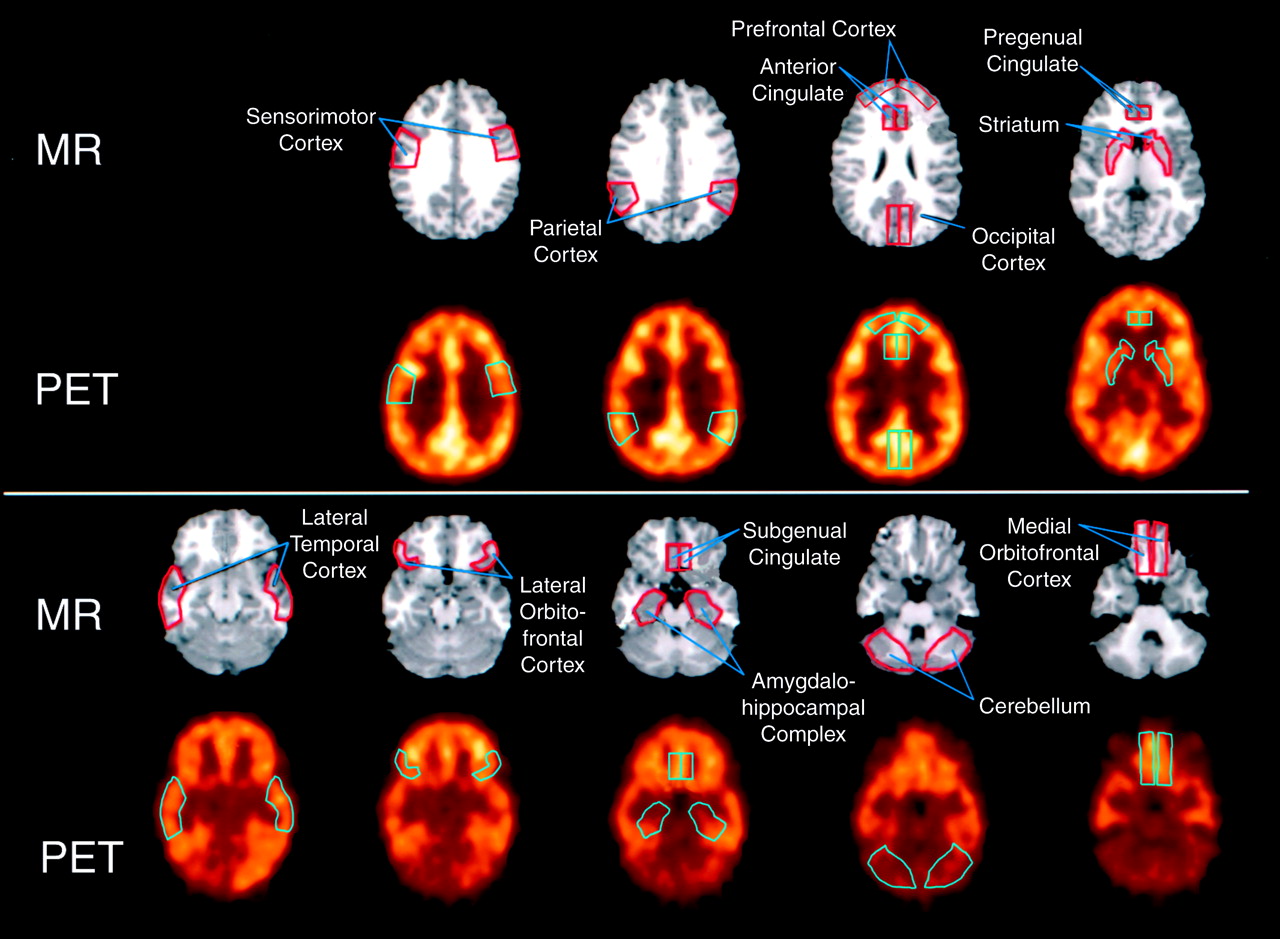
Dysregulation of the serotonin (5-HT) neurotransmitter system has been specifically implicated in depressive illness. In particular, considerable evidence indicates that the type 2A (5-HT
2A) receptor subtype has an important although poorly defined role in depressive illness and the mechanism of antidepressant medication
(3–
6). Selective degeneration of the 5-HT system, particularly the 5-HT
2A receptor, has also been linked to both mood and cognitive features of Alzheimer’s disease
(7–
10). In postmortem specimens, Bowen et al.
