Measures
Both in the initial questionnaire mailed to the twins and in the third wave of interviews, we asked the twins four questions about the psychological aspects of their premenstrual experiences. In this paper we will refer to these two assessments of premenstrual symptoms as “time 1” and “time 2.” These four items, which were similar to those used in other general population surveys
(10), were as follows:
A lot of women experience changes in their health and mood BEFORE they have their periods. Just BEFORE the start of your period, do you:
a) have less energy than usual or get tired more easily?
b) feel more sad, blue, or depressed?
c) feel more irritable or get upset more easily?
d) have any other changes in health or mood?
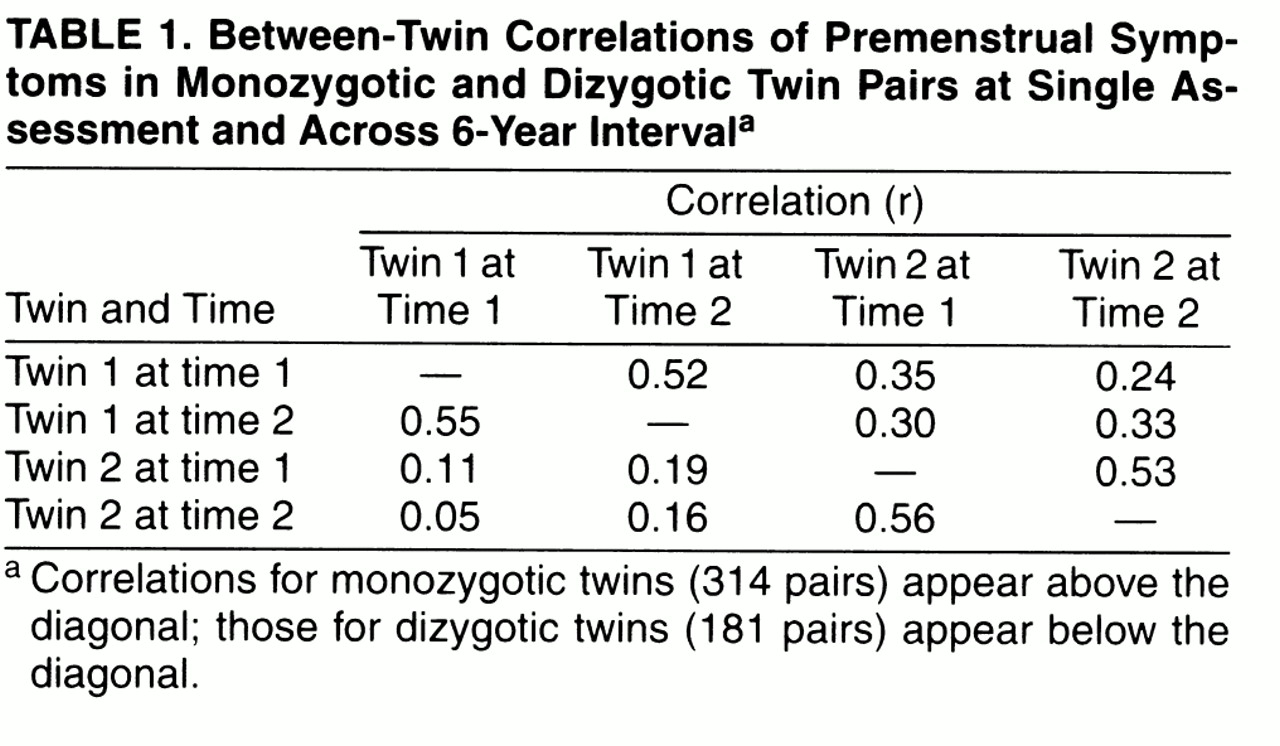
We called these items, respectively, “TIRED,” “SAD,” “IRRITABLE,” and “OTHER.” Four possible responses were provided: “A Lot,” “Some,” “A Little,” and “No.” Twins who reported that their menstrual symptoms had stopped completely (9.8% of the time 1 sample and 13.0% of the time 2 sample) were excluded from these analyses. Women who were pregnant, however, were asked to respond for their typical premenstrual experiences. At time 1 and time 2, 28.3% and 27.6% of the twins, respectively, reported currently using oral contraceptives. Individuals were included in this sample if they answered at least three of the four premenstrual items at both times of assessment. For those missing a single item at time 1 or time 2 (N=8 and N=4, respectively), the score on the missing item was imputed from theanswered items.
