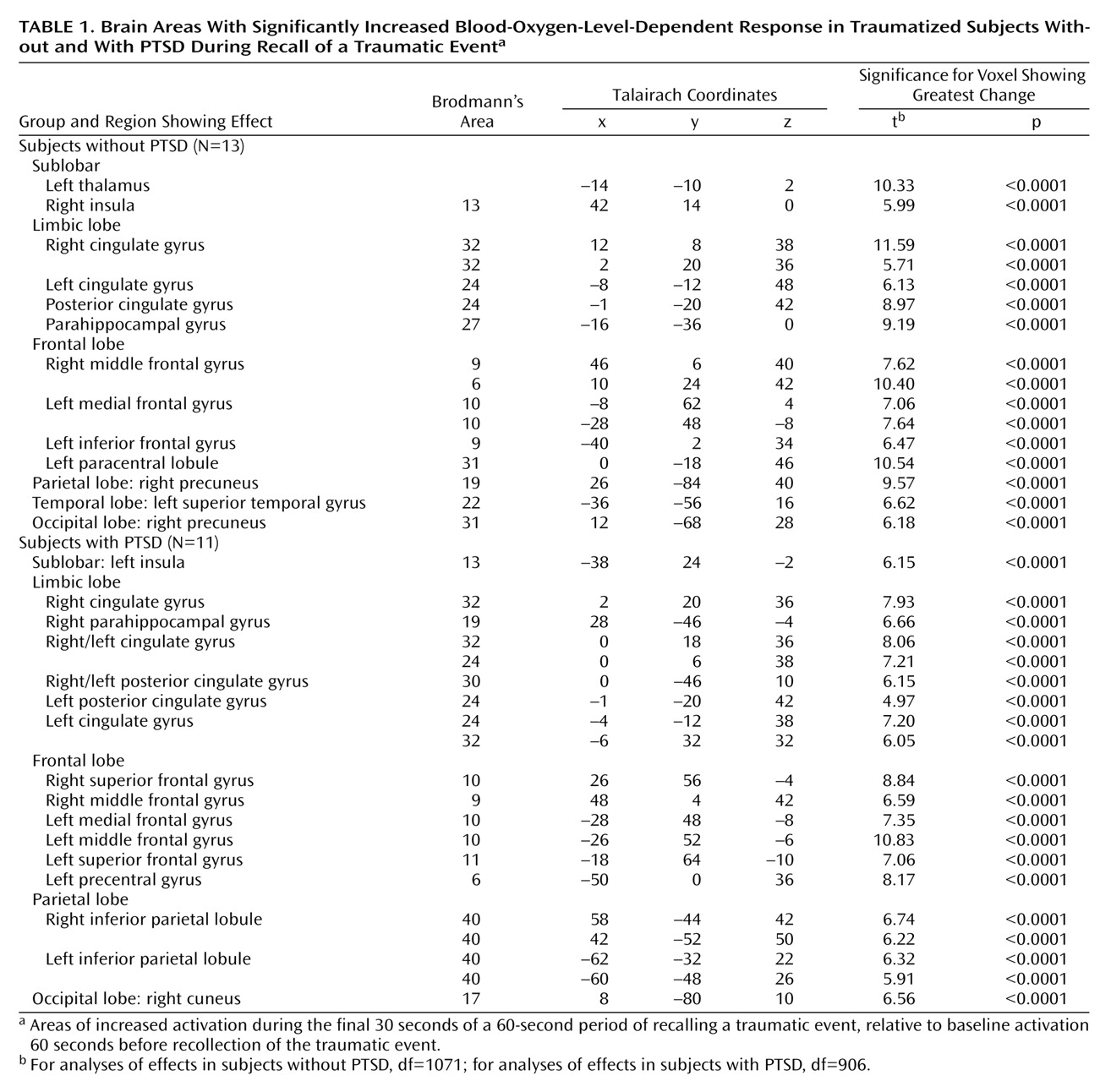
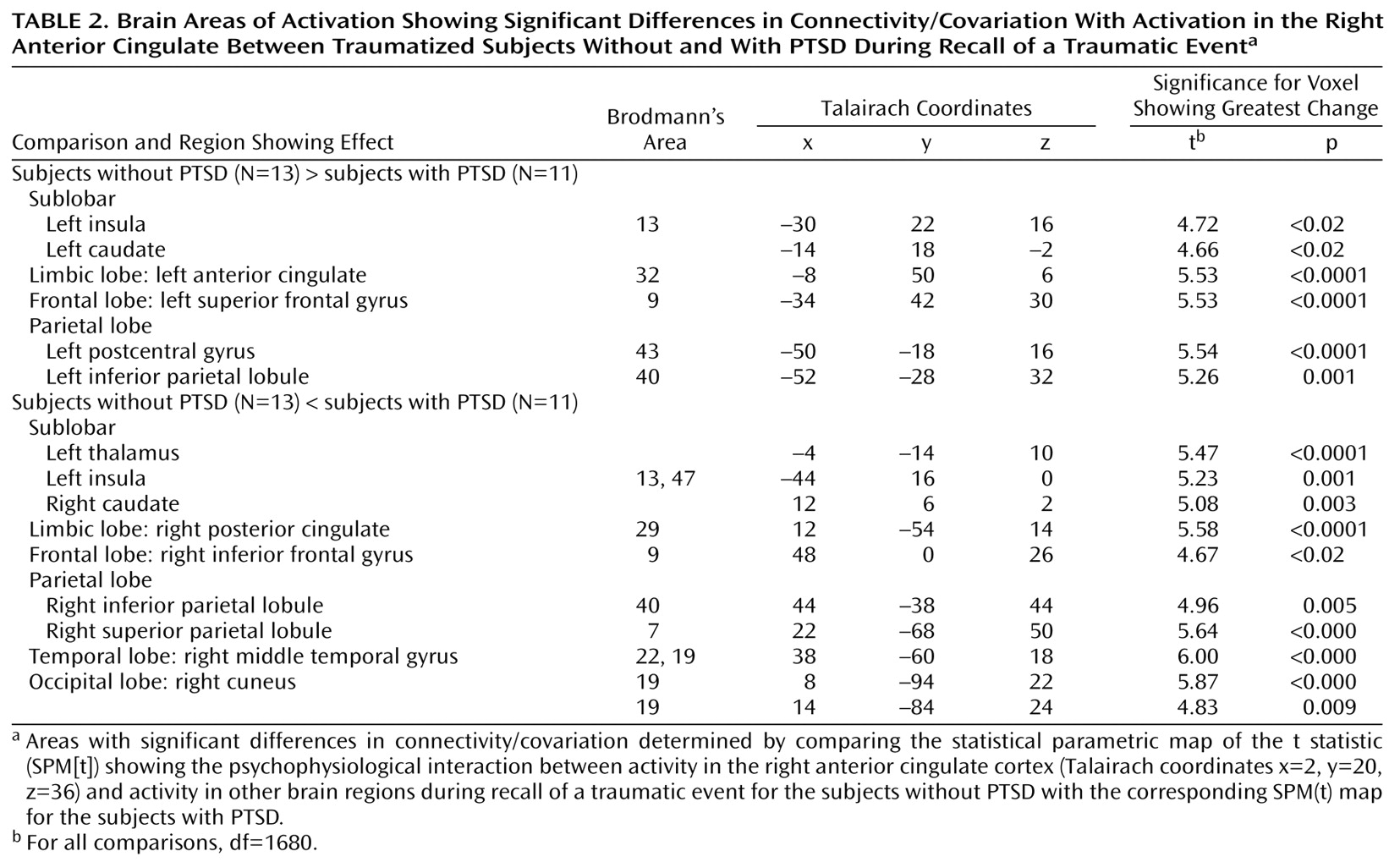
Previous neuroimaging studies of patients with posttraumatic stress disorder (PTSD) have examined specific brain regions involved in the recall of traumatic events and have implicated limbic, paralimbic, and prefrontal structures in the pathophysiology of PTSD
(1–
10). Paralimbic structures that are thought to underlie PTSD include the anterior cingulate gyrus (Brodmann’s areas 24 and 32) and the orbitofrontal cortex. Prefrontal structures implicated in PTSD include the left inferior prefrontal cortex and Broca’s area
(2–
6,
11). In support of these findings, our group, by using functional magnetic resonance imaging (fMRI), showed that PTSD patients who had a hyperarousal response to traumatic transcript-driven imagery had significantly less activation in the anterior cingulate gyrus (Brodmann’s area 32), medial prefrontal cortex (Brodmann’s area 11), and thalamus, as compared to traumatized subjects who never met the criteria for PTSD
(7).
