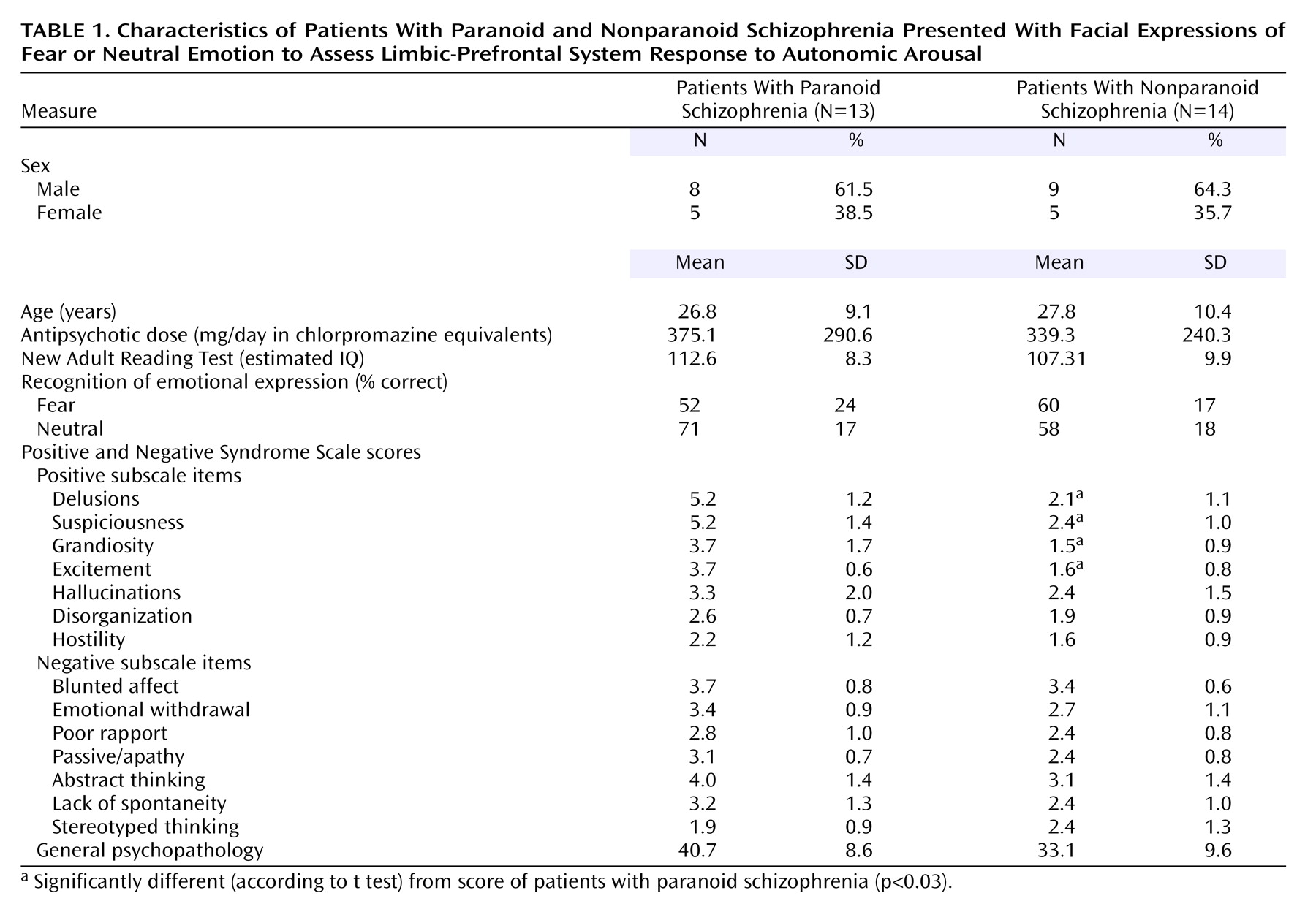
There is a convergence of evidence from neuropsychological, psychophysiological, and neuroimaging studies that breakdowns in temporolimbic-prefrontal circuits are central to the expression of schizophrenia
(1–
4). Limbic-prefrontal systems show a reciprocal modulatory relationship with autonomic (“body”) arousal via connections with brainstem arousal circuits
(5,
6). Abnormalities in both tonic and phasic autonomic arousal have been observed in chronic, first-episode, and high-risk samples
(7,
8). We investigated limbic-prefrontal and arousal dysfunction in schizophrenia during perception of facial expressions. A growing number of studies have observed that impairments in facial emotion perception in schizophrenia patients are most pronounced for threat-related expressions such as fear
(9,
10). In healthy subjects, fear stimuli typically evoke limbic and prefrontal activity, with preferential engagement of the amygdala
(11–
14).
