There has been evidence suggesting that several neuropsychological deficits found in schizophrenia may be associated with parietal lobe dysfunction
(1–
3). These abnormalities are associated with problems in attention, perception, affect recognition, and visuospatial processing
(2–
5), functions subserved by the inferior parietal lobule as part of a circuit in conjunction with other, particularly frontal, brain regions.
The inferior parietal lobule, also referred to as “posterior parietal cortex”
(6), consists of the supramarginal and angular gyri, which correspond approximately to Brodmann’s areas 39 and 40
(7). It is part of the heteromodal association cortex
(6,
8,
9), the regions of which are the last to evolve and mature and comprise higher-order cortical circuits that receive and process information from primary motor and sensory areas, as well as other unimodal and polymodal association cortical regions
(10,
11). The inferior parietal lobule also has extensive interconnections with the limbic system and hypothalamus
(12).
Heteromodal association cortex areas are usually highly lateralized (
figure 1), with the region in one hemisphere having greater area or volume than the other
(9,
13). These regions also exhibit normal sex differences, especially with regard to asymmetry
(9,
13–
24). We have previously shown that the left inferior parietal lobule is larger in healthy male subjects than in female comparison subjects
(13,
25). As the inferior parietal lobule is involved in visuospatial processing, in which men tend to outperform women
(26), such sex-based brain differences may mediate some of the normally observed subtle cognitive differences
(27–
30).
Several of the cognitive tasks mediated by the parietal lobe appear to be lateralized. For example, the right parietal lobe may be more involved in processing affect
(31–
33) and relationships between body parts
(34). In contrast, the left inferior parietal lobule is more involved in cognitive tasks related to perception and visuospatial processing
(35–
40).
Several functional neuroimaging studies in schizophrenia have demonstrated that parietal regions exhibit reduced regional cerebral blood flow (rCBF) in resting states
(2,
32,
41,
42) and a failure to deactivate rCBF appropriately during cognitive tasks
(43,
44). Some of these activation/deactivation patterns appear to lateralize
(44) or relate to symptoms
(2,
45). These rCBF patterns are often associated with concurrent inappropriate activation/deactivation in other brain regions, particularly frontal areas, which implies that the inferior parietal lobule is part of a functional circuit that is disturbed in patients with schizophrenia
(43).
Differential disease expression between the sexes has been observed in patients with schizophrenia
(46–
55). Both the sex-based brain asymmetries, particularly in heteromodal association cortex regions, seen in healthy subjects and the involvement of the heteromodal association cortex in schizophrenia suggest that disruption of these normal sex-based heteromodal association cortex asymmetries may occur in schizophrenia
(9,
16,
56). Most research that has examined the heteromodal association cortex, however, has focused on frontal and temporal lobe structure and function
(13,
19,
22,
57–
59). By means of a locally developed method for measuring inferior parietal lobule gray matter volume, we previously reported stronger asymmetry in normal male subjects than in normal female subjects
(25). Given this stronger male asymmetry, plus the possible disruptions of normal asymmetries seen with schizophrenia, we hypothesized that in patients with schizophrenia, the normal leftward male inferior parietal lobule asymmetry would be disturbed.