(11) found diminished 5-HT
2A receptor density in the brains of both elderly depressed and Alzheimer’s disease patients.
Substantial aging effects on the 5-HT system, and particularly the 5-HT
2A receptor, have been observed in both animals and human postmortem studies
(14–
18). We and others
(19,
20) have demonstrated a marked age-related decline in specific binding of [
18F]altanserin to 5-HT
2A receptors in humans in vivo. This finding supports the hypothesis that age-related changes in 5-HT function predispose the elderly to develop depression and suggests a continuum of serotonergic losses in normal aging, late-life depression, and Alzheimer’s disease. The purpose of this work was to test this hypothesis and thus to determine whether there are differences in 5-HT
2A binding among nondemented depressed elders, Alzheimer’s disease patients, and age-matched healthy subjects.
RESULTS
There were no significant gender (χ
2=0.75, df=2, n.s.) or age (F=2.91, df=2, 27, p=0.07) differences among the three subject groups, although the nondemented depressed patients tended to be younger than the normal or Alzheimer’s disease subjects (
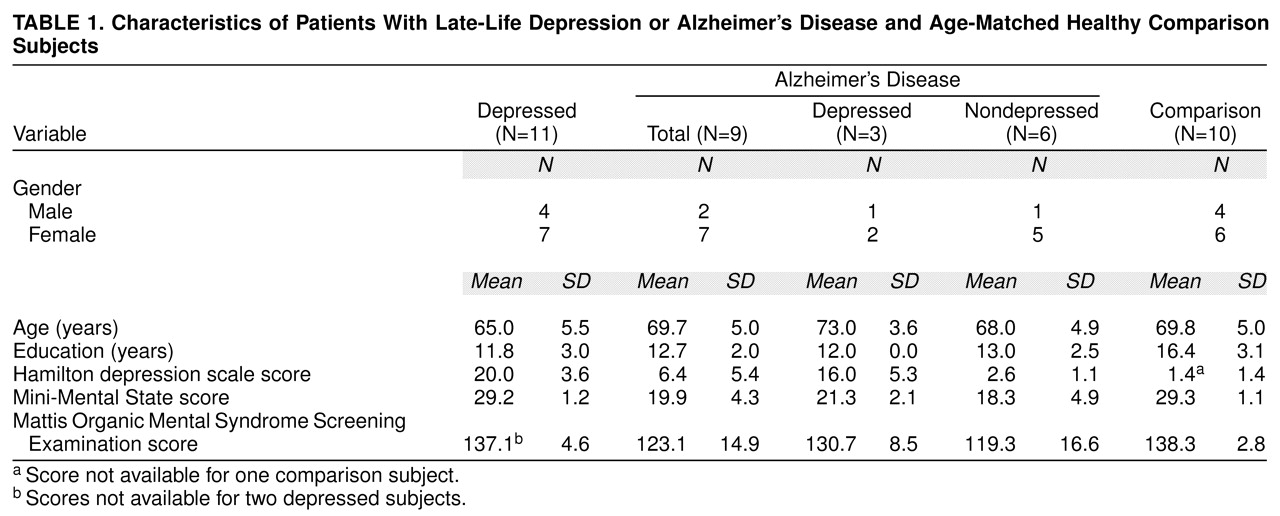
table 1). The comparison group was generally better educated than the patients (F=5.90, df=2, 27, p=0.007).
