Several studies have demonstrated morphological and functional changes in the thalamus of patients with schizophrenia. The thalamus is the vital relay station to the cortex and is important for consciousness, perception, and the integration of thought processes
(6). An aberrant thalamus could contribute to the perceptual and motor dysfunction of schizophrenia
(7). It has been postulated that abnormal prefrontal-thalamic-cerebellar circuitry may contribute to the cognitive impairment characteristic of the disease
(5,
8). Cell loss and reduced tissue volume of the thalamus have been described in several studies of schizophrenic brains
(9–
13). Furthermore, imaging studies have consistently revealed smaller thalamic volumes in patients with schizophrenia as well as altered thalamic perfusion and metabolism
(8, 14–
22).
The most recent emphasis on the neurodevelopmental model of schizophrenia
(1–
6) points to the importance of first-episode schizophrenia research. Studies of first-episode patients may reveal the biological basis of psychotic disorders without the potential confounds of prior treatment and illness chronicity. Prospective studies of this population can clarify the impact of illness course and treatment on neurobiology
(23). Because a specific thalamic role in schizophrenia remains unknown, and most structural and functional thalamic imaging studies have examined chronic, treated patients, we sought to measure thalamic volume in treatment-naive patients with first-episode schizophrenia. Furthermore, since the thalamus is composed of several nuclei, we conducted the first study to our knowledge that has measured subdivisions of the thalamus in this patient population. Our goal was to better understand the role of individual thalamic nuclei, particularly the dorsomedial nucleus and pulvinar, which have connections to brain regions previously implicated in the pathophysiology of schizophrenia
(6). We hypothesized that the regions mainly comprising the dorsomedial nucleus and pulvinar in patients with first-episode schizophrenia would differ from those of healthy comparison subjects.
Method
Subjects were 16 patients with schizophrenia and 25 healthy comparison subjects. The patients (15 with schizophrenia and one with schizoaffective disorder, depressed type) were diagnosed according to DSM-IV criteria at consensus conference meetings of senior diagnostic/clinical researchers approximately 1 month after entry into the study. All available clinical information and data gathered by using the Structured Clinical Interview for DSM-III-R Axis I disorders (SCID)
(24) were incorporated. All diagnoses were ascertained as part of a longitudinal, prospective study and included a careful diagnostic rereview to ensure diagnostic stability
(23). None of the subjects currently met criteria for DSM-IV psychoactive substance abuse or dependence; two subjects had a prior history of substance abuse and were dropped from the study. None of the patients had histories of significant head injury (with loss of consciousness of more than 10 minutes), neurological or medical illness, prior neuroleptic treatment, or mental retardation (IQ lower than 75). Illness duration was computed from the date of onset of prodromal symptoms to entry into the study. Clinical ratings were determined by using the Brief Psychiatric Rating Scale (BPRS)
(25).
The healthy comparison subjects were recruited through advertisements in the same local neighborhoods and communities in which the patients resided. These subjects were free from any current or past axis I disorder (including substance abuse or dependence) as determined by the nonpatient version of the SCID. None had any prior exposure to any psychotropic medication within 6 months of the baseline assessment; a history of neurologic disorders or any other chronic medical problems with potential to influence neurological function; or mental retardation (IQ lower than 75). None reported a history of schizophrenia or major mood disorder in a first-degree relative. The comparison subjects were matched, as a group, with the schizophrenia patients for age, gender, race, and parental socioeconomic status (from the Hollingshead Four-Factor Index
[26]). After an initial screening evaluation by telephone, these subjects were interviewed by a psychiatrist (M.S.K.) or clinical psychologist with the SCID (nonpatient version).
After complete description of the study to the subjects, written informed consent was obtained. The study was approved by the Biomedical Institutional Board of the University of Pittsburgh.