In a previous functional magnetic resonance imaging (fMRI) study of healthy subjects, we used simultaneous recording of skin conductance responses to examine the differentiation of limbic-prefrontal systems by autonomic arousal
(14). Amygdala and
medial prefrontal activity was associated specifically with fear stimuli that evoked a phasic skin conductance arousal response. This pattern of activity may represent a “visceral” system subserving the subjective appraisal of threat
(15–
19). By contrast, distinct hippocampus
lateral prefrontal activity was elicited by stimuli that did not evoke arousal responses and may represent a “context” system for integrating the declarative context of emotionally significant stimuli
(16,
19,
20). To date, schizophrenia deficits in the engagement of these limbic-prefrontal circuits have not been examined in relation to autonomic arousal.
In this study, we applied the technique for simultaneous fMRI and skin conductance recordings in a comparison of schizophrenia patients and a larger group of healthy comparison subjects. Schizophrenia was considered in terms of paranoid and nonparanoid subgroups, given our previous observation that neural responses to fear differ across these subtypes
(21). Our predictions drew on neurophysiological evidence for a disjunction in autonomic and central responses to emotion in schizophrenia
(7). We hypothesized that schizophrenia patients would show dysregulation most apparent in the visceral system: abnormally enhanced arousal (reflecting a heightened autonomic sensitivity to fear) but reduced activity in amygdala-medial prefrontal regions. Given the threat-related emotional content of paranoia, we predicted that this disjunction would distinguish paranoid patients in particular.
Discussion
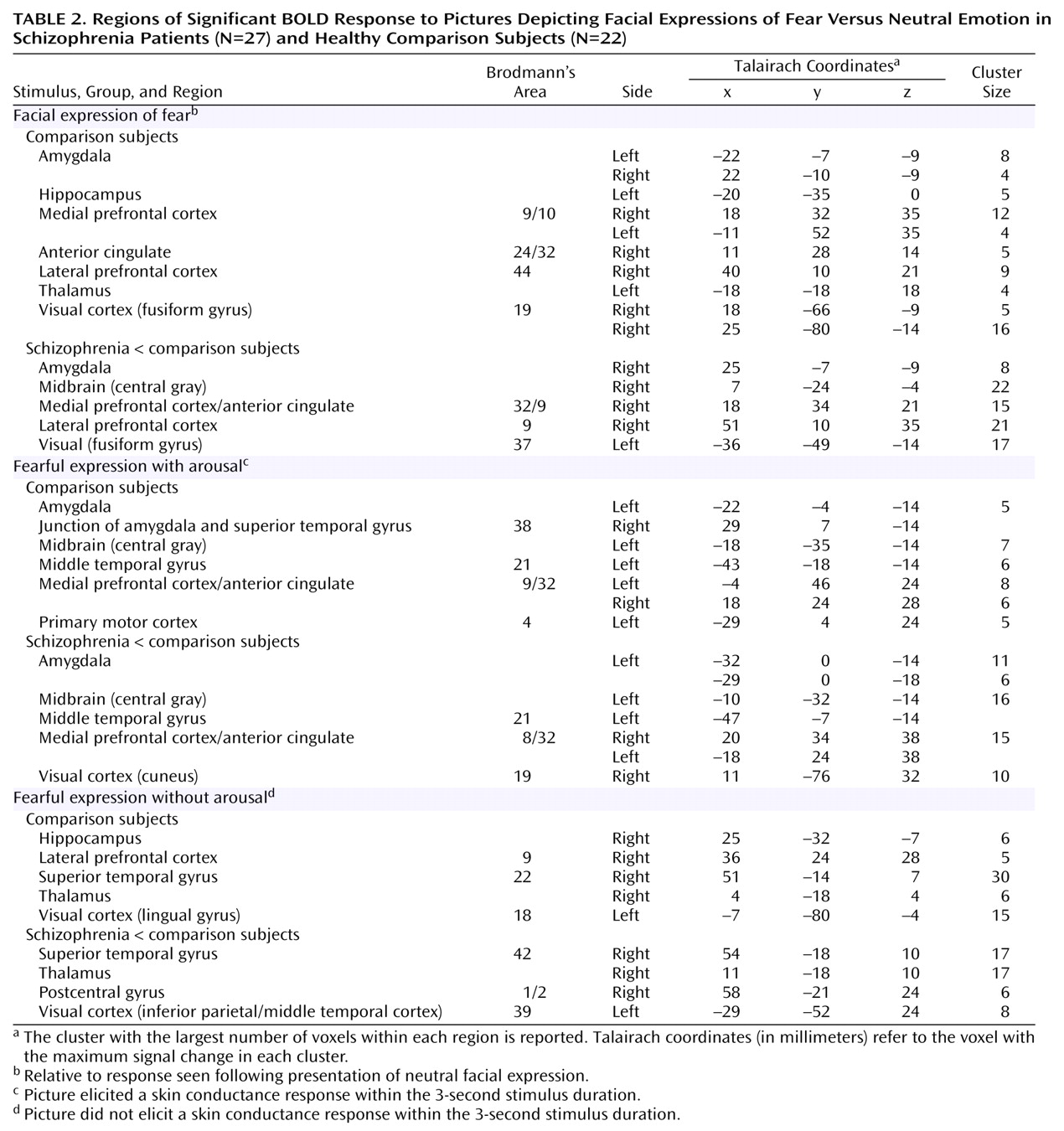
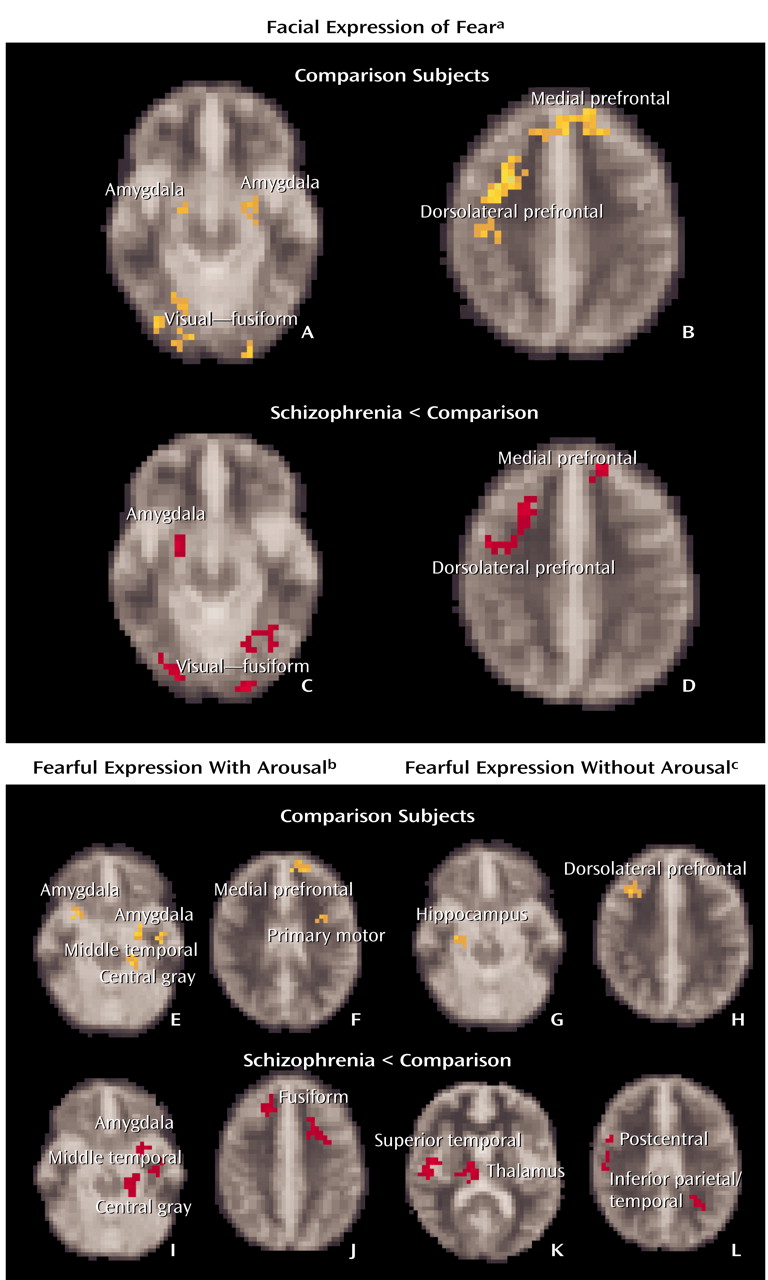
Consistent with previous findings
(11–
14), implicit processing of fearful faces in healthy subjects was subserved by activity in limbic, prefrontal, and visual brain regions. Skin conductance arousal responses differentiated distinct “with-arousal” amygdala/medial prefrontal activity from “without-arousal” hippocampus/lateral prefrontal activity. Amygdala activity was also associated with responses