Diagnoses of lifetime major depression were based on DSM-III-R criteria, as determined with an adaptation of the Structured Clinical Interview for DSM-III-R
(30) conducted by carefully trained interviewers with prior professional mental health experience; this assessment was made both during the initial face-to-face interview and during the third telephone interview. The interviewers were blind to the psychiatric history of the co-twin. For these analyses, individuals assessed at the third interview who reported an onset of major depression after the first interview were considered to be unaffected, so that we were obtaining two reports on each subject’s lifetime history for major depression before the first interview.
Statistical Analysis
We performed a principal components analysis factor analysis of the four premenstrual symptom items at both waves by using the PROCFACTOR procedure in SAS
(31). The number of factors was determined by an eigenvalue criterion. The twin-measurement model used in this investigation has been described previously
(32). Briefly, we assume that the variation in premenstrual symptoms can be ascribed to three sets of factors: 1) additive genetic factors (A), which contribute twice as much to the correlation in monozygotic twins as dizygotic twins (because monozygotic twins share all their genes identical by descent, while dizygotic twins share on average one-half of their genes); 2) family or “common” environment (the familial factors, such as parental attitudes, that are shared by members of a twin pair) (C), which contributes equally to the correlation in monozygotic and dizygotic twins; and 3) individual specific environment (E), which traditionally reflects environmental experiences not shared by both members of a twin pair and therefore contributes to differences between them in their reported levels of premenstrual symptoms. (We also explored the impact of adding dominance genetic variance in model fitting but do not report these results here as they did not result in improvements in overall fit.)
