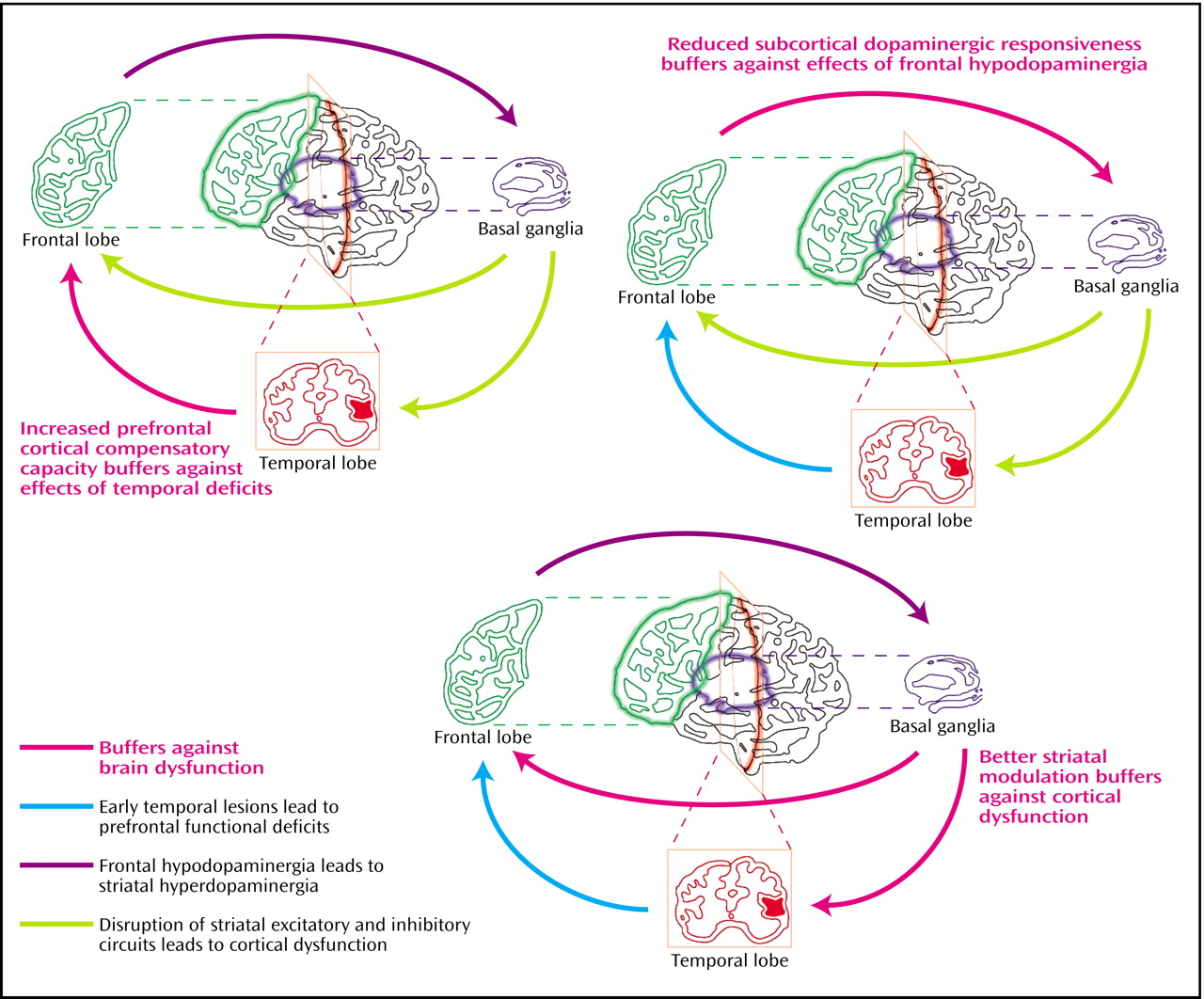
(114). This model would therefore predict that the temporal lobe and the hippocampus would be among the regions most consistently displaying reduced volume across the schizophrenia spectrum disorders, including schizotypal personality disorder, with consequences for frontal function. Patients with schizotypal personality disorder could be protected from the downstream effects of abnormalities in these lateral structures by compensatory strengths in intrinsic frontal functional capacity and/or greater intrinsic stability of subcortical structures (
Figure 1 and
Figure 2).