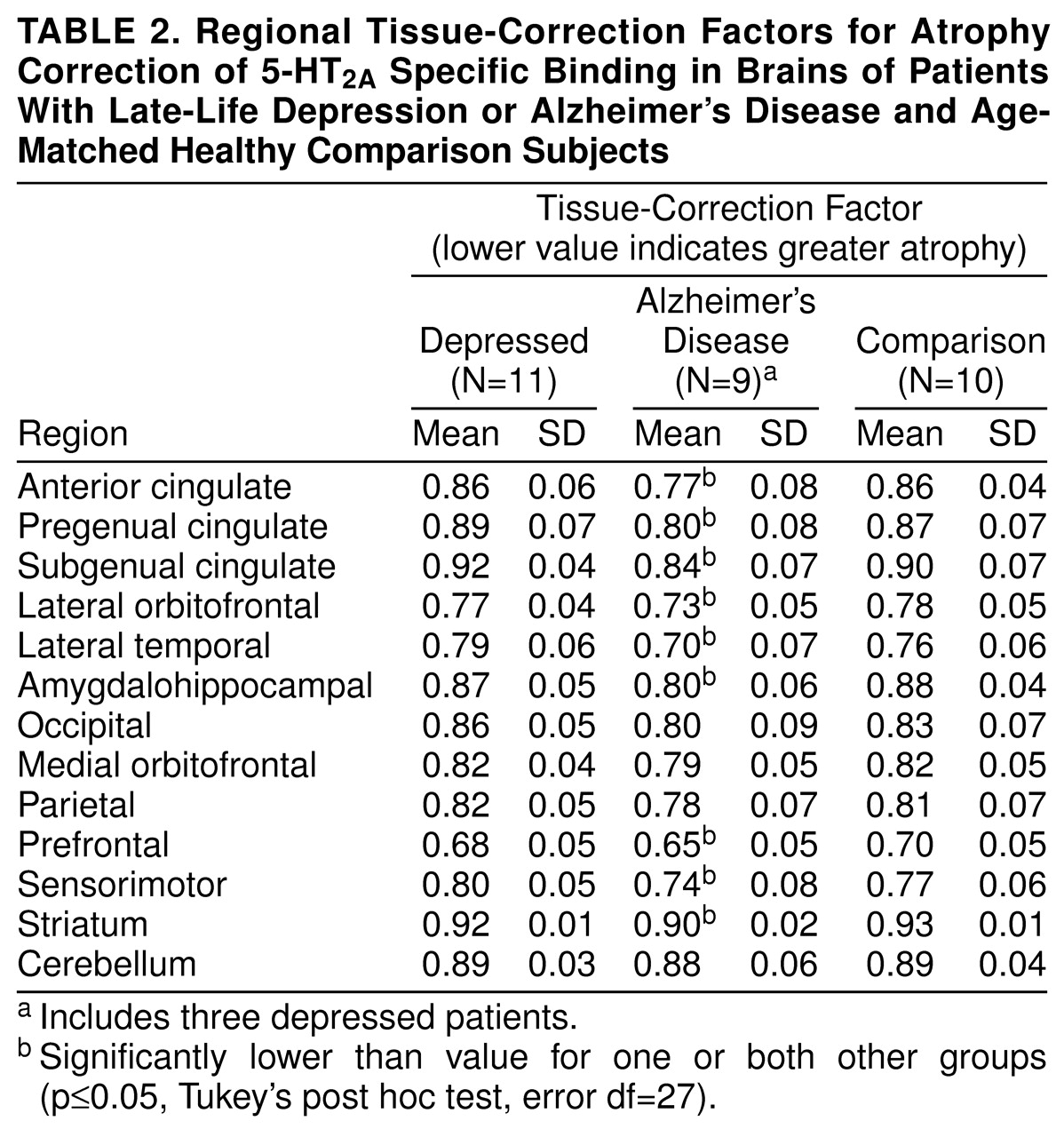
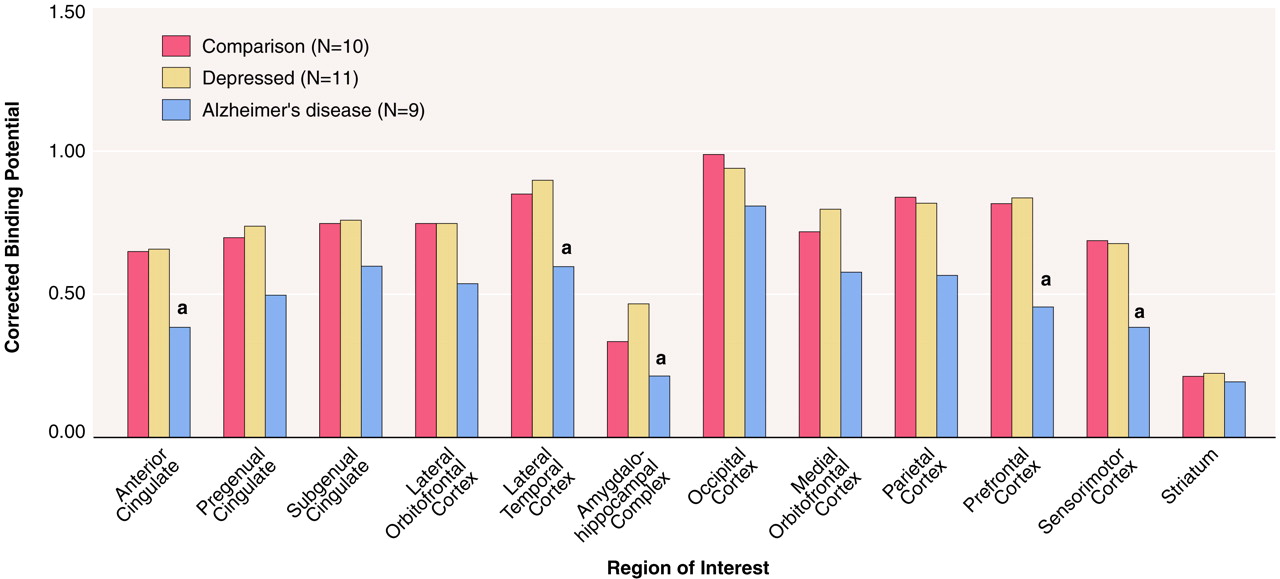
Using ANOVA followed by Tukey’s post hoc tests, we observed significant differences in the magnitude of the regional tissue-correction factors among the three groups. The Alzheimer’s disease subjects had significantly lower values (i.e., greater atrophy) than the comparison group and/or the nondemented depressed group in several brain regions (
table 2). These included the anterior cingulate (F=5.54, df=2, 27, p=0.01), pregenual cingulate (F=5.78, df=2, 27, p=0.01), subgenual cingulate (F=5.06, df=2, 27, p=0.02), striatum (F=7.89, df=2, 27, p=0.002), lateral orbitofrontal cortex (F=5.46, df=2, 27, p=0.01), lateral temporal cortex (F=6.42, df=2, 27, p=0.005), amygdalohippocampal complex (F=7.46, df=2, 27, p=0.003), prefrontal cortex (F=3.96, df=2, 27, p=0.03), and sensorimotor cortex (F=4.14, df=2, 27, p=0.03). The tissue-correction factors did not significantly differ between the nondemented depressed subjects and the comparison group in any region.