Discussion
The comparison of functional connectivity maps showed distinctly different connectivity patterns in the PTSD subjects and in the comparison subjects. The most striking findings arose from the connectivity analyses originating from activation in the cognitive division of the right anterior cingulate gyrus (Brodmann’s area 32) (coordinates x=2, y=20, z=36). The affective division of the anterior cingulate gyrus has been shown to be involved during the recall of traumatic material in PTSD
(2–
4,
7), and reciprocal connections between the cognitive and affective divisions of the anterior cingulate gyrus have been described
(22). In addition, the affective division of the anterior cingulate gyrus plays a role in the conscious experience of emotion and in linking autonomic changes to emotional stimuli
(22,
23). PTSD subjects did not show similar patterns of alterations in brain activation during recall of a neutral autobiographical memory. It is therefore unlikely that the between-group differences observed in response to the traumatic memories are generalizable to any autobiographical memory.
The differences in functional connectivity patterns observed between the PTSD subjects and the comparison subjects may be related to variations in episodic memory retrieval. All PTSD patients experienced the traumatic memories in the form of flashbacks, whereas the comparison subjects recalled the traumatic events as ordinary autobiographical memories.
Reexperiencing of traumatic events in the form of flashbacks is very different from the recall of events as ordinary autobiographical memories
(24,
25). Flashbacks often occur spontaneously and are triggered by internal or external events, and their occurrence usually cannot be controlled. Flashbacks also involve a subjective distortion in time. They are much more vivid in nature and are often experienced as though the event were happening again in the present. Flashbacks have also been shown to be experienced as fragments of the sensory components of the event, such as visual images or olfactory, auditory, or kinesthetic sensations
(25). They have also been reported to be unchanging over time
(26), compared with ordinary memories, which are altered by repeated recall
(27). Overall, flashbacks are therefore highly perceptual memories that are primarily image-based. In contrast, in ordinary autobiographical memory, the sensory elements of the experience are automatically integrated into a personal narrative