in the anatomically connected central gray region, which may reflect functional projections to the autonomic networks via the brainstem
(6,
15). These networks accord with our earlier finding, in a smaller sample, that arousal dissociates a “visceral” from “context” system
(14). Medial prefrontal involvement may allow for the cognitive appraisal of visceral input and subsequent decision making
(18,
19). By contrast, the
lateral prefrontal cortex receives considerable innervation from the hippocampus (with only meager amygdala connections) and may subserve processing of stimulus context in working memory
(16,
20).
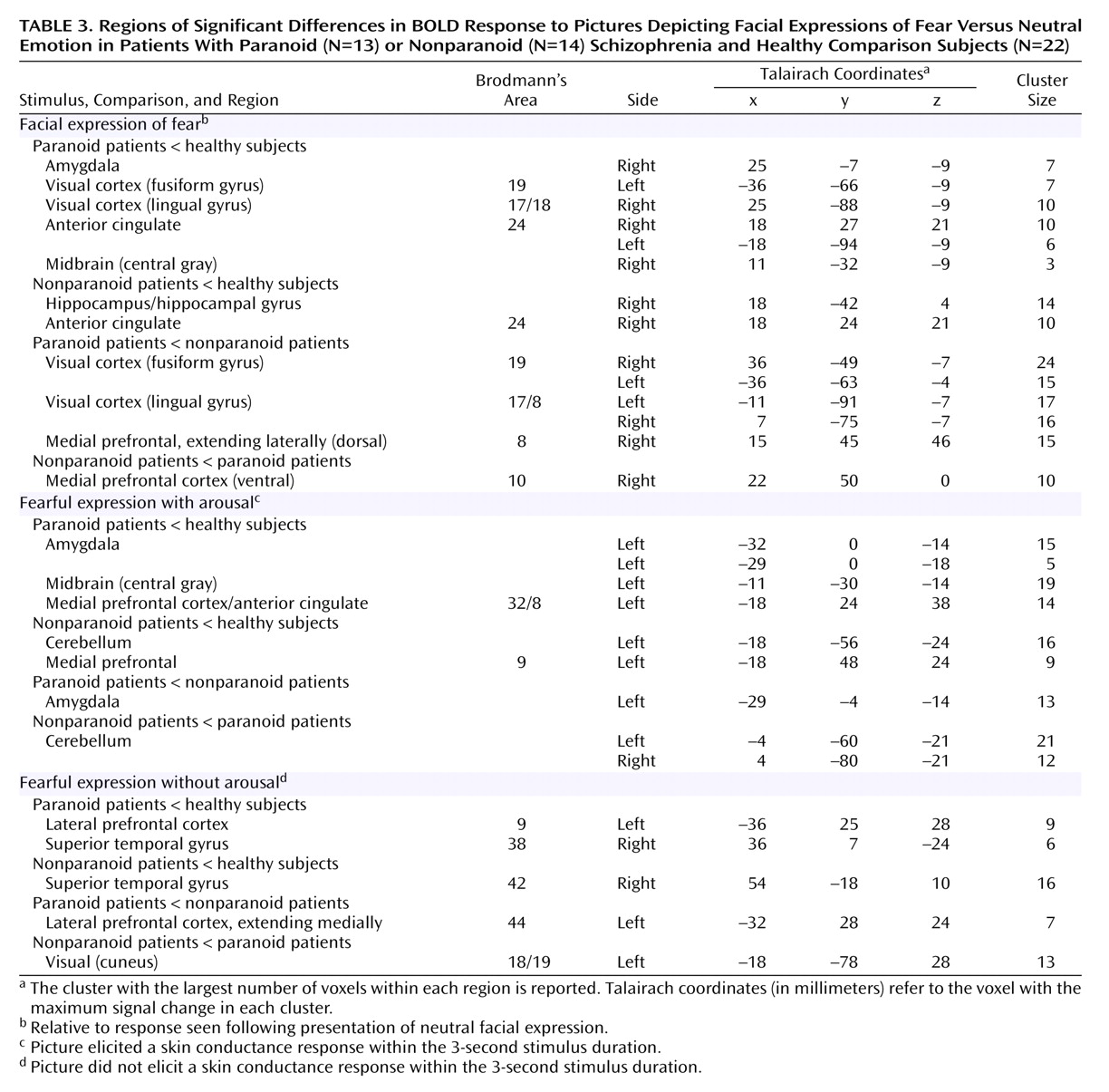
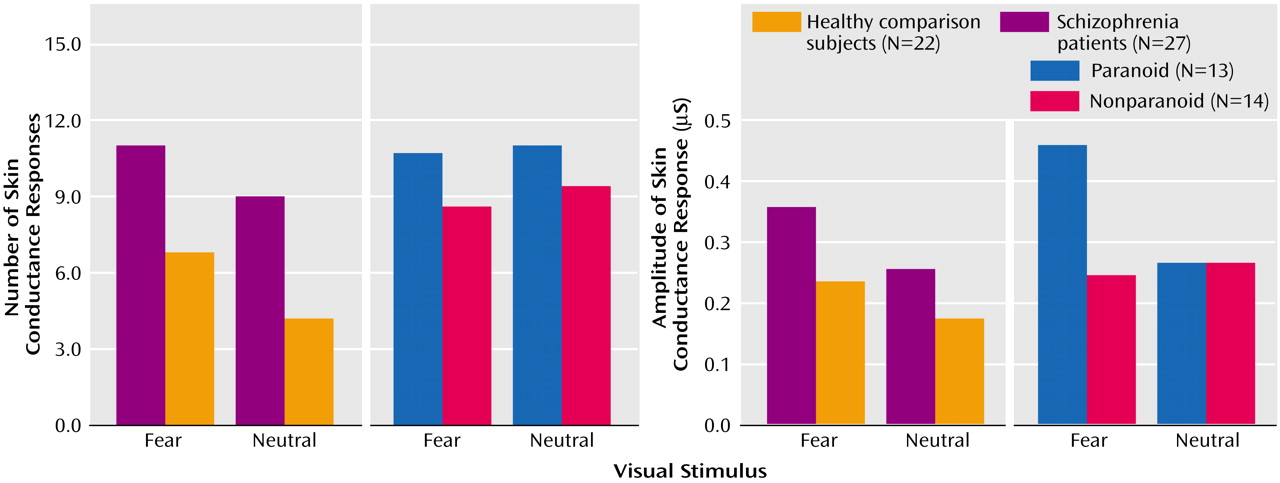
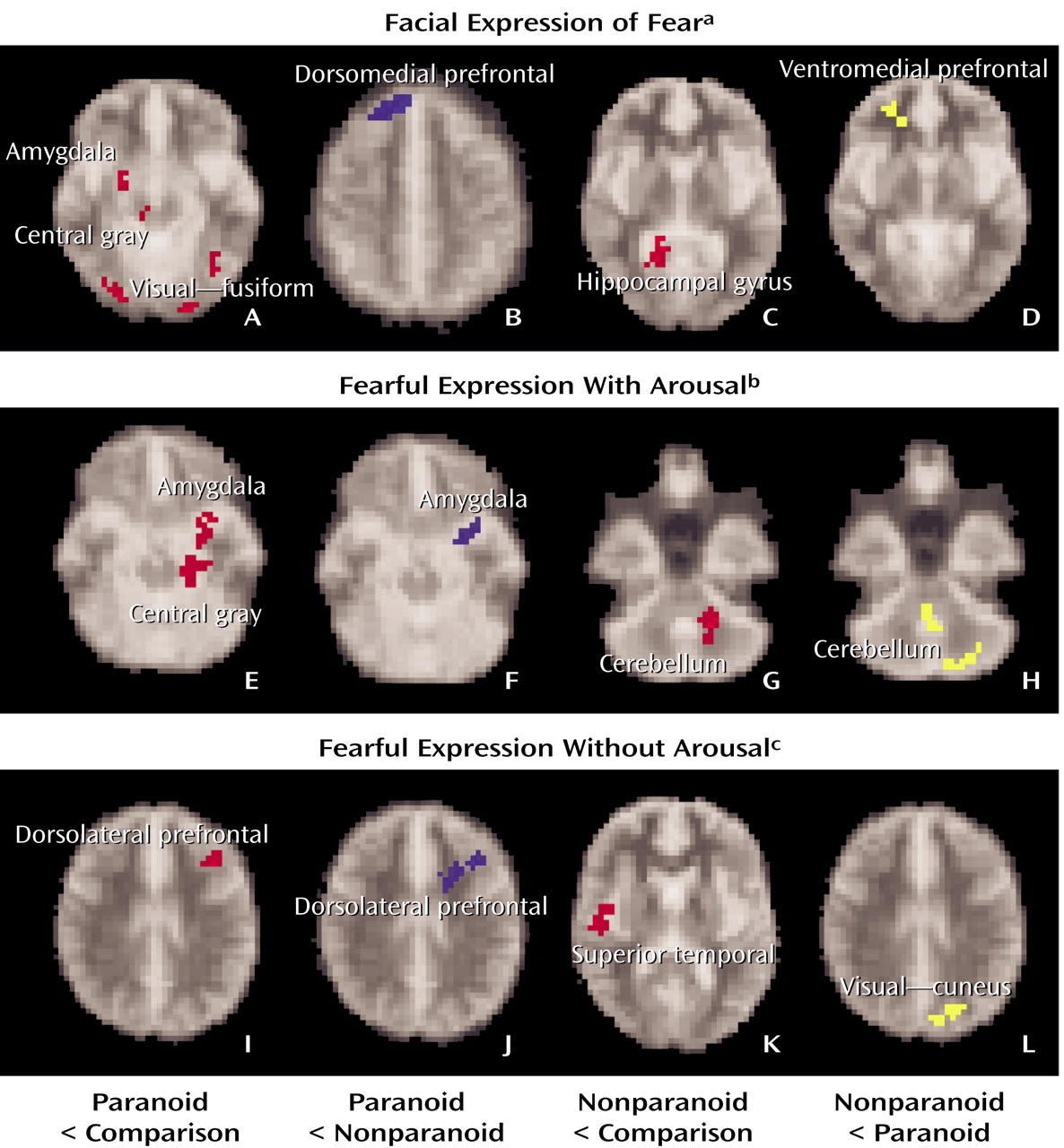
Schizophrenia impairments were due primarily to a dysfunction of the visceral network, which was most pronounced in paranoid patients. Notably, paranoid patients produced excessive arousal responses relative to both comparison subjects and to nonparanoid patients, suggesting a heightened autonomic responsivity to threat-related signals in this group. By contrast, arousal responses for nonparanoid patients were similar to those for healthy comparison subjects. Despite enhanced arousal, paranoid patients showed a reduction in “with-arousal” amygdala, central gray area, and dorsomedial prefrontal activity. In these patients, there was a particularly marked reduction in “with-arousal” amygdala activity relative to both healthy subjects and nonparanoid patients.