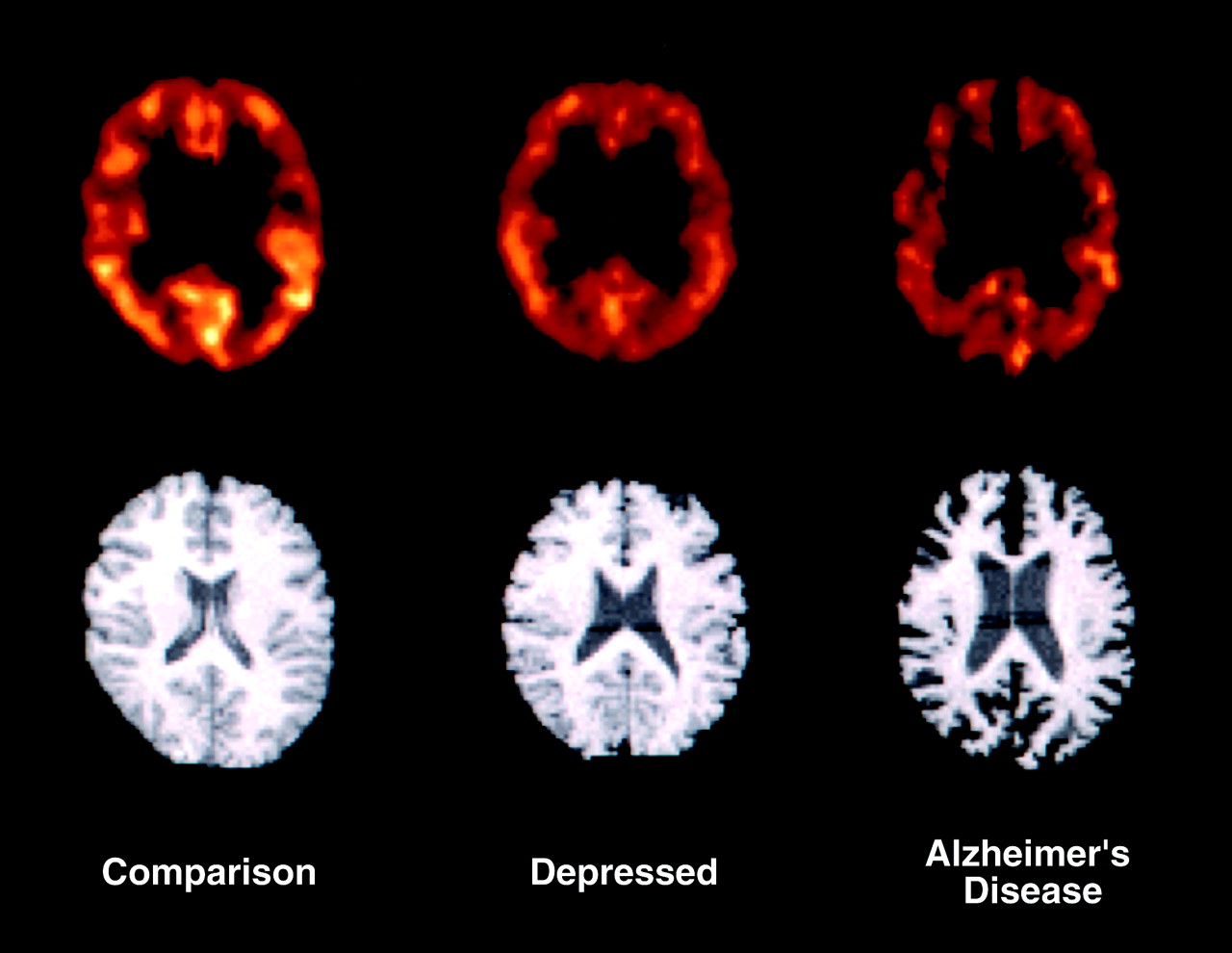
Overall, regional specific binding followed the known rank order of 5-HT
2A receptor density
(30). After an initial ANOVA of the data revealed no group-by-hemisphere interactions (p>0.15), paired right and left regions of interest were averaged for subsequent analyses. There were no significant differences in corrected binding potential between the nondemented depressed patients and the normal comparison group in any brain region examined (
figure 2 and
figure 3). No effect of depression on binding potential was noted in the nondemented depressed subjects or within the Alzheimer’s disease group; therefore, the Alzheimer’s disease subjects with and without depression were pooled for further analyses. The patients with Alzheimer’s disease had significantly lower atrophy-corrected binding potential than both the nondemented depressed and comparison groups in several brain areas, according to ANOVA. These regions were the anterior cingulate (F=3.74, df=2, 25, p=0.04), prefrontal cortex (F=4.65, df=2, 25, p=0.02), and sensorimotor cortex (F=3.75, df=2, 25, p=0.04). In addition, the atrophy-corrected binding potential in the Alzheimer’s disease group was significantly lower than that of the nondemented depressed patients but not the comparison group in the lateral temporal cortex (F=3.35, df=2, 25, p=0.05) and amygdalohippocampal complex (F=4.15, df=2, 22, p=0.03).
The cerebellar distribution volumes, representative of the free radioligand concentration and nonspecific binding, did not differ significantly among the nondemented patients with late-life depression (mean=1.69, SD=0.54), patients with Alzheimer’s disease (mean=1.94, SD=0.53), and comparison subjects (mean=1.68, SD=0.50) (F=0.71, df=2, 27, p=0.50).