(26).
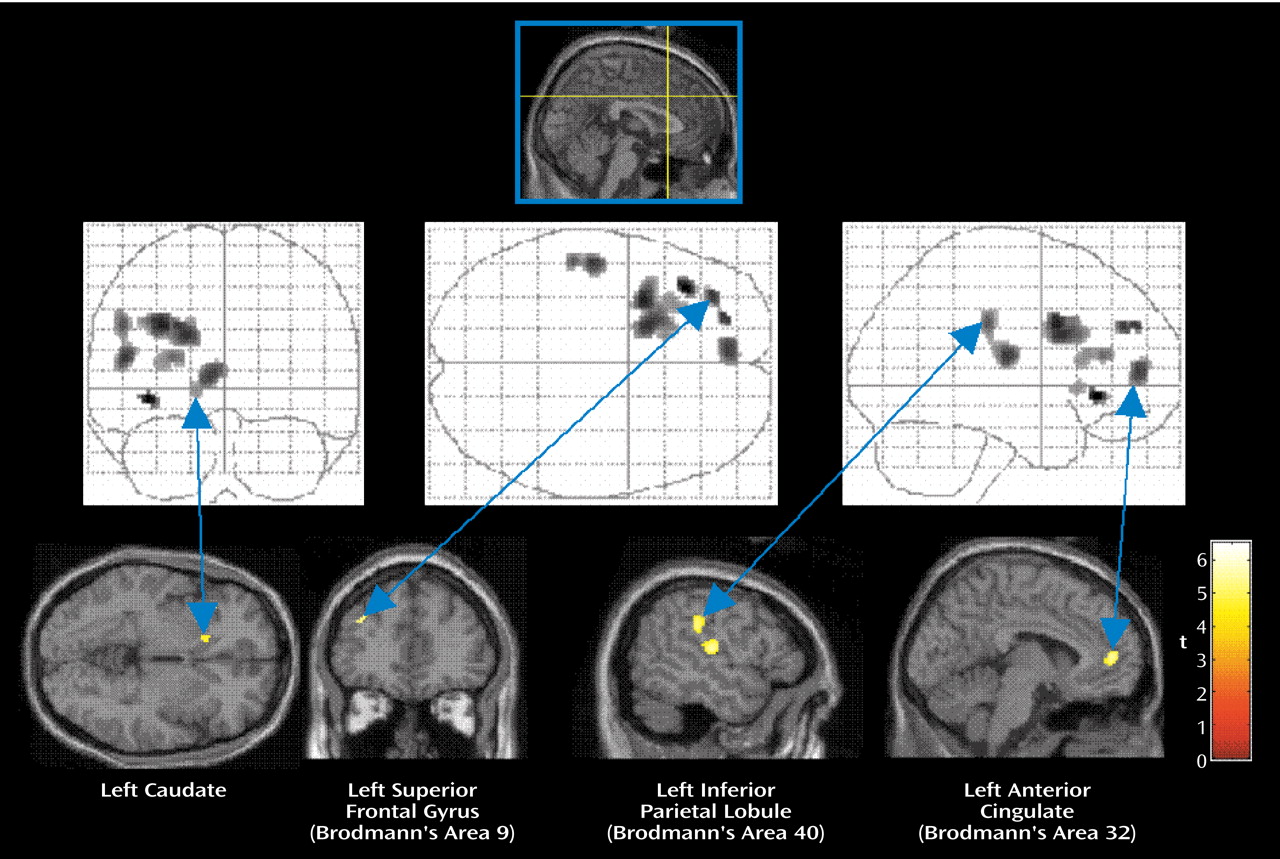
It is interesting to note that the functional connectivity analyses for the comparison subjects showed areas of brain activation that are consistent with verbal episodic memory retrieval. Comparison of the functional connectivity maps of the PTSD subjects and the comparison subjects showed that the comparison subjects had greater levels of brain activation in the left superior frontal gyrus (Brodmann’s area 9), left anterior cingulate gyrus (Brodmann’s area 32), left striatum (caudate), left parietal lobe (Brodmann’s areas 40 and 43), and left insula (Brodmann’s area 13). Prefrontal activations during episodic memory retrieval have sometimes been shown to be bilateral, but overall to have a clear tendency for right lateralization
(28). Anterior cingulate activation during memory recall has been hypothesized to be related to language processes, since activation of this area is more frequent for verbal than nonverbal materials.
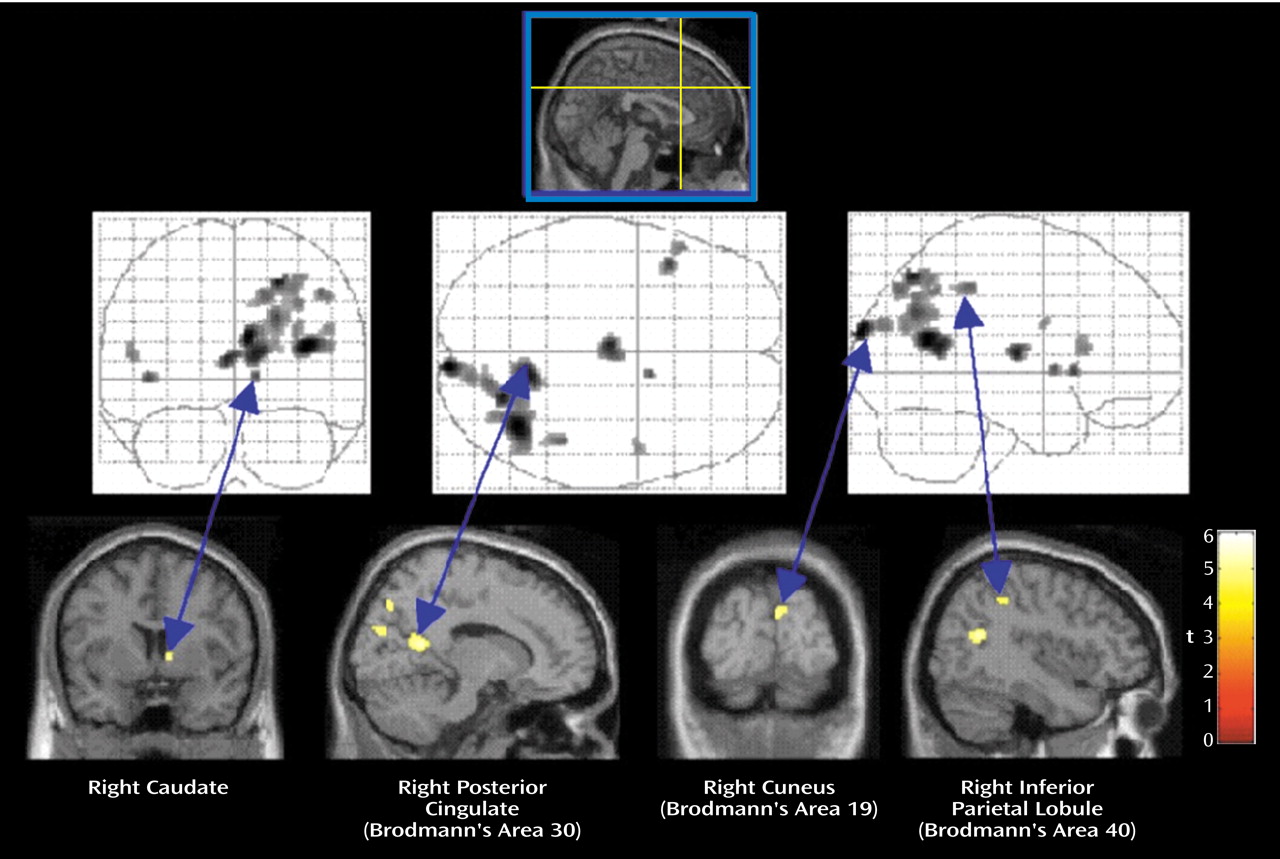
The functional connectivity analyses for PTSD patients revealed a much more nonverbal pattern of memory retrieval. Comparison of the functional connectivity maps of the PTSD subjects and the comparison subjects showed that the PTSD subjects had greater levels of brain activation in the right posterior cingulate gyrus (Brodmann’s area 29), right caudate, right parietal lobe (Brodmann’s areas 7 and 40), and right occipital lobe (Brodmann’s area 19). For nonverbal episodic retrieval, occipital, right parietal, and posterior cingulate activations have been shown to predominate
(28,
29). Moreover, the posterior cingulate gyrus has recently been shown to mediate interactions of emotional and memory-related processes
(30). Functional connectivity analyses may therefore help to elucidate the differences in the nature of memory recall (verbal versus nonverbal) between comparison subjects and PTSD patients. It is interesting to note that Paivio
(31) has proposed a dual coding theory in which affective and emotional reactions become associated primarily with a nonverbal representational system because they are learned in the context of nonverbal events.
Functional neuroimaging techniques (PET and fMRI) have been used to investigate the neuronal circuitry underlying autobiographical memory. In a review of 11 neuroimaging studies of autobiographical memory, Maguire