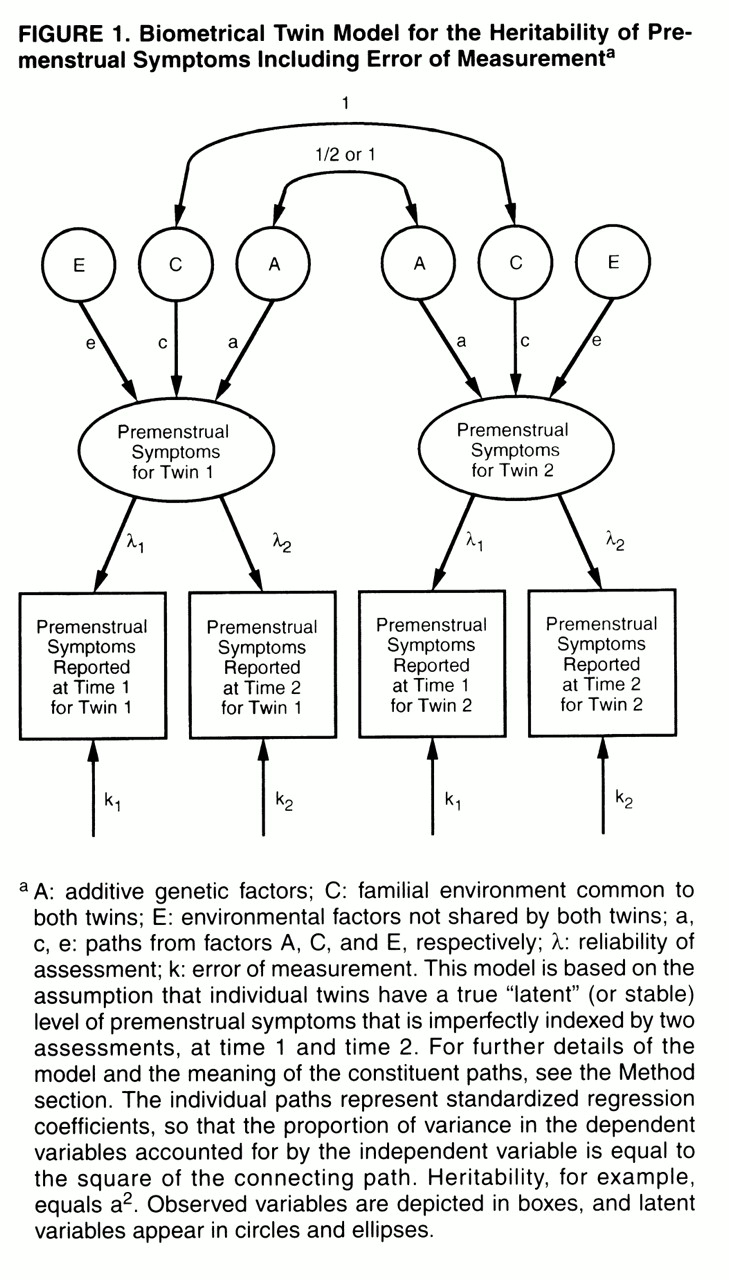
As outlined in
figure 1, the twin-measurement model uses the premenstrual symptom scores of twins at both time 1 and time 2. The model assumes that each twin has a true but unobserved (or “latent”) level of premenstrual symptoms. We measured premenstrual symptoms twice in these twins, but each measurement is fallible in that it partially reflects the true level of premenstrual symptoms and partly reflects short-term temporal fluctuations or error. The paths λ
1 and λ
2 represent the degree to which the assessments of premenstrual symptoms obtained at time 1 and time 2, respectively, reflect this true level of symptoms. The higher the value of λ, the more accurately any one measure of premenstrual symptoms reflects anindividual’s true level of symptoms. The other path to assessments of premenstrual symptoms at the two time points (k
1 and k
2, respectively) represents error in the individual assessments. By definition, λ
2+k
2=1.0. These models assume that the errors of measurement are uncorrelated within time. The true, or latent, level of premenstrual symptoms is then modeled in a standard twin design, as already outlined, with the sources of variance in liability divided between additive genetic, common environmental, and individual specific environmental factors.
Three differences between a measurement model and the standard twin model, based on a single time of assessment, are noteworthy. First, this model provides separate estimates for error of measurement (k) and true individual specific environment (e), which are confounded in the standard twin model. Second, it provides a direct estimate of the reliability of the phenotypic assessment support (λ). Third, while a standard twin model estimates the heritability of the observed phenotype (including error), the measurement model estimates the heritability of the latent phenotype, correcting for the effects of error. The latter will always be greater than the former.