METHOD
Subjects
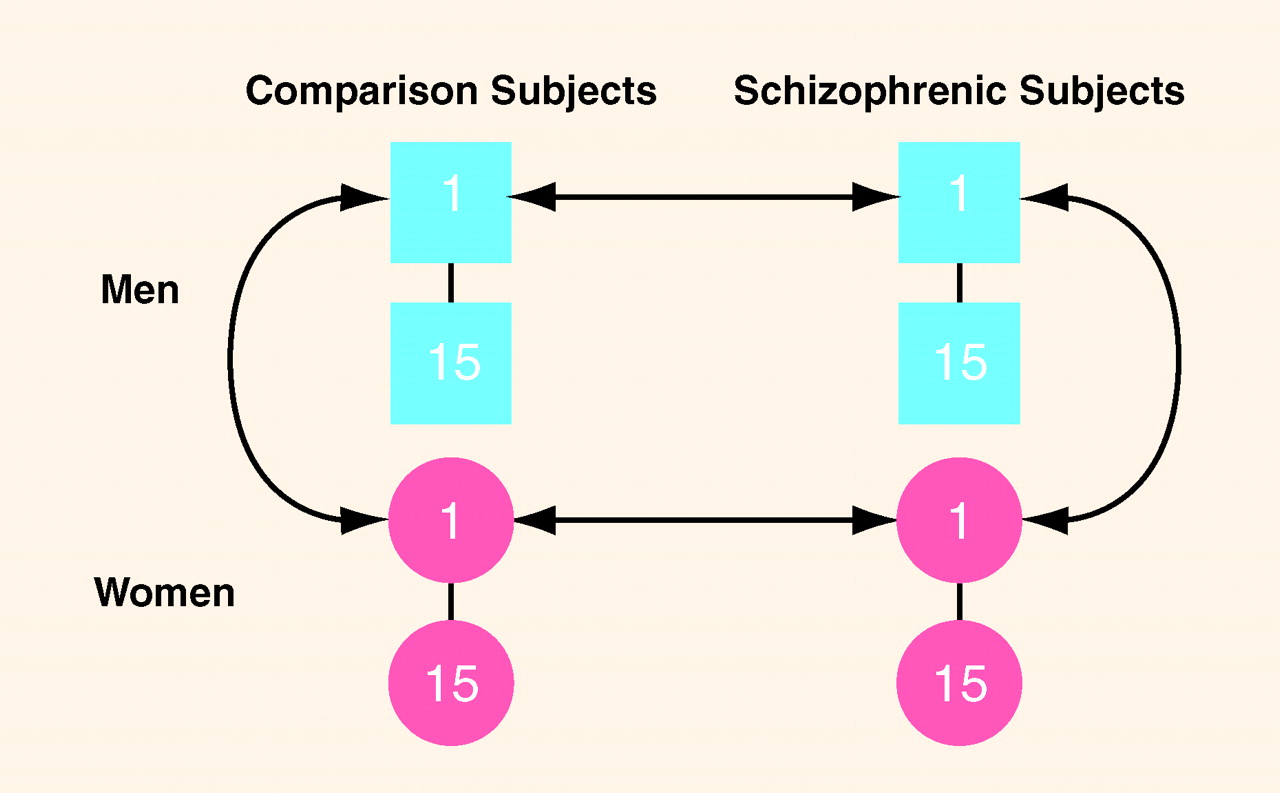
Figure 2 depicts the individual matching design between subjects. We examined a total of 60 subjects in which 15 pairs of normal male and female subjects, individually matched to each other on age (plus or minus 5 years), race, and parental socioeconomic status
(60), were also individually matched on the same variables as well as by sex to 15 pairs of similarly matched male and female subjects with DSM-III-R schizophrenia (as assessed by the SCID
[61]). In terms of diagnostic reliability, all patients continue to be followed by the investigators, and there have been no diagnostic changes. All 60 subjects were right-handed as assessed by the Chapman inventory
(62).