(32) noted that the most consistent result seen is a medial and left-lateralized activation pattern
(33,
34). In one study
(35), however, subjects were instructed to recall particularly affect-laden memories, resulting in a predominantly right hemispheric activation pattern that included the temporal cortex, posterior cingulate, insula, and prefrontal cortex. Our results add an interesting dimension to this literature, as they suggest that the subjects without PTSD do not process their “traumatic” memories as affect-laden memories, whereas PTSD subjects show the right-lateralized pattern of activation consistent with the results of Fink et al.
(33).
The functional connectivity analyses also found differences in the brain regions whose activity covaried with that of the left posterior cingulate gyrus. Of particular interest was the finding that activity in the right cingulate gyrus was lower in the PTSD subjects than in the comparison subjects, while activity in the right anterior cingulate gyrus was higher in the PTSD subjects than in the comparison subjects. There are reciprocal connections between the rostral cingulate and both the dorsal anterior and subgenual cingulate gyrus
(36). Mayberg et al.
(37,
38) found that pretreatment metabolism in the rostral anterior cingulate gyrus uniquely differentiated treatment responders from nonresponders in a group of patients with unipolar depression. Furthermore, activity in both the left posterior cingulate (Brodmann’s area 31) and right dorsal anterior cingulate (Brodmann’s area 24b) increased after 6 weeks of treatment with fluoxetine in the treatment responders. In studies of patients with obsessive-compulsive disorder (OCD), Rauch et al. found that high pretreatment activation levels in the posterior cingulate cortex predicted treatment response to fluvoxamine
(39) and to anterior cingulotomy
(40). These predictive findings may be relevant to our study for two reasons. First, there is a high degree of comorbidity in patients with PTSD, who often experience depression and/or other anxiety disorders, raising the question of whether similar predictive patterns may be seen in PTSD patients treated with these agents. Second, given these similarities in activation pattern between the anxiety disorders and depression, follow-up studies will be necessary to examine whether consistent differences in activation patterns might serve to differentiate between the two groups of patients.