MRI Studies
All volumetric magnetic resonance imaging (MRI) scans were conducted at the University of Pittsburgh Medical Center (1.5-T Signa Whole Body Scanner, GE Medical Systems, Milwaukee). Image quality and clarity as well as patient head position were determined with a sagittal scout series. Total brain volume was measured with a three-dimensional spoiled gradient recalled acquisition in the steady state pulse sequence that obtained 124 1.5-mm thick contiguous coronal images (TE=5 msec, TR=25 msec, acquisition matrix=256 × 192, field of view=24 cm, flip angle=40°). In order to facilitate image orientation, coronal slices were obtained perpendicular to the anterior commissure-posterior commissure line. Axial proton density and T
2-weighted images were obtained for thalamic measurement and to exclude structural abnormalities on MRI scans. Image software (version 1.56) developed by the National Institutes of Health was used to measure brain anatomy
(27). This technique yields valid and reliable neuroanatomic measurements of the regions of interest with a semiautomated segmentation approach
(28). None of the MRI scans in this data set showed motion or magnetic field inhomogeneity artifacts.
Neuroanatomic Measures
We measured the right and left hemispheres of the thalamus, their subdivisions, and total intracranial volume, which was used as a covariate. We also measured the volume of the dorsolateral prefrontal cortex in view of the implications of this structure in the pathophysiology of schizophrenia
(3,
28) and its reciprocal connections with the thalamic nuclei
(6). Neuroanatomic boundaries were determined by reference to standard neuroanatomical atlases
(29,
30), and detailed definitions were adapted from previously published psychiatric neuroimaging studies of the thalamus
(8,
15,
16, 31–
33).
Thalamic Volume
Separate measurements were obtained for the left and right hemispheres of the thalamus by using a manual tracing technique. The mamillary body was used as the anterior boundary. The internal capsule was considered the lateral boundary, the third ventricle the medial boundary, and the inferior border of the third ventricle the inferior boundary. The posterior boundary was defined by where the hemispheres of the thalamus merged under the crux fornix. The superior boundary was the main body of the lateral ventricle
(32). Measurements were made by a single well-trained and reliable rater (A.R.G.) who was blind to the study hypothesis, subject identification, and clinical data.
Thalamic Subdivisions
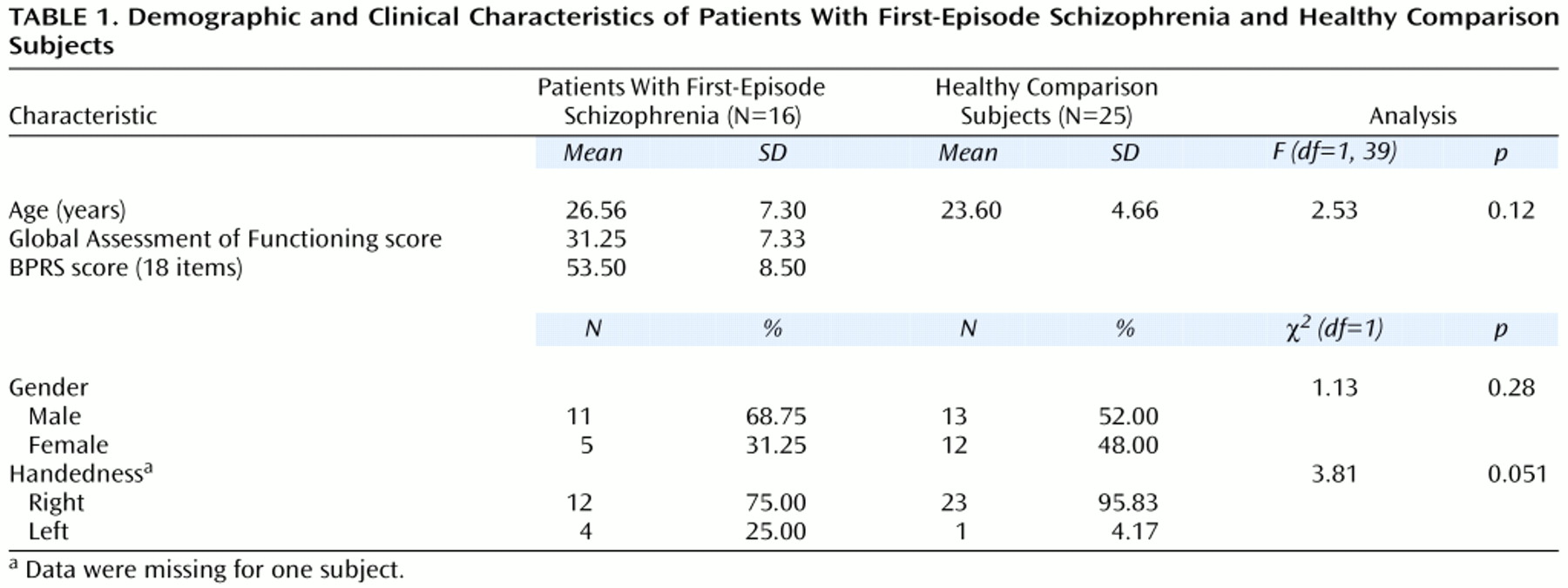
The thalamus was first divided into two distinct medial and lateral regions (axial view in
Figure 1). The point of reference for the line that bisected the thalamus into medial and lateral regions was the emergence of the fourth ventricle as it appeared as a vertical line through the pons. A line drawn parallel to the lateral border of the midbrain, interhemispheric fissure, and cerebral aqueduct represented our line of vertical bisection of each thalamus (coronal view in
Figure 1). This line was carried on through all thalamic slices to create a plane of bisection parallel to the interhemispheric fissure. We chose this approach because no significant differences have been seen between patients with schizophrenia and comparison subjects with regard to brainstem structures
