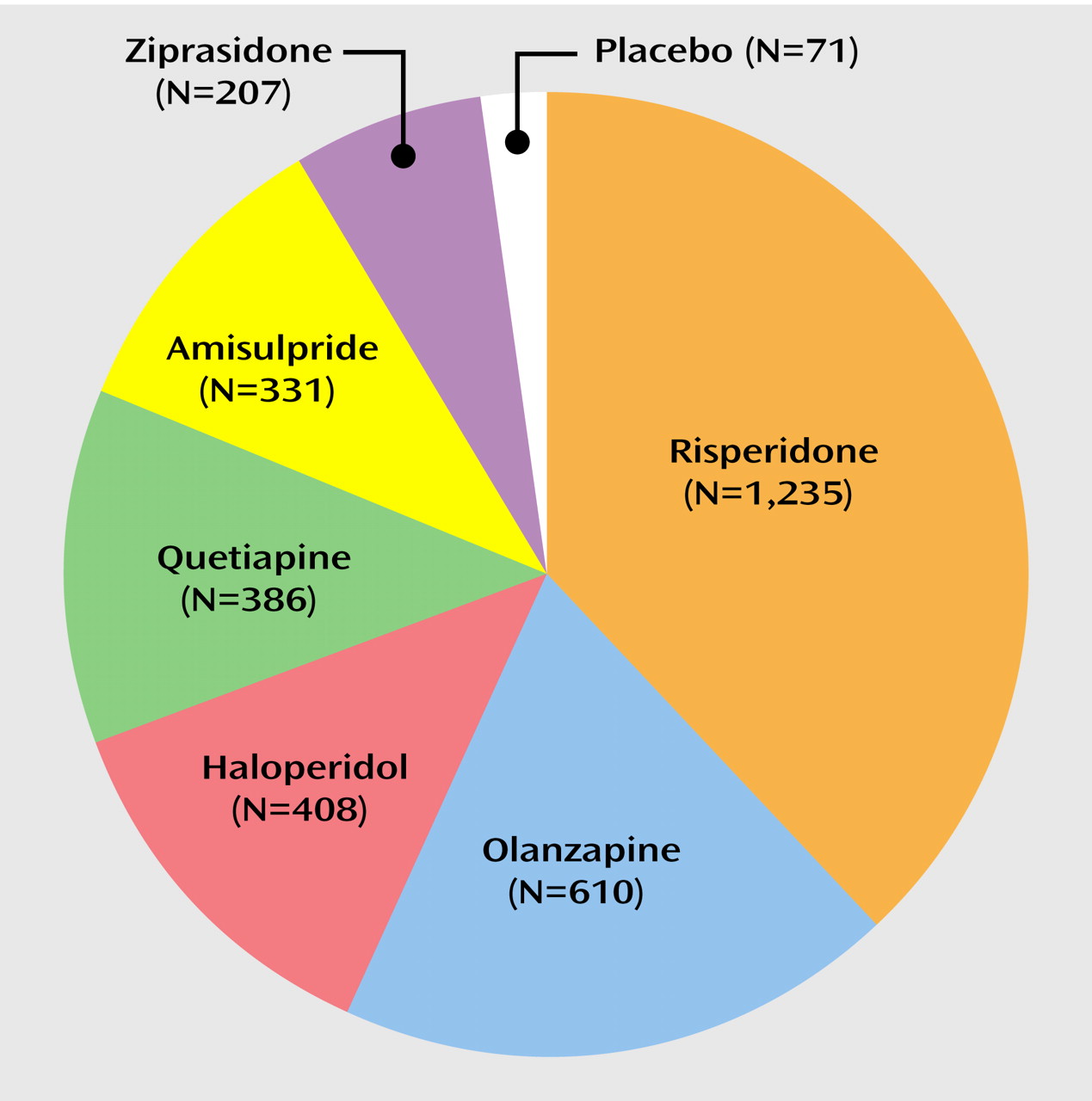
Major Findings
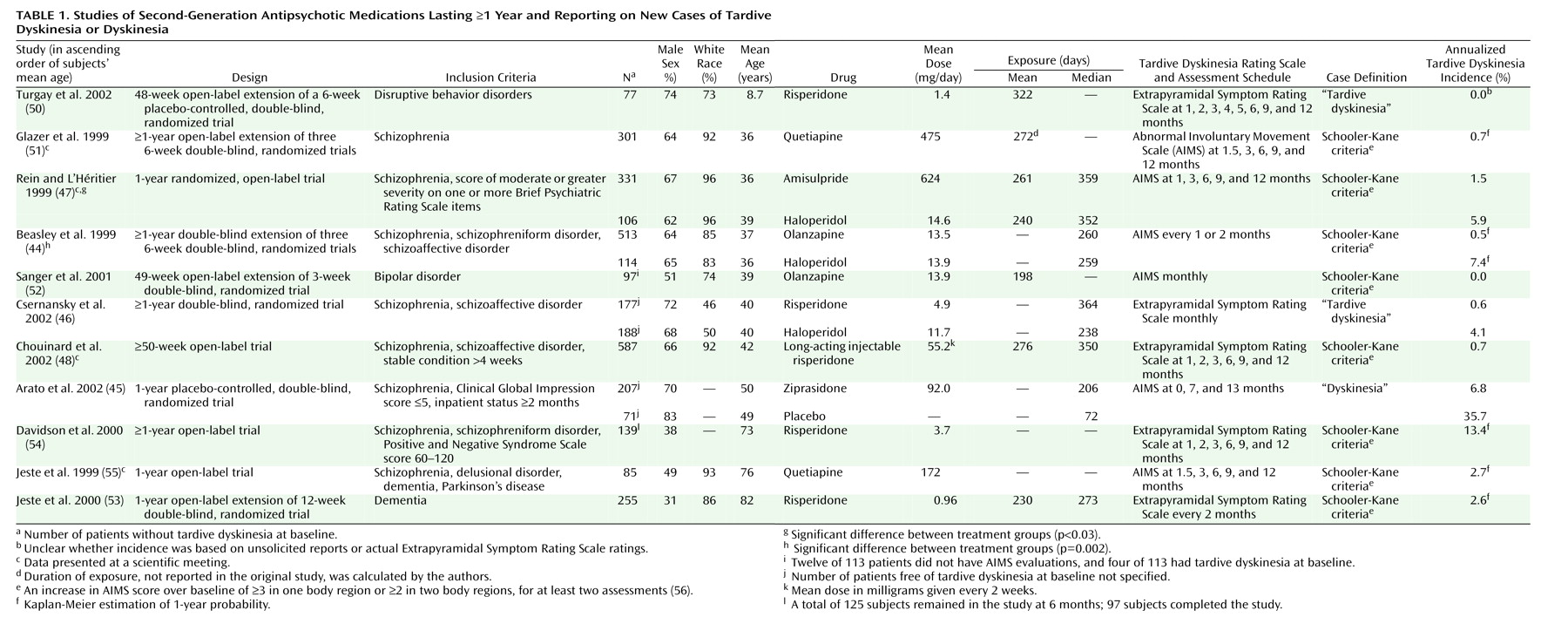
The major finding of this systematic review is that the available data from 11 long-term studies involving 2,769 patients seem to support the expectation that second-generation antipsychotic agents have a reduced risk for tardive dyskinesia, compared with conventional antipsychotics
(12,
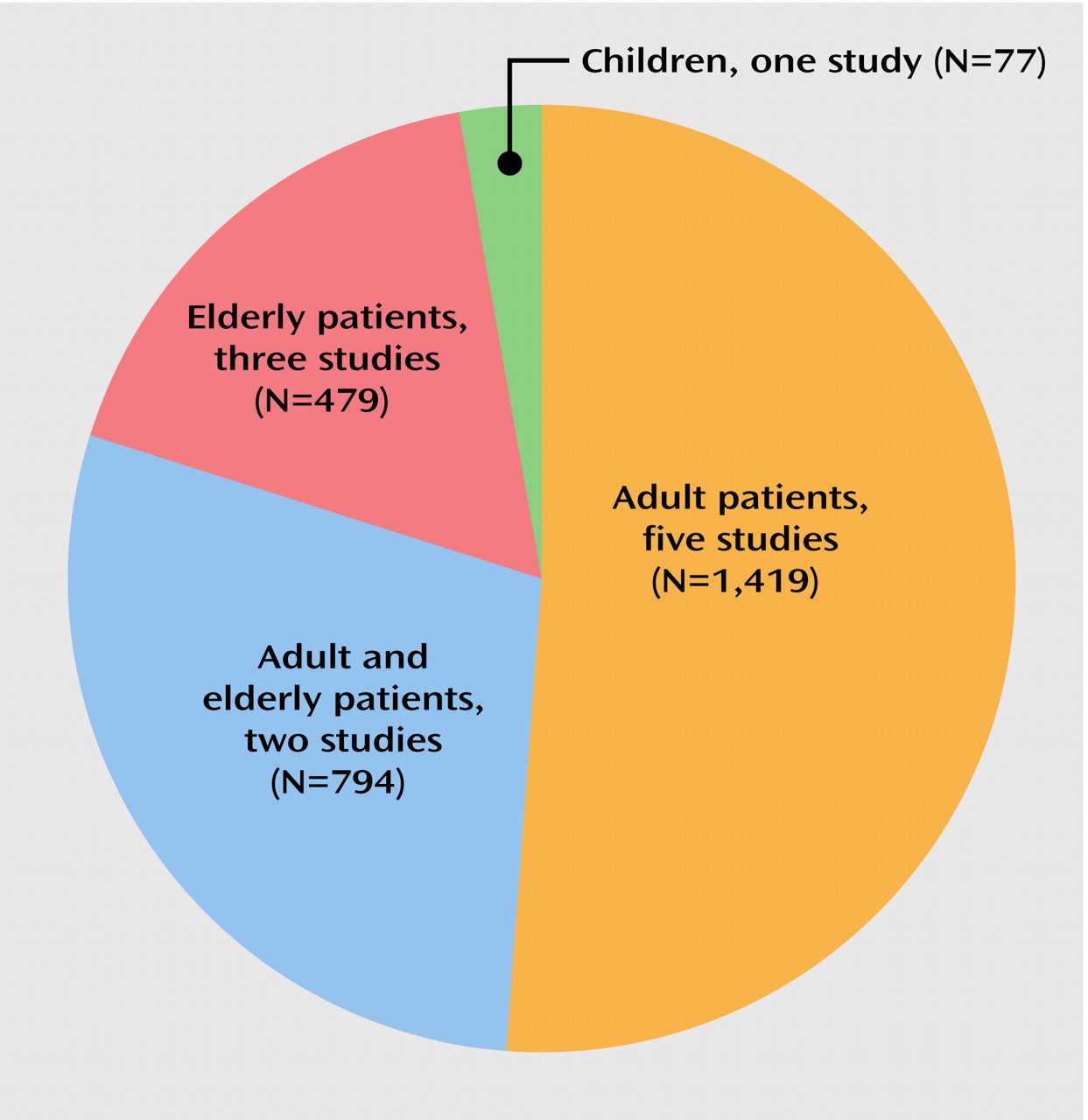
20). It is important to note that this finding was true for children, adults, and the particularly vulnerable elderly population.
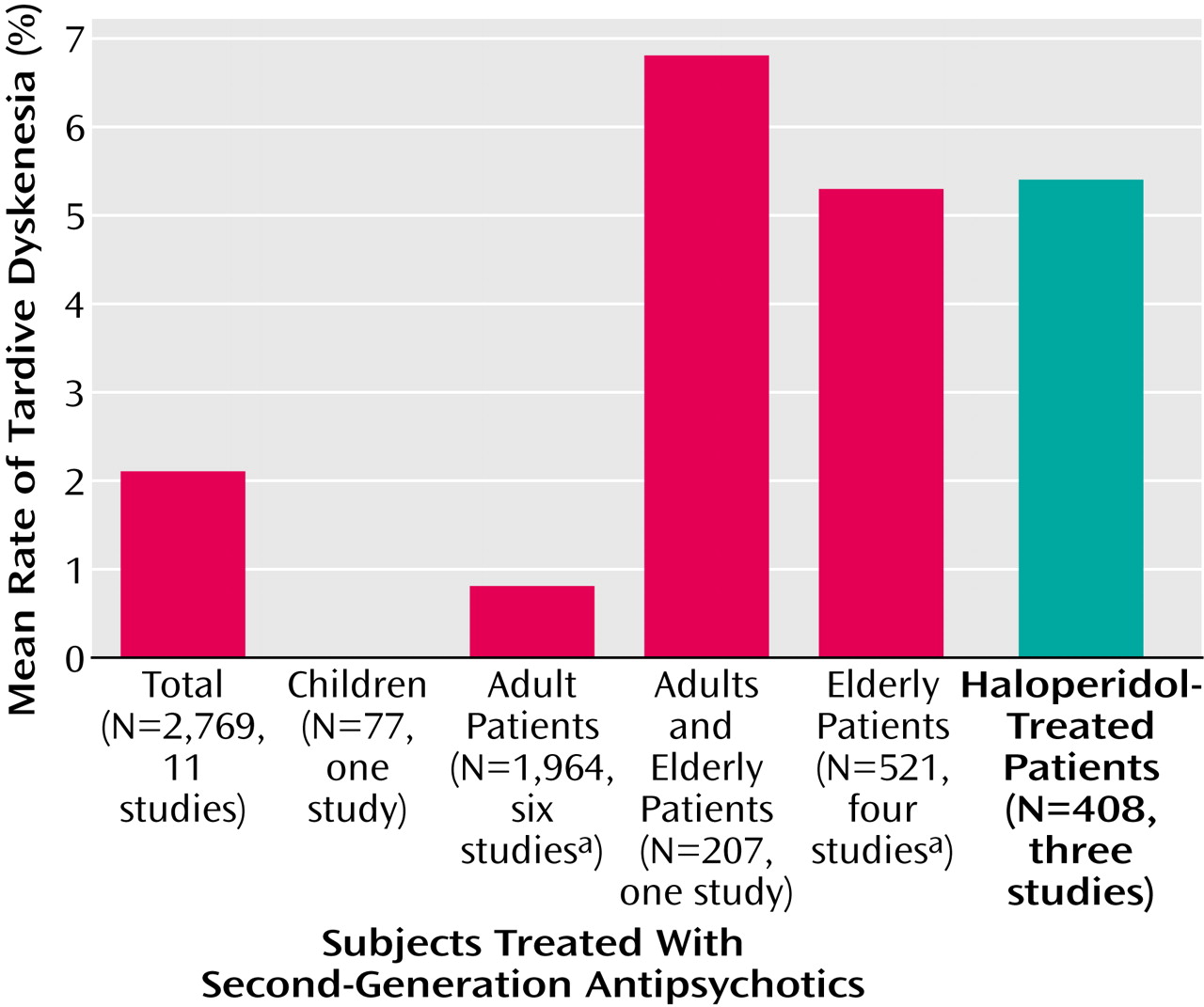
Across the six studies including 1,964 adults, the weighted mean annual tardive dyskinesia incidence for second-generation antipsychotics was only 0.8%. Except for one trial without any new cases of tardive dyskinesia during olanzapine treatment of 97 patients with bipolar disorder (in which 12 patients were excluded from the tardive dyskinesia analysis for unclear reasons), the rates from two studies with risperidone and one study each with amisulpride, olanzapine, and quetiapine were relatively similar (0.5%–1.5%). Furthermore, the mean annual tardive dyskinesia risk of less than 1% per year is considerably lower than the mean annualized tardive dyskinesia incidence of 5.4% for haloperidol in the three studies that included haloperidol as an active comparator. Moreover, the tardive dyskinesia rates for haloperidol were significantly higher, compared to those for olanzapine and amisulpride, in the studies that reported comparative statistical analyses