Functional connectivity analyses also suggested differences in laterality, with PTSD subjects showing a right hemisphere dominance effect. The importance of lateralized responses in PTSD has been previously examined by using EEG and auditory probe evoked potential attenuation. Schiffer et al.
(41), using auditory probe evoked potential attenuation as a measure of hemispheric activity, reported that subjects who had experienced early trauma displayed significant left-dominant asymmetry during neutral memory recall and relative right dominance during traumatic memory recall. In addition, psychological abuse has been shown to be associated with an increased prevalence of left-sided EEG abnormalities and an increased prevalence of right-left hemisphere asymmetries
(42). EEG coherence studies also reported that abused children had greater average left hemisphere coherence than did nonabused children but that the two groups had a comparable degree of right hemisphere coherence. Teicher et al.
(43) suggested that these findings may be related to diminished left hemisphere differentiation in the abused group and thus may provide evidence that childhood abuse has a significant effect on cortical development.
Previous neuroimaging studies in PTSD using the script-driven imagery symptom provocation paradigm have also suggested lateralized responses. Using traumatic script-driven imagery, Rauch et al.
(8) found that in the traumatic condition, relative to the neutral condition, regional cerebral blood flow increases occurred in the
right medial orbitofrontal cortex, insula, amygdala, and anterior temporal pole. However, lateralized responses have been reported in other disorders besides PTSD. Pooling data from patients with three different anxiety disorders, including OCD, simple phobia, and PTSD in a study that used symptom provocation paradigms, Rauch et al.
(44) found that the right inferior and medial orbital prefrontal cortex as well as the bilateral insular cortex, lenticulate nuclei, and brainstem foci were consistently activated across the three different groups. These provocation studies indicate the consistent involvement of both paralimbic and subcortical regions in the mediation of acute anxiety, independent of stimulus type and patient group. These results illustrate that the specific mechanisms underlying the different anxiety disorders remain unclear and that studies are needed to address functional connectivity/neuronal networks rather than solely examining specific brain regions involved in the acute anxiety experience. The importance of examining neuronal networks in addition to regionally specific changes has also recently been described by Shaw et al.
(45), who used covariance-based multivariate analysis to investigate distributed brain systems in PTSD patients and matched comparison subjects during performance of a working memory task. However, it is not possible to directly compare our findings to those of Shaw et al. because of the different nature of the memory systems (autobiographical memory versus working memory) investigated in the two studies.
The present study had several limitations that must be considered in interpreting the findings. The relatively small number of study participants (N=11 in the PTSD group and N=13 in the comparison group) was not large enough for a random-effects analysis, which would have allowed the extrapolation of the results from this study to a larger population. Studies using a larger number of subjects are currently in progress. In addition, the comorbidity profile of the PTSD subjects may be a confounding factor in the study. Some PTSD subjects included in the present study had significant comorbidity, including dysthymic disorder, panic disorder, lifetime history of polysubstance dependence, and major depressive disorder. Furthermore, the current study included subjects who had experienced a mixture of traumata (sexual/physical abuse and motor vehicle accidents). Future studies will need to address possible pretrauma differences in imagery and verbal thinking habits between PTSD subjects and comparison subjects.
In summary, the present functional connectivity findings illustrate significant differences in brain connectivity between PTSD and comparison subjects during the recall of traumatic events. These variations may account for the differences in the nature (i.e., nonverbal versus verbal) of traumatic memory recall between PTSD subjects and comparison subjects. Moreover, the connectivity analyses used in this study may also allow a better understanding of the functional connectivity underlying various anxiety disorders and depression. Many of the same patterns of activation are seen in paralimbic and subcortical structures in both types of disorders, and there is a high degree of comorbidity between the two types. Functional connectivity analyses may help to elucidate the differences in the dynamic neuronal networks involved in these disorders.