(34). Using the spoiled gradient recalled acquisition protocol, we measured contiguous coronal slices of the thalamus, dividing the regions into anterior, central, and posterior divisions. The anterior and central divisions each contained 40% of the total number of slices; the posterior division contained 20% of the total number of slices (axial and sagittal views in
Figure 1). These divisions are based upon neuroanatomical atlases of the human brain as well as a previously published article examining the thalamus in schizophrenia
(16,
29,
30). The divisions roughly reflect the individual nuclei that are located in these thalamic regions (axial view in
Figure 1). Each individual subdivision was measured as a percentage of the total number of slices. Measurements were made by a single, well-trained, and reliable rater (A.R.G.) who was blind to the study hypothesis, subject identification, and clinical data.
Dorsolateral Prefrontal Cortical Volume
The dorsolateral prefrontal cortex was measured in the coronal plane by a method similar to that used by Seidman et al.
(35). The most posterior part of the genu was located and used as the first slice in measuring the dorsolateral prefrontal cortex. The borders of the dorsolateral prefrontal cortex were first manually outlined, and the gray matter was then segmented. The superior boundary was the superior frontal sulcus, and the inferior boundary was the posterior lateral fissure and the horizontal ramus of the anterior lateral fissure. The lateral border was the edge of the cerebral cortex, and the medial border was created by connecting the deepest points on the superior frontal sulcus and the lateral fissure. Ten successive anterior slices were measured. The dorsolateral prefrontal cortex was segmented, and the gray matter and total volume were measured.
Intracranial Volume
Intracranial volume included the following volumes: total brain, dura, ventricular and extraventricular CSF, brainstem, and cerebellum; this was measured by manually outlining the outer limit of the tracing along the inner border of the inner table of skull. This measurement was made from the coronal slices. The intracranial volume was used as a covariate in comparison to volumetric measures of the aforementioned structures across diagnostic groups.
Statistical Analysis
The thalamic volumes and their subdivisions were compared between subjects by using analysis of covariance, with the intracranial volume as a covariate. The relations between thalamic volumes and subject characteristics (age, symptom severity) were examined by using Pearson correlation coefficients. Interrater (K.H., A.R.G.) reliabilities (intraclass correlations; N=10) for all measurements and intrarater reliabilities for measurements by the same rater (A.R.G.), conducted approximately 1 month apart on 10 scans, were all greater than 0.90.
Results
The two groups did not significantly differ with regard to age, gender, or handedness (
Table 1). The right hemisphere, left hemisphere, and total thalamic volumes of the patient group were significantly smaller than those of the comparison subjects, after we controlled for intracranial volume (
Table 2). Intracranial volume did not significantly differ between the groups. When only right-handed subjects (12 patients and 23 comparison subjects) were examined, there were continuing differences between the groups (left hemisphere: F=9.34, df=1, 33, p=0.004; right hemisphere: F=3.86, df=1, 33, p=0.06; total thalamus: F=7.14, df=1, 33, p=0.01). There were no differences observed when we controlled for handedness.