(44,
47), and the rates for haloperidol were consistent with 1-year incidences of about 5% for first-generation antipsychotics described in the literature
(12,
60). As the mean haloperidol doses were above 10 mg/day in all three trials, a dose effect in the haloperidol group cannot be excluded. However, the rates of tardive dyskinesia in subjects taking haloperidol were very similar to the rates in subjects taking conventional antipsychotics in the Hillside longitudinal study of tardive dyskinesia development, a naturalistic study that involved multiple neuroleptics and doses (unpublished paper of J.M. Kane et al.). On the other hand, a recently published study found a 12-month incidence of probable or persistent tardive dyskinesia of 12.3% in 57 subjects with a first episode of a nonaffective psychotic disorder treated with low-dose haloperidol at a mean dose of 1.7 mg/day
(61). Although in this study a higher haloperidol dose was a significant predictor of tardive dyskinesia, these results suggest that, at least, the high-potency first-generation antipsychotic haloperidol carries a significant risk for the development of tardive dyskinesia, even if given at low doses
(61). Similarly, an earlier study in an elderly population also demonstrated a high risk of tardive dyskinesia after 1 year of treatment with low median doses of conventional antipsychotics (68 mg/day of chlorpromazine equivalents)
(7). In addition to the results from the long-term studies included in this analysis, lower rates of tardive dyskinesia with second-generation antipsychotics have been reported in several shorter-term studies involving adults with similar mean ages (i.e., 35–40 years). Based on pooled data from a mix of 12 open-label and 15 double-blind studies with risperidone (nine lasting ≥3 months, and seven lasting ≥1 year), the incidence of tardive dyskinesia was observed to be 0.2% in 878 adults (median age=35 years, range=17–85) who completed at least 3 months of treatment
(62) and was only slightly higher (0.3%) when the analysis was restricted to the seven 1-year studies
(63). However, these data were derived from heterogeneous studies and did not take individual exposure times into consideration, and most important, both of these incidences were extrapolated only from spontaneous reports of adverse effects. Although common for many industry databases, the use of unsolicited reports of tardive dyskinesia as an adverse event is problematic since it may markedly underestimate the true risk for tardive dyskinesia. In the study by Davidson et al.
(54), for example, no “spontaneous reports” of tardive dyskinesia as an adverse event were recorded; yet, six new cases of tardive dyskinesia could be detected among 139 patients who were free of tardive dyskinesia at baseline when the patients underwent formal assessment with the Extrapyramidal Symptom Rating Scale. Similarly, neither the rating scale nor the case definition of tardive dyskinesia was specified in another 6-month, open-label, adjunctive trial with risperidone involving 541 adults (430 completers) with bipolar disorder and schizoaffective disorder
(30). In that study, no new case of tardive dyskinesia was detected during three follow-up visits (at 1, 3, and 6 months). On the other hand, in an 18-week double-blind, placebo-controlled trial (subjects’ mean age=39 years, range=18–70) that compared olanzapine and clozapine and included 90 patients with treatment-resistant schizophrenia in each treatment group, the tardive dyskinesia incidence of 2.2% in the olanzapine group was comparatively high and significantly higher (p<0.03) than in the clozapine group, which had no new cases
(64). This finding, however, may have been related to the fact that the subjects were patients with treatment-resistant schizophrenia who had, by definition, been exposed to relatively long periods and high doses of antipsychotic treatment before study entry.