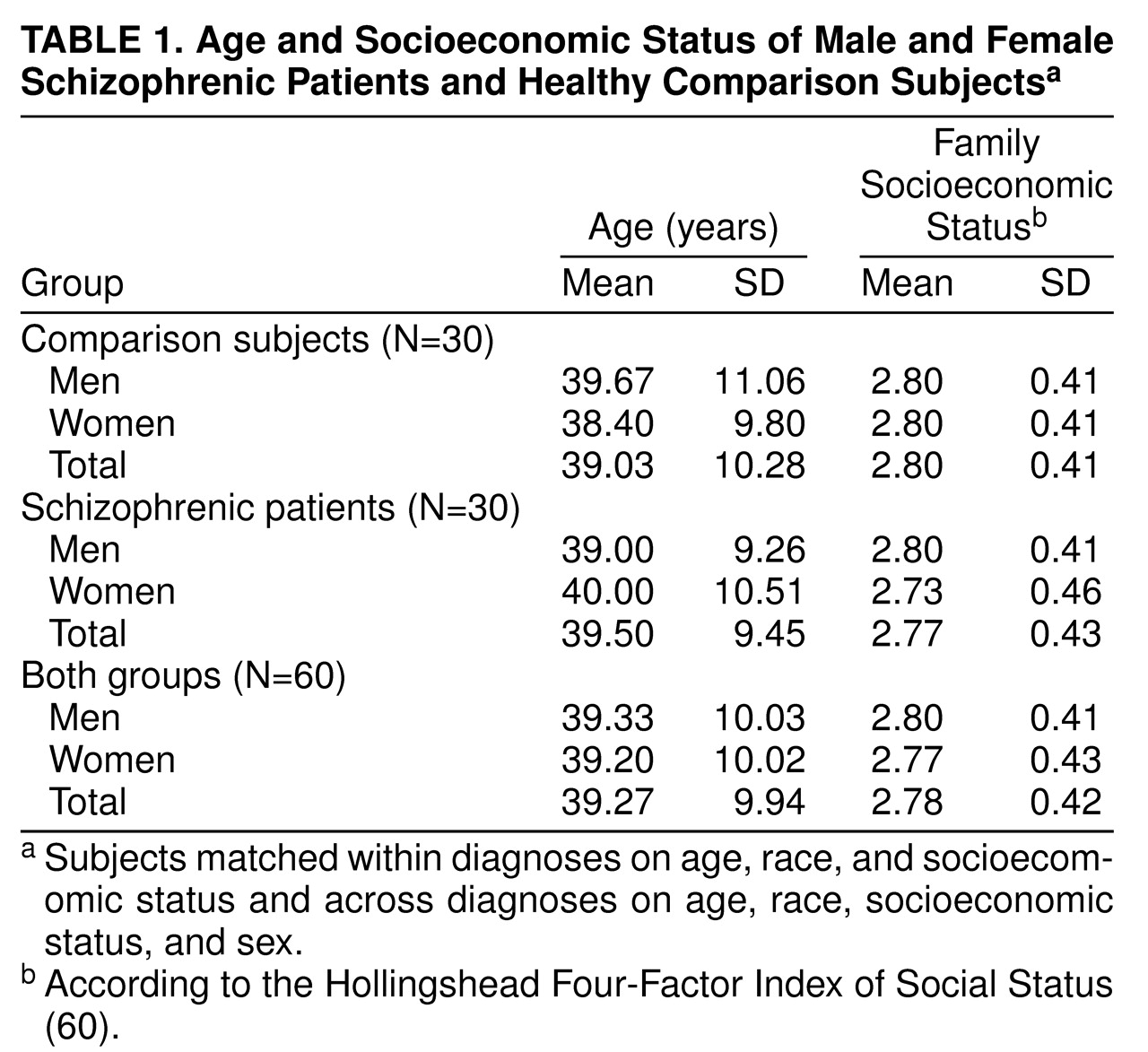
Table 1 outlines group demographic data.
Comparison and schizophrenic subjects were recruited from the community through advertisements (N=18) or had been reached through a random telephone digit dialing protocol as part of a community-based aging study (N=4). Additional subjects were taken from the Epidemiologic Catchment Area study
(63) (N=6) and the Maryland Epidemiologic Sample
(64) (N=10) or were employees of Johns Hopkins Hospital (N=3). In addition, nine schizophrenic and 10 comparison subjects were derived from a parallel study by a collaborator in London (T.S.). Exclusion criteria included history or magnetic resonance imaging (MRI) evidence of overt brain disease as determined by radiologist interpretation; lifetime history of substance abuse/dependence; or any medical illnesses known to affect the brain, e.g., severe head injury with loss of consciousness for more than 1 hour, severe hypertension (e.g., requiring treatment with two or more medications), or clinically significant cardiovascular disease that required medical or surgical treatment. Additional exclusionary criteria for comparison subjects included lifetime history of significant psychopathology or history of major mental illness in a first-degree relative as assessed by a locally developed questionnaire. After complete description of the study to the subjects, written informed consent was obtained from all subjects in accordance with institutional internal review board standards.