No significant gender effect was seen for left-hemisphere thalamic volume (F=1.75, df=1, 36, p=0.19), but female subjects had smaller right-hemisphere thalamic volumes (F=4.18, df=1, 36, p=0.04). No gender-by-diagnosis interactions were seen for either structure. Right hemisphere, left hemisphere, and total thalamic volumes, after we controlled for intracranial volume, did not correlate with age in either the patients (partial correlation coefficients<0.25, df=13, p>0.50) or the comparison subjects (partial correlation coefficients<0.25, df=22, p>0.50). In the patient group, thalamic volumes were not correlated with Global Assessment of Functioning scores (r<0.15, df=14, p>0.60) or total BPRS scores (r<0.15, df=14, p>0.60). When the patient with schizoaffective disorder was excluded, thalamic volume differences remained significant (left hemisphere: F=8.53, df=1, 37, p=0.006; right hemisphere: F=5.84, df=1, 37, p=0.02; total: F=8.08, df=1, 37, p=0.007). The number of coronal slices used to quantify the thalamus ranged from 13 to 21 slices, with an average of 17.5 slices. There were no significant differences between groups in the number of slices.
Thalamic Subdivisions
Measurements of thalamic subdivisions were conducted in 16 patients and 25 comparison subjects (
Table 3). The 18% difference in volume for the left central medial subdivision was significant, and the 17% difference in volume for the right central medial subdivision approached significance. There was a highly significant difference in right posterior medial subdivision volume (43% smaller in the patient group). Significant volume differences were also found in the right and left anterior medial subdivisions (13% and 14%, respectively). There were no significant volume differences in the lateral subdivisions.
Dorsolateral Prefrontal Cortical Volume
The dorsolateral prefrontal cortex volumes (gray, white, total) did not differ significantly between patients and comparison subjects. However, the left dorsolateral prefrontal cortex gray matter volume was smaller in the patients than in the comparison subjects (mean=9.94 cm3 [SD=1.35] versus 10.56 cm3 [SD=1.13], respectively) (F=2.86, df=1, 38, p<0.10). No significant correlations were seen between the thalamic and thalamic subdivision volumes on the one hand and the gray, white, and total dorsolateral prefrontal cortex volumes on the other (all partial correlations [after partialing out intracranial volume] <0.50, df=13, p>0.20).
Discussion
Consistent with the results of other early course as well as chronic schizophrenia studies, we have demonstrated significant thalamic volume differences in patients with first-episode schizophrenia relative to those of healthy comparison subjects. Furthermore, by examining treatment-naive, first-episode patients, we have eliminated the potentially confounding effects of prior treatment and illness chronicity.
Thalamic volume differences in patients with schizophrenia may reflect abnormal thalamic circuitry—a potential contributor to the cognitive dysfunction characteristic of schizophrenia. The thalamus is a recognized component of frontal, cerebellar, and limbic circuits
(36–
38). Lesions of thalamic nuclei have been reported to result in neuropsychological and behavioral disturbances similar to the deficits of executive function in patients with schizophrenia. Vascular and degenerative disorders affecting the thalamus are often indistinguishable from “frontal lobe”-type syndromes
(36). With many cortical and subcortical connections, the thalamus is thought to filter sensory information before it reaches the cortex. Therefore, abnormal thalamic function could result in the inability to filter or “gate” sensory input to the cortex, creating the type of sensory overload often experienced by patients with schizophrenia
(5,
39). Furthermore, an aberrant thalamic component to thalamocortical circuitry may contribute to or result from the frontal lobe dysfunction described in previous schizophrenia research
(8). However, our inability to find a correlation between thalamic and dorsolateral prefrontal cortex volume alterations suggests that caution is warranted for such conclusions.
Thalamic volume differences in patients with schizophrenia, suggestive of aberrant thalamic circuitry, are consistent with previous structural and functional studies that have demonstrated abnormalities in other brain regions. Several postmortem studies have revealed differences in superior temporal gyrus cell and total cortical volumes as well as lower prefrontal and anterior cingulate cell densities in schizophrenic brains
(40–
43). Differences in cell number, density, and volume in the mediodorsal thalamic nucleus have been reported as well as lower cell volume in the globus pallidus
(9,
13,
44). Imaging studies have revealed smaller thalamic volumes and less glucose metabolism and neuronal activity in patients with schizophrenia
(15,
16,
20,
21). Furthermore, abnormal striatal
(45,
46) as well as superior temporal gyral volumes
(47) have been observed. Functional imaging studies have consistently demonstrated abnormal perfusion and metabolism in cortical and cerebellar regions of schizophrenic brains
(8, 48–
51). These findings support the concept of abnormal cortical-subcortical circuitry underlying the pathophysiology of schizophrenia. A greater understanding of the thalamic components of these circuits is critical.