In the five trials that also included elderly patients or that were restricted to elderly patients (mean age ≥50 years), the weighted annual rates for dyskinesia and tardive dyskinesia associated with second-generation antipsychotics were 6.8% and 5.3%, respectively (including the unusual finding of no case with tardive dyskinesia among 42 elderly subjects treated for an unreported duration with long-acting injectable risperidone
[48]). These results are consistent with the findings of a 9-month open-label study involving 61 adults with schizophrenia, mood disorders, dementia, and organic mental disorders who were older than age 45 years (mean age=66 years)
(65). That study found a cumulative incidence of tardive dyskinesia of 5% with risperidone (median dose=1.0 mg/day), compared to 32% in haloperidol-treated comparison subjects (median dose=1.0 mg/day), matched for age, sex, and duration of treatment with neuroleptics (p<0.05). However, the individual 1-year tardive dyskinesia rates in the reviewed studies involving elderly patients were more dissimilar than those for adults, ranging from 0% to 13.4%. Nevertheless, this disparity appears to be largely due to methodological differences. For example, the relatively high annualized rate of 6.8% with ziprasidone in the study by Arato et al.
(45) was based on cases with dyskinesia, not tardive dyskinesia. Moreover, this comparatively high rate may be at least partly attributable to early withdrawal dyskinesias, since previous antipsychotic medications were discontinued rapidly over 1–3 days. This rate is also consistent with the otherwise counterintuitive finding that in the placebo group the annualized risk for dyskinesia was as high as 35.8%. Conversely, in a 28-week randomized, double-blind study involving adults age 18–65 years (mean age=39 years) that compared ziprasidone (N=148, mean dose=116.5 mg) and haloperidol (N=153, mean dose=8.6 mg) and included a 3–14-day washout phase, not one case of tardive dyskinesia was observed in the ziprasidone group, compared to an annualized tardive dyskinesia rate of 3.4% with haloperidol
(34). Although this trial did not include subjects older than age 65 years and the AIMS ratings took place only at baseline and week 28, these data suggest that the annualized dyskinesia rate of 6.8% for ziprasidone in a mixed population may be an overestimate of the true risk for tardive dyskinesia. On the other hand, the marked difference between the annual tardive dyskinesia risks in the two studies with oral risperidone in elderly patients, i.e., 2.6%
(53) versus 13.4%
(54), seems to be due to higher doses in the latter trial (mean daily dose of 3.7 mg, compared with 0.96 mg), which included markedly ill patients with chronic schizophrenia. In addition, these individuals probably had a higher lifetime exposure to antipsychotic agents than the subjects with dementia in the study by Jeste et al.
(53). Furthermore, the tardive dyskinesia incidence could also have been increased by early withdrawal dyskinesia, as previous antipsychotic and anticholinergic medications were stopped abruptly and “gradual” discontinuation was used only “when medically appropriate”
(54). In contrast, in the trial by Jeste et al.
(53), the patients had undergone a 3-month lead-in with risperidone or placebo. Notwithstanding these differences in annual tardive dyskinesia rates in elderly patients, however, the reviewed data still support a fivefold reduction in risk for the development of tardive dyskinesia with second-generation antipsychotics in this particularly vulnerable population, compared to reported rates of new-onset tardive dyskinesia in 25%–30% of patients per year with first-generation antipsychotics
(7–
10). It is interesting to note that in the reviewed long-term studies with second-generation antipsychotics, the ratio between annual tardive dyskinesia rates in adults and in elderly patients was quite similar to the ratio for first-generation antipsychotics in those two age groups reported in the literature (i.e., about 1:5), adding face validity to the rates observed in the long-term studies with second-generation antipsychotics.
However, the reviewed data also seem to indicate that the benefits of lower tardive dyskinesia risk with second-generation antipsychotics may be reduced at higher doses. This effect for higher doses is suggested for risperidone by the significant dose effect in one trial
(53) and by the more than fivefold higher tardive dyskinesia rate in the study by Davidson et al.
(54), in which patients received a 3.8 times higher risperidone dose, compared to the dose in the study by Jeste et al.
(53), irrespective of having a lower mean age (73 years versus 82 years). The effect of higher doses is also consistent with the finding that at higher doses second-generation antipsychotics are associated with more extrapyramidal side effects
(12,
13) and that the presence of acute extrapyramidal side effects
(10,
18,
19), as well as utilization of anticholinergic medications
(66,
67), has been associated with an increased risk for tardive dyskinesia. However, the question of whether a similar dose relationship exists for second-generation antipsychotics other than risperidone awaits further study. Finally, as discussed for acute extrapyramidal side effects
(68), similar tardive dyskinesia rates for amisulpride (which does not block serotonin receptors), compared to other second-generation antipsychotics, suggest that serotonin blockade may not be a necessary factor in the reduced risk of tardive dyskinesia associated with second-generation antipsychotics, in contrast to first-generation antipsychotics.