MRI Protocol
Coronal MRI scans from throughout the entire brain were obtained by means of high-resolution contiguous 1.5-mm thick spoiled gradient recall acquisition in the steady state (TE=5 msec, TR=35 msec, field of view=20 or 24 cm, image matrix=256 × 256 pixels, flip angle of 45°) on one of three Signa 1.5-T units that used the same General Electric software. An approximately equal number of patients and comparison subjects were imaged on each scanner. Scans were then transferred from the MRI archive and stored on CD-ROMs.
Image Processing and Measurement
All raters were blind to subject sex and diagnosis. By using the software developed in our laboratory (Measure)
(65), raters stripped all brains of skull and dura by means of reliable semiautomated techniques
(59,
66) and rendered realistic three-dimensional brain images, which enabled easy visualization of sulcal-gyral patterns. All brains were aligned along the anterior-posterior commissural line and the interhemispheric fissure.
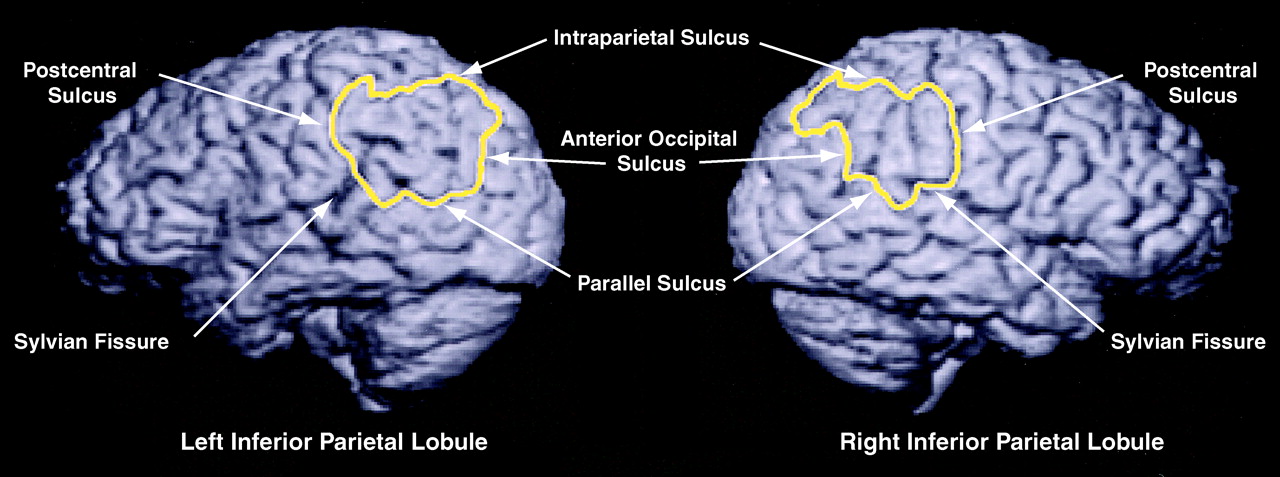
By means of a “paint and point-counting” method described elsewhere
(25,
59), the inferior parietal lobule was first delineated by painting the sulcal and gyral landmarks on a three-dimensional rendering of the cortical surface. The anterior boundary was the postcentral sulcus and the superior boundary was the intraparietal sulcus. The inferior boundary consisted of 1) the Sylvian fissure from the postcentral sulcus to the posterior lateral edge of the planum temporale, 2) a plane that passed through this posterior lateral edge and the temporo-occipital incisure to the superior temporal (or parallel) sulcus, and 3) the parallel sulcus to its horizontal segment (anterior occipital sulcus) and its connection with the intraparietal sulcus.
The spoiled gradient recall acquisition data set was filtered by using locally developed anisotropic diffusion filtering software (K=1.5 × average sigma value of 10 random values within the caudate nucleus; number of iterations=3) to better visualize the gray-white boundary
(67). A three-dimensional grid of points, spaced 4.5 mm apart and yielding approximately 200 points per inferior parietal lobule, was superimposed on the entire volume
(25). “Paint” that demarcated the inferior parietal lobule anatomical borders was then superimposed on the filtered image set, and gray matter points lying within the painted borders were selected. This stereological volume estimation method (Cavalieri) has been discussed in previous publications
(59,
65). Because of imprecise white matter boundaries for this and other cortical regions, inferior parietal lobule white matter volumes were not obtained. As reported previously
(25), intra- and interrater reliability of the inferior parietal lobule volume stereological measurements separately yielded intraclass correlation coefficients of 0.98 in five randomly selected brains.