We have previously proposed that a developmentally mediated neural dysplasia in cortical-subcortical circuitry may be associated with the pathogenesis of schizophrenia. A substantial reorganization of cortical connections, involving a programmed synaptic pruning, takes place during human development
(28). It has been suggested that in schizophrenia, aberrant migration or programmed cell death in the subplate zone of the developing cortex may impair normal patterns of synaptic connections in the overlying cortex and lead to compromised circuitry
(6). Recent studies have revealed abnormal thalamic projections to the prefrontal cortex and other signs of impaired thalamocortical connectivity in schizophrenia
(52,
53). The primary versus secondary nature of thalamic dysfunction in schizophrenia remains unclear; however, it is understood that neuronal loss in the thalamus is commonly a retrograde degeneration ensuing from pathology in the cerebral cortex that affects the axon terminations of thalamocortical relay cells. Furthermore, retrograde degeneration secondary to cortical damage in the developing brain is typically unaccompanied by gliosis
(6). It is interesting to note that the lower neuronal density described in postmortem analysis of the thalamus occurred in the absence of gliosis, which suggests a neurodevelopmental rather than neurodegenerative process
(8).
While smaller thalamic volumes in patients with schizophrenia has been a consistent finding in both in vivo and postmortem studies, the majority of this research has examined chronic or treated patients
(9,
14,
15,
17,
20,
54,
55). We have demonstrated that caudate volumetric measurements may differ between neuroleptic-naive and treated schizophrenia patients, perhaps secondary to the effects of neuroleptic treatment
(28). In light of these potential treatment-induced changes, we sought to examine patients with untreated schizophrenia. It is possible that, similar to our caudate findings
(28), volumetric measurements following 1 year of antipsychotic treatment may reveal significant treatment effects. A previous study
(18) revealed no change in thalamic volumes, as compared to control subjects, in five schizophrenia patients following approximately 70 days of risperidone treatment, although higher doses of atypical neuroleptics were associated with larger thalamic volumes. It was suggested that medication-induced hypertrophy may have been responsible for the volumetric changes.
The inability to precisely measure discrete thalamic nuclei limits the interpretation of our findings. Our current measurements of thalamic subdivisions revealed significant and nearly significant volume differences in the regions primarily comprising the dorsomedial nucleus. As previously mentioned, postmortem analysis of schizophrenic brains has revealed a significant neuronal deficit in the dorsomedial nucleus. It is of interest that these changes appeared to be unaffected by treatment
(9). Because of its connections to the prefrontal cortex, medial temporal cortex, and basal forebrain, all of which have been implicated in the pathophysiology of schizophrenia, the dorsomedial nucleus is thought to be an important component of aberrant circuitry
(6). We also found significant volume differences in the right posterior medial subdivision, a thalamic region roughly reflecting the right medial half of the pulvinar. The pulvinar may also represent a critical thalamic nucleus in that it projects to temporal lobe regions reported to be abnormal in patients with schizophrenia and might be associated with altered visual perceptual functions
(6,
42,
47).
It is important to note that our measurement of thalamic subdivisions has several limitations. We were unable to delineate and employ an intrathalamic marker as a consistent landmark for our regional subdivisions. Rather, we relied upon approximate percentage-based divisions of total thalamic area as a means of dividing the thalamus. It is possible that our approach may not account for thalamic volume loss that is inconsistent with these percentage-based divisions. Our approach assumes that all thalamic nuclei will be consistently represented by these rigid subdivisions. It is possible that some patients have thalamic nuclei that are not in proportion to the percentage-based parameters that we defined in this study. Furthermore, an enlarged third ventricle causing a mass-effect shift in thalamic volume could be misinterpreted as thalamic volume loss. Higher resolution imaging may allow for the delineation of an intrathalamic marker that may improve the reliability of our approach. Furthermore, by using more advanced imaging techniques, such as diffusion tensor imaging
(56), it may be possible to delineate the white matter tracts that connect thalamic nuclei with their cortical projections and better understand the role of these circuits in schizophrenia.