Limitations of the Database
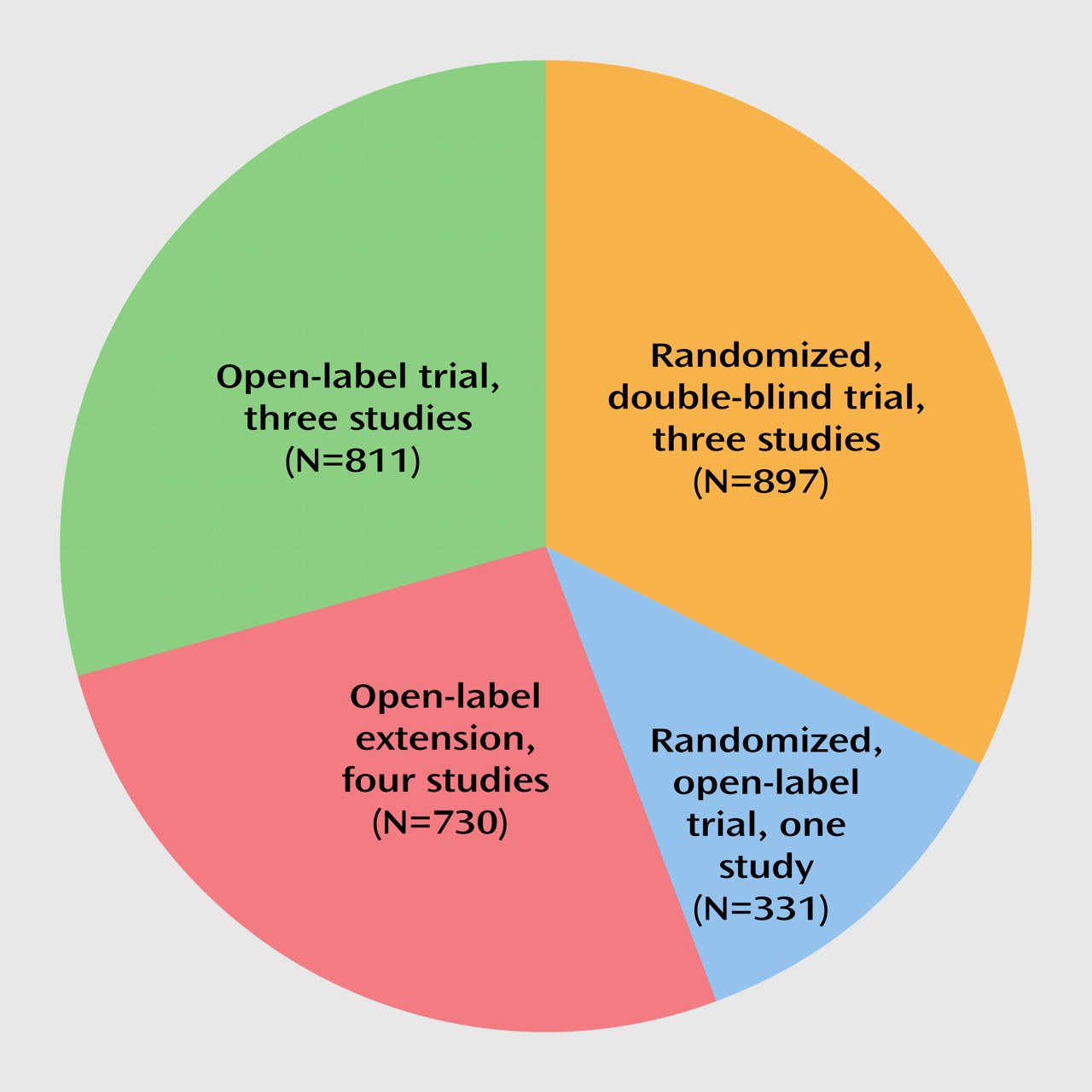
Although the results of the reviewed long-term trials strongly support the assumption that second-generation antipsychotics induce less tardive dyskinesia than first-generation antipsychotics, the available database is still small in light of the wide utilization of these agents, particularly in the United States
(69–
71). Although the subjects in eight studies were randomly assigned to study groups at study entry, only three studies maintained randomization throughout the trial, and only three trials had a blinded design. Furthermore, the comparison with first-generation antipsychotics was based mostly on historical data, as only three of the studies involving adults and none of the studies involving elderly patients included a first-generation antipsychotic (i.e., haloperidol) as an active comparator. However, in the geriatric population the confirmed high risk for tardive dyskinesia in association with first-generation antipsychotics may render a direct comparison in this population unethical. Moreover, in the three active-comparator studies, haloperidol doses were 14.7, 14.6, and 11.7 mg/day, respectively, which is higher than generally considered necessary
(12). Thus, studies using lower doses of first-generation antipsychotics (e.g., 5 mg/day of haloperidol) are necessary to exclude a dose-dependent phenomenon and to confirm the superior safety of second-generation antipsychotics regarding the development of tardive dyskinesia. Furthermore, to our knowledge, there are no studies in which second-generation antipsychotics are compared with low-potency first-generation antipsychotics, even though preliminary data suggest a reduced risk for tardive dyskinesia with phenothiazines compared to haloperidol (adjusted odds ratio=1.56)
(59) and despite the fact that chlorpromazine, which is relatively inexpensive, is still the most widely used antipsychotic in most of the developing world
(72). Finally, none of the long-term studies evaluated the potential confound of an increased tardive dyskinesia risk with haloperidol due to intermittent or interrupted treatment that could lead to withdrawal dyskinesia, given the fact that noncompliance and early medication discontinuation have been found to occur more frequently with first-generation antipsychotics, compared to second-generation antipsychotics
(73,
74), even in controlled settings
(75).