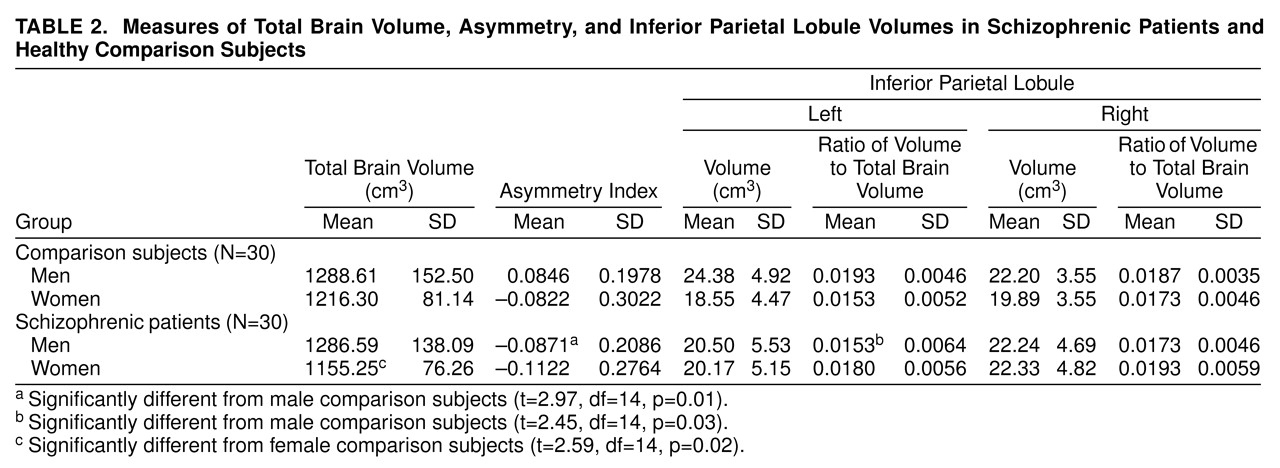
In addition to total brain volume and volumes of the left and right inferior parietal lobules, asymmetry indexes of inferior parietal lobule volume ([left – right]/0.5[left + right]) and ratios of left and right inferior parietal lobule volumes to total brain volume were calculated for each subject and are presented in
table 2.
Statistical Analysis
Given our previous finding of sex differences in the inferior parietal lobule in normal subjects
(25), we compared subjects within the same sex group so as not to obscure potentially important disease state findings. In order to retain the greatest statistical power from our individually matched group, we conducted paired-samples t tests for total brain volume, asymmetry index, and ratios of left and right inferior parietal lobule volume to total brain volume within each sex group. As asymmetry indexes were unitless and were derived from the mean inferior parietal lobule volumes, no total brain volume correction was required.
Chi-square analyses or t tests, as appropriate, were used to examine group differences on demographic variables. Analyses of variance (ANOVAs) of total brain volume, left and right inferior parietal lobule volumes, and asymmetry indexes were performed to examine main effects of MRI scanner.
RESULTS
Neither the male versus female nor the schizophrenic versus comparison groups differed significantly on age, race (86.7% of the entire group was Caucasian), or family socioeconomic status (
table 1). ANOVA showed no significant main effect of MRI scanner on total brain volume (F=0.04, df=2, 59, p=0.97), left inferior parietal lobule volume (F=0.22, df=2, 59, p=0.81), right inferior parietal lobule volume (F=0.12, df=2, 59, p=0.88), or asymmetry index (F=0.52, df=2, 59, p=0.60).
Results of the statistical analyses for the measurement data are summarized in
table 2. Regarding our primary hypothesis, male schizophrenic patients differed significantly from male comparison subjects on asymmetry index and the left inferior parietal lobule to total brain volume ratio, but not on total brain volume or the right inferior parietal lobule to total brain volume ratio. Female schizophrenic patients differed significantly from female comparison subjects only on total brain volume.