No effect of subject group was observed in the rate of metabolism of [
18F]altanserin in plasma (F=0.04, df=2, 26, p=0.76). At 2 minutes 20 seconds postinjection, similar mean fractions of the parent compound were measured in the plasma of the normal subjects (mean=96.0%, SD=2.2%), nondemented depressed patients (mean=96.1%, SD=2.0%), and patients with Alzheimer’s disease (mean=94.9%, SD=3.7%). At the end of the 90-minute scanning period, the average fraction of unchanged [
18F]altanserin was 42.7% (SD=6.2%) in the comparison group, 42.0% (SD=16.2%) in the depressed patients, and 45.3% (SD=10.8%) in the Alzheimer’s disease group. Although radiolabeled metabolites of [
18F]altanserin appear to cross the blood-brain barrier, these metabolites have been demonstrated to have no significant specific binding to the 5-HT
2A receptor in vivo and, therefore, contribute to free radioligand concentration and nonspecific binding only
(31–
34).
The correlations between regional binding potential for [18F]altanserin and Hamilton depression scale score or time to recovery in the depressed patients did not achieve statistical significance. Also, no statistically significant correlations between binding potential and Mini-Mental State scores for the Alzheimer’s disease subjects were observed.
DISCUSSION
The major finding of this work is the absence of a significant abnormality in central 5-HT
2A receptor binding of [
18F]altanserin in late-life depression, in contrast to significantly lower than normal binding in Alzheimer’s disease. This suggests that the 5-HT
2A receptor may not play a key role in depressive illness and differs from the finding of a specific regional [
18F]altanserin binding abnormality in midlife depression
(13). In work by Biver et al.
(13), statistical parametric mapping techniques were applied to PET images from eight midlife depressed patients and 22 healthy comparison subjects. These data showed significantly lower uptake of [
18F]altanserin in a brain region encompassing the right orbitofrontal cortex and anterior portion of the insula in the depressed patients; a similar trend was observed in the homologous region in the left hemisphere. An analogous brain region (lateral orbitofrontal cortex) was sampled in the current study by using a region-of-interest approach; no specific binding differences were found between the elderly nondemented depressed patients and the age-matched comparison group. The study of Biver et al.
(13) supports the theory that 5-HT
2A receptor activity is impaired in depressive illness; however, the potential influence of age-related factors on the PET measurements was not fully considered and thus may potentially confound their interpretation. First, the mean age of the patients was 10 years higher than that of the nondepressed comparison group. Given the steep age-related decline in 5-HT
2A binding that has been demonstrated with [
18F]altanserin PET imaging
(19,
20), it is possible that the age difference between the subject groups may, in part, have accounted for the apparent differences in depression-related binding. Second, although structural brain differences would be expected to be small in the midlife group studied, the finding of significant binding differences in a brain region adjacent to a major (Sylvian) fissure raises the issue of the potential dilutional influence of greater age-related cerebral volume loss in the depressed subjects than the (younger) comparison group. In the current study, the subjects in the comparison group were slightly older than the depressed patients overall, but the subjects’ ages were restricted to a total interval of 18 years (58–76 years of age), and the potential influence of age on the binding estimates was accounted for in the statistical analysis. Further, the effect of possible age-related differences in cerebral volume loss on the PET measurements was accounted for by correcting the data for partial-volume effects.
In addition to a small (but nonsignificant) age difference between subject groups, reports of accentuated brain atrophy in late-life depression
(41–
43) reinforced the need to correct the PET measurements for the potential dilutional effect of cerebral atrophy. However, the lack of a significant difference in regional tissue-correction factors between the nondemented depressed and comparison groups suggested that the abnormalities in cerebral volume in depression were small, and indeed, they did not alter the study results. Substantial atrophy did exist in several regions in the Alzheimer’s disease group. Therefore, atrophy correction was applied to all data to eliminate this source of bias in the PET measurements. A previous PET study
(12) indicating 5-HT
2A receptor abnormalities in Alzheimer’s disease examined moderately to severely demented patients but did not correct for the potential confounding effects of greater atrophy in the patients than in the normal elderly subjects.