Another complication in interpreting the available data stems from the fact that patients in the reviewed studies had only a weighted mean median exposure of 306 days or 10 months (with mean exposures being considerably lower, whenever reported). This finding implies that the majority of patients did not complete 12 months of treatment, even in the trials that extended beyond 1 year
(44,
46,
50,
51,
54). To account for a potential underestimation of the true 1-year tardive dyskinesia incidence with second-generation antipsychotics, five studies calculated annualized tardive dyskinesia risk estimates using Kaplan-Meier survival analysis, yet five reported only raw incidences of tardive dyskinesia and one reported the raw incidence of dyskinesia. Even though the annualized raw incidences and Kaplan-Meier risk estimates for tardive dyskinesia were not too disparate, at least in the studies involving adult patients, the importance of calculating survival rates is highlighted by the difference between the population-based incidence of 4.3% and the annualized Kaplan-Meier survival probability of 13.6% in the study with risperidone by Davidson et al.
(54). In addition, the lack of explicit exclusion of patients with baseline tardive dyskinesia
(45,
46,
52) could lead to either an underestimation of true tardive dyskinesia rates, as incidences were based on the entire study population, or an overestimation, if patients with tardive dyskinesia at baseline were included. Finally, in the study by Arato et al.
(45), the potential for an underestimation of the true risk was further increased by the rather infrequent follow-up AIMS examinations (at 7 and 13 months), as was the potential for an overestimation because of high rates of withdrawal dyskinesia in the placebo group and a low threshold for caseness.
On the other hand, the true risk for tardive dyskinesia with second-generation antipsychotics may turn out to be even lower than currently assumed, as studies involving patients who have never been exposed to first-generation antipsychotics are still missing. In this regard, the inclusion of patients with subthreshold dyskinetic movements in all available studies introduces a considerable confound for the etiological evaluation of “new-onset” tardive dyskinesia, as tardive dyskinesia may already have been incipient due to previous antipsychotic treatment before the switch to a second-generation antipsychotic. That baseline AIMS scores may turn out to be a relevant factor for the understanding of the true long-term risk for tardive dyskinesia with second-generation antipsychotics is suggested by an important finding in the study by Beasley et al.
(44). When the Kaplan-Meier annualized risk analysis was stratified by using baseline AIMS scores of zero versus scores greater than zero, the risk for development of tardive dyskinesia with olanzapine was substantially lower for patients without dyskinetic movements at study entry than for patients with subthreshold dyskinesia (1.1% versus 6.0% for the overall period and 0% versus 2.0% for the period after week 6, respectively). Conversely, such a stratification by AIMS scores did not significantly alter the risk for tardive dyskinesia in patients treated with haloperidol for the overall trial period (7.1% versus 10.0%) and after week 6 (7.5% versus 6.4%). This finding indicates that for patients treated with haloperidol the tardive dyskinesia risk was independent of whether a pathophysiological process involved in the development of dyskinesia had already begun, whereas it mattered for patients treated with olanzapine. It is interesting to note that, at least in the data set analyzed by Beasley et al.
(44), treatment with olanzapine for a median of 260 days seemed not to have produced any new cases of tardive dyskinesia among 375 patients without preexistent dyskinetic movements, a compelling finding that clearly needs replication.