Nonparanoid schizophrenia patients also showed a reduction in “with-arousal” medial prefrontal activity (in this case, the ventral portion) but were distinguished by a “with-arousal” reduction in cerebellar, rather than amygdala, activity. There was a further distinctive reduction in hippocampal gyrus activity for nonparanoid patients in the general averaged response to fear. This pattern suggests that nonparanoid schizophrenia impairments in emotion perception do not reflect a specific dysfunction of the visceral network. Rather, they may be due to a general inability to coordinate and contextualize salient stimuli via cerebellar-hippocampal-prefrontal circuits, which is independent of any abnormalities in feedback from autonomic arousal. This proposal accords with neurophysiological evidence that the failure to contextualize task-relevant signals is apparent in frontal brain regions in negative symptom schizophrenia during periods of phasic arousal
(23).
For “without-arousal” stimuli, schizophrenia patients as a group did not show specific reductions in the context (hippocampus/lateral prefrontal) network. However, paranoid patients were distinguished by a reduction in the lateral prefrontal cortex (extending medially) relative to both healthy subjects and to nonparanoid patients. This reduction suggests that paranoid patients may have an additional deficit in the integration of threat-related signals in working memory. This deficit may reflect a consequence of the ineffective processing of these incoming signals via the visceral network. Both paranoid and nonparanoid patients showed an impairment in “without-arousal” superior temporal gyrus function, consistent with the impairment in general face and emotion processing observed in these patients
(13).
Taken together, the findings suggest that paranoid schizophrenia is characterized by a specific disjunction of arousal and amygdala-prefrontal circuits that leads to impaired processing of significant, particularly threat-related, signals. The pattern of excessive arousal but reduced amygdala activity in paranoid patients points to a dysregulation in the normal cycle of mutual feedback between amygdala function and somatic state (autonomic activity). The concomitant lack of “with-arousal” medial prefrontal engagement suggests that this region cannot undertake its usual role in regulating amygdala-autonomic function, leading to a perseveration and exacerbation of arousal responses
(18,
19). A functional breakdown of autonomic-amygdala-prefrontal systems could readily lead to an internally generated cycle of hypervigilance and misattribution that feeds into paranoid cognition
(31).
Given evidence for habituation of amygdala responses
(32,
33), group differences in habituation might be considered as an alternative account of the present findings. However, the abnormally high number and magnitude of skin conductance responses in schizophrenia subjects, and their spread across all trials and face stimuli, indicated that we might discount a simple hypothesis of greater amygdala habituation in these individuals. Previous skin conductance response studies have also reported a comparative failure of habituation in schizophrenia and in “at-risk” samples
(34,
35).
Combined functional and structural imaging studies are warranted to explore the possible role of structural deficits
(1) in these limbic-prefrontal disturbances. Consideration of more specifically defined syndromes and gender differences with a larger sample is also warranted, given evidence from neuroimaging, neuropsychology, and neurophysiology studies that brain function and structure differ across both sex and symptom profile
(1,
23,
36). In addition, this study demonstrates the value of the concurrent fMRI-skin conductance response technique for future investigations of treatment in schizophrenia. The arousal-inhibiting effect of atypical antipsychotic treatments was a key factor in initiating their development, and these agents are designed to more specifically target dopamine receptors in the limbic system
(37). The approach employed in this study provides a means to study both the arousal and limbic-prefrontal effects of atypical medications within a single paradigm.