DISCUSSION
This structural MRI study provides evidence of inferior parietal lobule abnormality in patients with schizophrenia that appears to be mediated by sex. Previously, we had shown that inferior parietal lobule volume in healthy men exhibited a left-greater-than-right asymmetry and that left inferior parietal lobule gray matter volumes were significantly larger in men than in women (25). Conversely, this study demonstrated that male schizophrenic subjects had significantly smaller left inferior parietal lobule volumes than healthy male comparison subjects, as well as a reversal of the normal left-greater-than-right asymmetry. Whereas female schizophrenic patients had significantly smaller total brain volumes than healthy female comparison subjects, these differences were not accompanied by significant differences in left or right inferior parietal lobule volume or asymmetry. Hence, in patients with schizophrenia, disruption of the normal inferior parietal lobule asymmetry appears to be limited to men only. To our knowledge, this is the first study to demonstrate such a finding. Such sexual dimorphisms may underlie some of the sex-based cognitive and clinical differences observed between male and female schizophrenic patients (46–55).
Given our previous finding in normal subjects of larger left inferior parietal lobule volumes in men versus women
(25), it is important when comparing diseased and healthy subjects to analyze data by sex so as not to obscure potentially important sex-based findings. Closer inspection of
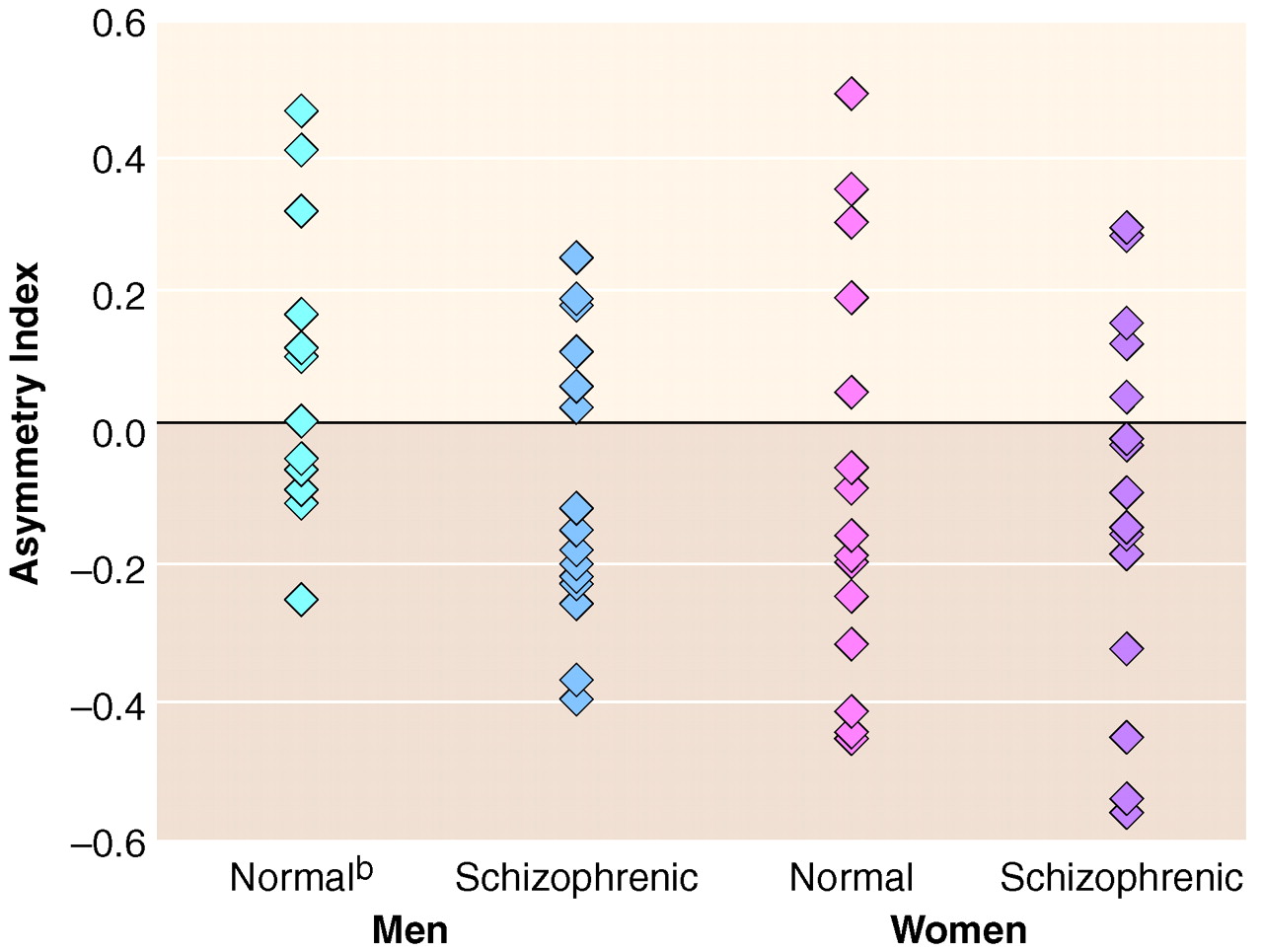
table 2 reveals that combining male and female inferior parietal lobule data for each diagnostic group may lead to the incorrect conclusion of no inferior parietal lobule volume differences in schizophrenia. While not statistically significant, scrutiny of the data also reveals that female schizophrenic patients not only had larger inferior parietal lobule volumes than female comparison subjects, even when corrected for total brain volume, but that female schizophrenic patients exhibited the greatest degree of inferior parietal lobule rightward asymmetry overall (
figure 3). Thus, the inferior parietal lobule may be affected in female schizophrenic patients, and further studies are required.
The inferior parietal lobule is one component of the heteromodal association cortex family of regions that also include the planum temporale, Broca’s area, and the dorsolateral prefrontal cortex
(6,
8,