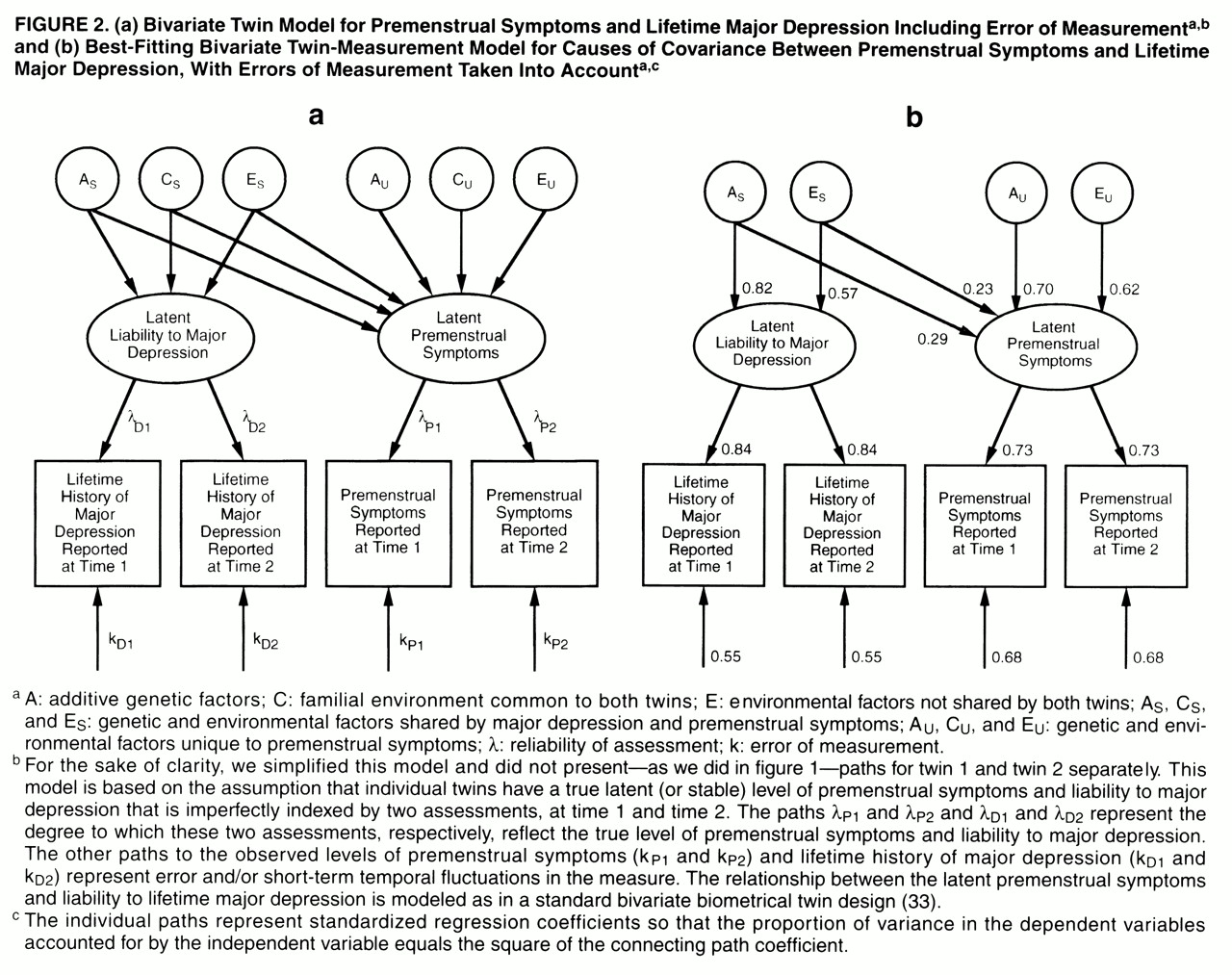
We present here, for the first time to our knowledge, a bivariate measurement model that combines features of the measurement model with those of a standard bivariate twin model (
figure 2a)
(33,
34). Basically, in a bivariate model we try to decompose the
covariation between two disorders into its genetic and environmental components. However, in the bivariate measurement model, instead of decomposing covariation between
observed disorders, we explore the etiologic relationship between two latent disorders, which are each, in turn, assessed at two occasions of measurement. Thus, this model permits us to assess the genetic and environmental covariance between two disorders while correcting for the effects of measurement error. As outlined in
figure 2a, we use a Cholesky decomposition
(34) that includes two “sets” of genetic and environmental factors—those that are shared by major depression and premenstrual symptoms (A
S, C
S, and E
S) and those that are unique to premenstrual symptoms (A
U, C
U, and E
U). Lower-case letters (a, c, and e) are used to label the paths from these factors.
These analyses are based on the assumption that the exposure to environmental factors that influence premenstrual symptoms are similarly correlated in monozygotic and dizygotic twins. We tested these two assumptions by using regression analysis to examine whether, with controls for zygosity, measures of childhood or adult environmental similarity could predict the difference in premenstrual symptoms in members of a twin pair.
Modeling fitting was performed with the program Mx
(35) by using weighted least squares to fit to matrices of polychoric correlations computed by PRELIS 2.14
(36). Because the full-weight matrices were almost singular, we used only the diagonal of the weight matrix to fit the model. The best-fit model was selected by using Akaike’s information criterion (AIC)
(37).