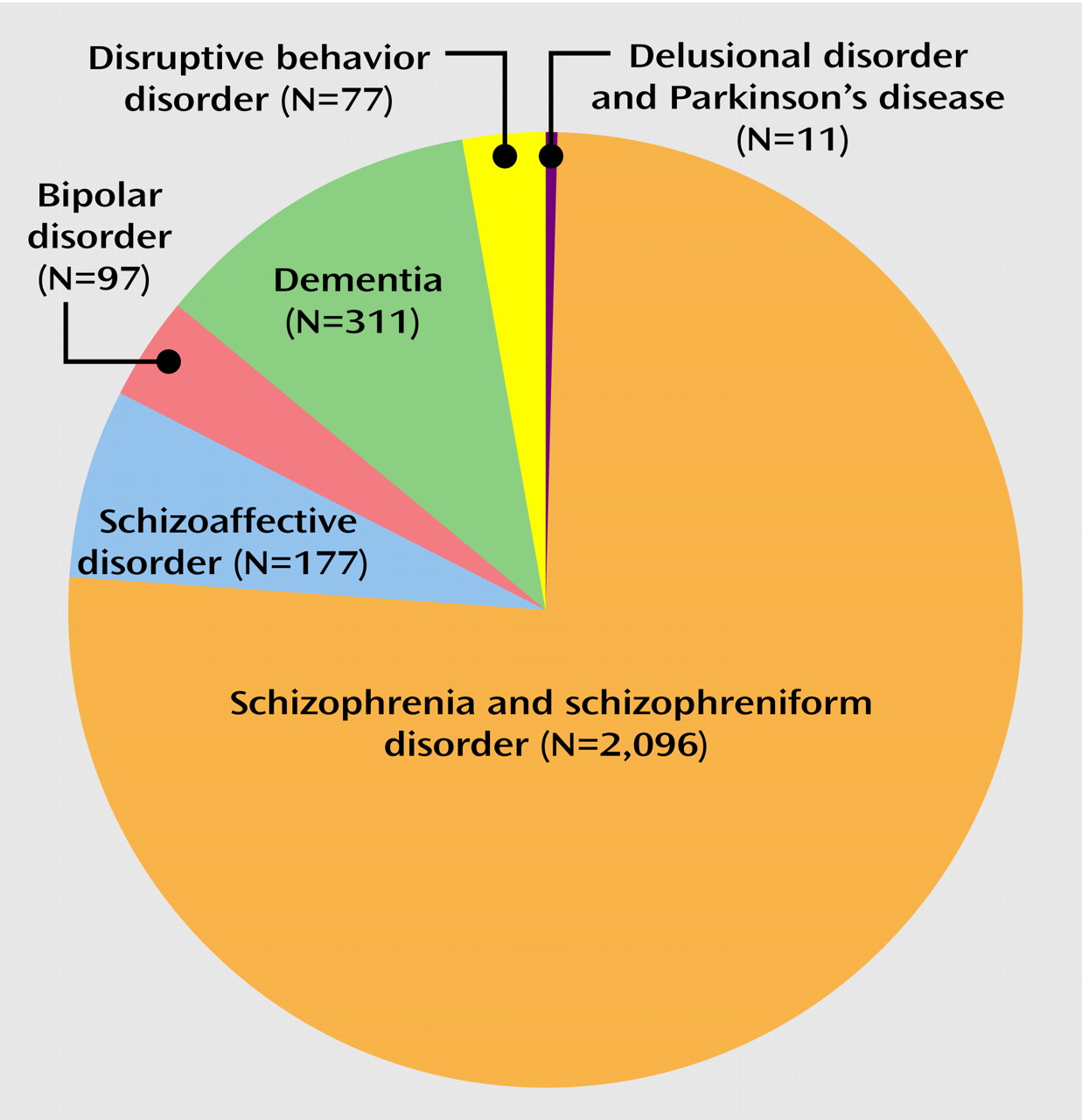
Another limitation of the currently available database is that it does not provide information about the comparative risk for tardive dyskinesia among different second-generation antipsychotics, that sex differences have not been examined, that nonwhite patients are markedly underrepresented (14.2% of subjects in the nine studies with information on ethnicity were nonwhite), and that patients with bipolar disorder or schizoaffective disorder represented only 9.9% of the subjects in the reviewed studies. The latter is in stark contrast to the wide utilization of second-generation antipsychotics as adjunctive or even primary treatment for manic symptoms
(15) and to data suggesting that patients suffering from mood disorders may be at higher risk for developing tardive dyskinesia, compared to patients with schizophrenia
(76). In addition, more and larger-scale long-term studies are needed to examine risk in children and adolescents, who despite the favorable findings in the one available study
(50) may represent a particularly vulnerable patient group
(77,
78) but for whom second-generation antipsychotics are used widely in treatment of psychotic, mood, and disruptive behavior disorders
(17). In addition, the quality of the tardive dyskinesia assessments may also vary between the few studies that were conducted in a limited number of sites and with well-trained raters (e.g., the study by Jeste et al.
[53]) and the overwhelming majority of studies that had multiple sites and inevitable difficulties regarding interrater reliability. Finally, we cannot exclude the possibility that studies showing no reduced risk for tardive dyskinesia with second-generation antipsychotics, compared to first-generation antipsychotics, may have been withheld from publication. However, few studies have been conducted with tardive dyskinesia as the primary outcome measure, and, therefore, the available data are usually from clinical trials where tardive dyskinesia was a secondary focus.