In postmortem studies by Bowen and colleagues
(11), late-life depression was associated with low 5-HT
2A receptor binding in several cortical regions (temporal, frontal, parietal). Greater differences from normal were seen in the brains of patients with Alzheimer’s disease, although these may be in part due to the fact that the Alzheimer’s disease patients were older (mean age=82 years) than both the comparison group and the nondemented depressed patients. Also, the results of such postmortem studies are likely to be biased toward Alzheimer’s disease of end-stage severity. In the current study, the low number of subjects precluded a definitive examination of 5-HT
2A receptor status in patients with coincident depression and Alzheimer’s disease. However, the absence of an abnormality in [
18F]altanserin binding in the nondemented depressed group, coupled with the lack of evidence for an effect of depression within the Alzheimer’s disease group, suggests that the 5-HT
2A receptor is differentially affected in late-life depression and Alzheimer’s disease. Potential reasons for conflicting findings in late-life depression between the work of Bowen et al.
(11) and the present in vivo study include the inherent difficulty in postmortem studies of ensuring uniform selection of subjects with uncomplicated major depression and controlling for exposure to psychotropic medications. Although the possibility of a type 1 error due to limited sample size in the PET study must be considered, the group sizes were sufficiently large to detect an approximately 30% difference in regional binding measures in the lateral orbitofrontal cortex between the late-life depressed and comparison groups. This magnitude was consistent with our expected effect size based on postmortem studies of similar brain regions in suicide victims
(3,
45). Further, there was no evidence for trends in any of the regional binding measures suggesting a difference between the late-life depressed subjects and the comparison group (
Figure 3).
There is substantial evidence to support the influence of gender on the functional status of the 5-HT system
(46–
49). Particularly relevant to this study is the reported contribution of postmenopausal decreases in estrogen levels to alterations in circulating 5-HT and central 5-HT
2A receptor densities in women
(46,
49,
50). For these reasons, the influence of gender on [
18F]altanserin binding to 5-HT
2A receptors was considered. An effort was made to minimize the potential confounding impact of gender on the PET measurements by gender matching the subject groups (depressed patients: 36% male, Alzheimer’s disease patients: 22% male, comparison group: 40% male) and excluding female subjects receiving estrogen replacement therapy from study participation.
Although radiolabeled metabolites of [
18F]altanserin appear to cross the blood-brain barrier, these metabolites have been demonstrated to have no significant specific binding to the 5-HT
2A receptor in vivo and, therefore, contribute to free and nonspecifically bound radioactivity concentration only
(31,
34). These metabolites have been well characterized and do not appear to confound the interpretation of specific binding measures obtained by using the Logan graphical analysis
(31). We have also previously shown
(19) that age does not affect the rate of metabolism of [
18F]altanserin over the 90 minutes after injection.
Since binding potential may reflect changes in receptor density (B
max) and/or receptor affinity (K
D), abnormalities in either or both measures may contribute to the finding of a difference or, alternatively, a lack of difference in regional binding potential between study groups. However, most postmortem binding assays of 5-HT
2A receptor ligands in the brains of suicide victims
(3) and Alzheimer’s disease patients
(51,
52) have shown abnormal B
max values and normal K
D values. Therefore, it is likely that the low specific binding of [
18F]altanserin in several brain regions is the result of a corresponding loss of 5-HT
2A receptors, as similarly suggested by Blin et al.
(12), who conducted a PET study using [
18F]setoperone.
This study addressed our hypothesis—based on prior findings of much lower than normal [
18F]altanserin binding in older subjects
(19) and controversial postmortem and neuroendocrine evidence for 5-HT
2A receptor abnormalities in depressive illness
(3,
11,
53,
54)—that specific 5-HT
2A receptor binding may be even lower in elderly individuals who suffer from depression. Our data, however, indicate no difference in specific binding of [
18F]altanserin in selective brain regions between elderly depressed patients and elderly healthy individuals. This finding is consistent with those of several postmortem studies showing no effect of depression on 5-HT
2A receptor binding in younger age groups
(50,
54–
56). Significantly lower than normal 5-HT
2A binding in Alzheimer’s disease was observed and is consistent with results from postmortem and prior in vivo studies suggesting specific serotonergic degeneration in Alzheimer’s disease
(7,
8,
11). In contrast to the postmortem work by Bowen et al.
(11), who showed similar low levels of 5-HT
2A receptors in late-life depression and Alzheimer’s disease, this study suggests that the 5-HT
2A receptor may be differentially affected in the two disorders.