9). Heteromodal association cortex regions normally exhibit asymmetries
(9,
13), especially with regard to sex differences
(9,
13,
17). In schizophrenia, heteromodal association cortex regions show disruption of these normal asymmetries, as well as volume losses, e.g., in Broca’s area
(32,
45) and the planum temporale
(13,
68,
69). Functionally, activation abnormalities of rCBF in several heteromodal association cortex regions, including the inferior parietal lobule, suggest impairment of a neural circuit in schizophrenia
(43).
Possible deficiencies of this study include lack of white matter volume data and all of the subjects being right-handed. Many asymmetrical brain measures (e.g., planum temporale and radius of gyration) correlate with handedness
(18,
70). Thus, left-handed individuals may show variants of the pattern described here. As for white matter data, precise white matter boundaries for the inferior parietal lobule, or other cortical surface regions, do not exist, and appropriate valid and reliable measurement techniques remain to be developed.
As we have noted, the inferior parietal lobule is involved in neurological functions related to attention, perception, and visuospatial processing, with studies demonstrating hemispheric lateralization of these functions. For example, the left inferior parietal lobule appears to mediate visuospatial functions such as mental rotation of three-dimensional figures
(38) and judgments of target speed
(35) and position
(39). Studies have also demonstrated that normal male subjects tend to perform better than normal women on visuospatial tasks
(26).
In schizophrenia, male/female comparisons of cognitive functions have provided mixed results. Men with schizophrenia tend to develop the disorder earlier
(47) and exhibit more negative symptoms
(48,
53,
55), poorer social functioning
(46,
49,
50,
52), and more abnormal neurologic signs (51). They may also exhibit more cognitive impairment
(46,
71), although other studies do not report this
(54,
72–
74). While our study was conducted in a very well-matched subject population and used a highly reliable measurement method, the lack of accompanying detailed neuropsychological and clinical data makes direct comparison between brain findings and clinical symptoms speculative. Future studies (e.g., functional neuroimaging studies) that assess activation during cognitive tests specific to parietal regions, with an emphasis on sex differences and lateralization in normal and disease states, would be useful.