While patients with schizophrenia show volume reductions in the frontal lobe in many but not all studies, subjects with schizotypal personality disorder exhibit normal to increased frontal lobe volumes in the limited data available
(72). In this model, we propose that greater frontal reserves in schizotypal personality disorder compared to schizophrenia protect the schizotypal individual from the severe cognitive deterioration and social deficits associated with chronic schizophrenia. Schizotypal individuals may also be able to better use their frontal capacities, perhaps by compensating for dysfunction in regions such as the dorsolateral prefrontal cortex, used by healthy volunteers to accomplish working memory and executive tasks, by activating other frontal regions not normally recruited by normal volunteers
(72,
94). Thus, schizotypal individuals would be more resistant to the downstream effects of temporal pathology. This model predicts that frontal volumes would be more preserved in schizotypal subjects than in schizophrenia subjects and that schizotypal individuals would be able to activate compensatory prefrontal regions. In contrast, normal volunteers do not need to activate these regions, whereas patients with schizophrenia, unlike schizotypal individuals, are unable to compensate substantially for dorsolateral prefrontal cortex dysfunction by activating supplemental brain areas.
The thalamus, a nodal way station linking diverse regions of the brain, has been hypothesized to play a central role in the altered circuitry in schizophrenia
(115). The finding of reduced pulvinar volume in schizotypal personality disorder, as also observed in patients with schizophrenia, and unchanged medial dorsal nucleus volume, in contrast to the reduced volume of this region in patients with schizophrenia
(84), suggests that abnormalities in these thalamic nuclei parallel those of their cortical projections and implicate the temporal cortex and pulvinar as part of circuitry that is impaired across the schizophrenia spectrum disorders, while prefrontal circuitry is impaired primarily in schizophrenia.
Finally, subcortical structures that are modulated by the frontal cortex may be better protected from the impact of relative dysfunction of the frontal cortex and functional deafferentation of subcortical structures in schizotypal subjects compared to schizophrenia subjects, perhaps because frontal function is better preserved and/or because intrinsic dopamine activity in the striatum may be more effectively regulated. Lesions of prefrontal cortical dopamine systems or disconnection of the frontal cortex and the striatum result in up-regulation of subcortical dopaminergic activity, with increases in presynaptic dopamine release and dopamine receptor sensitivity
(116,
117), providing a possible animal analogue of frontal cortical hypodopaminergia leading to subcortical dopaminergic overactivation in schizophrenia (
Figure 1). Different rodent strains vary in the degree of up-regulation observed in response to these kinds of perturbations
(113,
117). Comparable individual differences between people might also modulate the degree to which striatal dopaminergic up-regulation occurs in the face of frontal dysfunction, for example, as may be induced by stress paradigms
(107) or secondary to functional reductions in frontal dopaminergic activity. Schizotypal individuals may be constrained in their capacity for up-regulation of subcortical dopaminergic activity
(1), secondary to differences in intrinsic subcortical dopaminergic activity, receptor regulation, or other key modulator systems, such as the glutamate system. Thus, they may be less likely to up-regulate subcortical dopaminergic systems than patients with schizophrenia in response to frontal hypodopaminergia, protecting them from the emergence of overt psychosis (
Figure 2).
This hypothesis is consistent with findings of reduced release of dopamine in subjects with schizotypal personality disorder compared to patients with schizophrenia after the administration of pharmacological releasing agents, such as amphetamine
(106) or stressors that perturb dopaminergic activity
(109). Furthermore, it would suggest that psychotic-like symptoms would not worsen in patients with schizotypal personality disorder after amphetamine administration and might be accompanied by improvement of deficit symptoms in subjects with schizotypal personality disorder, as has been observed in studies of schizotypal personality disorder without comorbid borderline personality disorder using oral amphetamine
(118,
119).
This model also allows an evaluation of intermediate phenotypes or endophenotypes that are more likely to have underlying genetic foundations and are associated with specific brain dysfunction in schizophrenia
(19,
39). If intermediate phenotypes could be identified at a neurobiological level as, for example, in a specific psychophysiological, biochemical, or cognitive abnormality, we may be more successful in identifying the underlying genes for the schizophrenia syndrome. Phenotypes characterized by asociality, cognitive disorganization, and restricted affectivity, traits that are common to the entire spectrum of schizophrenia disorders, might reflect a genetic impairment, particularly prominently expressed in temporal or hippocampal regions, that is common to the entire schizophrenia spectrum.
One promising example of an endophenotype that is intermediate between the genotype and clinical phenotype is the P50 suppression abnormality, which is associated with the alpha-7 nicotinic receptor gene in relatives of patients with schizophrenia and reflects an abnormality in sensory gating or filtering function of the hippocampus, where these receptors are localized
(25,
39). Another intermediate phenotype may be characterized by severe executive dysfunction and failure of planning and abstraction related to aberrant frontal activity and observed in more chronic schizophrenia and to a lesser degree in schizotypal individuals. For example, a common allele (the Val allele) of a functional polymorphism, the Val(108/158)Met, for catechol
O-methyltransferase (COMT), the major enzyme for extraneuronal metabolism of dopamine, is associated with increased activity of COMT compared to the lower-activity form of COMT coded by the Met allele, thus resulting in relatively reduced availability of dopamine in the prefrontal cortex. The Val allele is associated with greater perseverative errors on the Wisconsin Card Sorting Test and poorer performance on the N-back Continuous Performance Test, reflecting deficits in executive function and working memory. Thus, working memory impairment may serve as an endophenotype for a gene that appears to account for a modest but significant proportion of the variance for the susceptibility to schizophrenia
(59) and may be associated with schizotypal personality disorder in preliminary studies
(120). Finally, a third intermediate phenotype may be characterized by psychosis, without necessarily outstanding prefrontal cognitive impairment or deficit symptoms, related to underlying increased subcortical dopaminergic activity. Candidate genes related to dopamine activity, such as the genes for dopamine beta-hydroxylase
(121,
122), a key biosynthetic enzyme for dopamine, or the dopamine D
4 receptor (DRD4)
(123,
124), have been associated in these studies with psychosis per se rather than schizophrenia specifically. While these examples are certainly not definitive, they illustrate the power of a multifactorial or multidimensional perspective applied to candidate genes and intermediate phenotypes in schizophrenia.
These phenotypic dimensions may be interactive so that, for example, the predispositions to neurodevelopmental aberration in the hippocampus or the temporal cortex, frontal dysfunction, and psychosis may interact synergistically to increase the probability of developing schizophrenia, while hippocampal impairment might lead to P50 abnormalities and perhaps spectrum pathology but not necessarily schizophrenia. Vulnerability and protective factors within a single domain, e.g., hippocampal function, may also be interactive so that, for example, the relatives of schizophrenia probands with P50 abnormalities have been found to have larger hippocampi than those without the P50 abnormality, consistent with the hypothesis that larger hippocampi reflecting increased processing capacity may partially compensate for the sensory gating deficit in these relatives, thus protecting them from schizophrenia
(125). Available data testing multiple endophenotypes in the same subjects with spectrum pathology suggest that endophenotypes such as P50 and prepulse inhibition are partially distinct in that they do characterize different subpopulations
(126). A better understanding of these partially distinct pathophysiological processes as they manifest themselves across the schizophrenia spectrum may help point to new promising endophenotypes and candidate genes and help clarify how they interact.