DISCUSSION
The main finding was that low concentrations of raclopride, iodobenzamide, or dopamine could (within minutes) displace some of the [
3H]clozapine or [
3H]quetiapine prebound to D
2 receptors (
Figure 3,
Figure 4,
Figure 5), in contrast to [
3H]haloperidol and other [
3H]antipsychotic drugs. This finding is relevant and applicable to clinical brain imaging as follows. The amount of [
11C]raclopride injected into human volunteers for determining D
2 occupancy by PET is between 15 and 100 nmol, with an average of 36 nmol (A. Wilson and S. Kapur, personal communication, and references
55–
59). This average dose, when rapidly distributed in 5 liters of blood, yields a peak molarity in plasma of the order of 7 nM, with a final free concentration in the plasma water of the order of 0.03–0.3 nM raclopride (see references
31,
53, and
54 for data on free molarities of antipsychotic drugs). This concentration range of raclopride may displace some of the clozapine bound to the D
2 receptors in a human subject’s striatum within 10 minutes, at which time the brain imaging measurements are started. As shown in
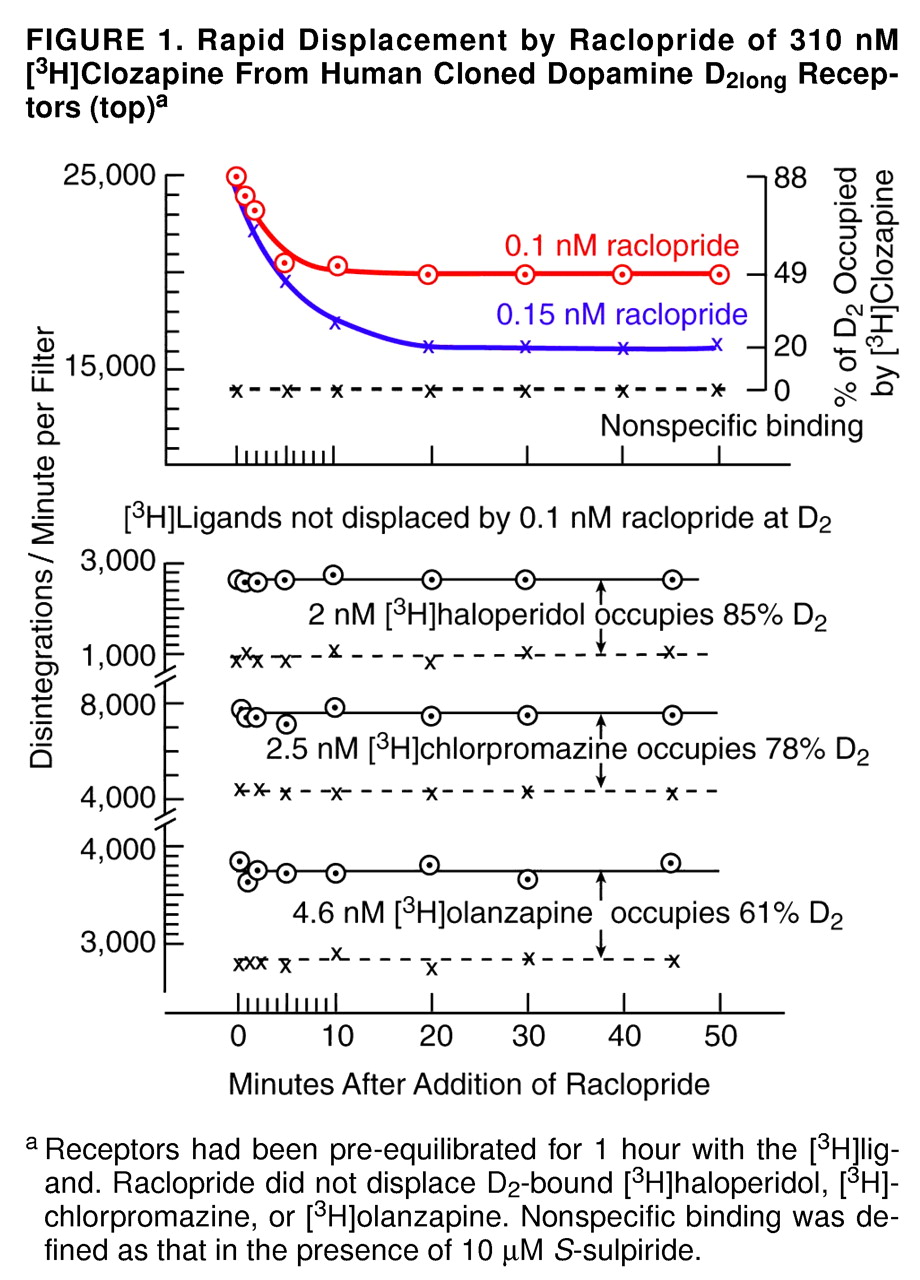
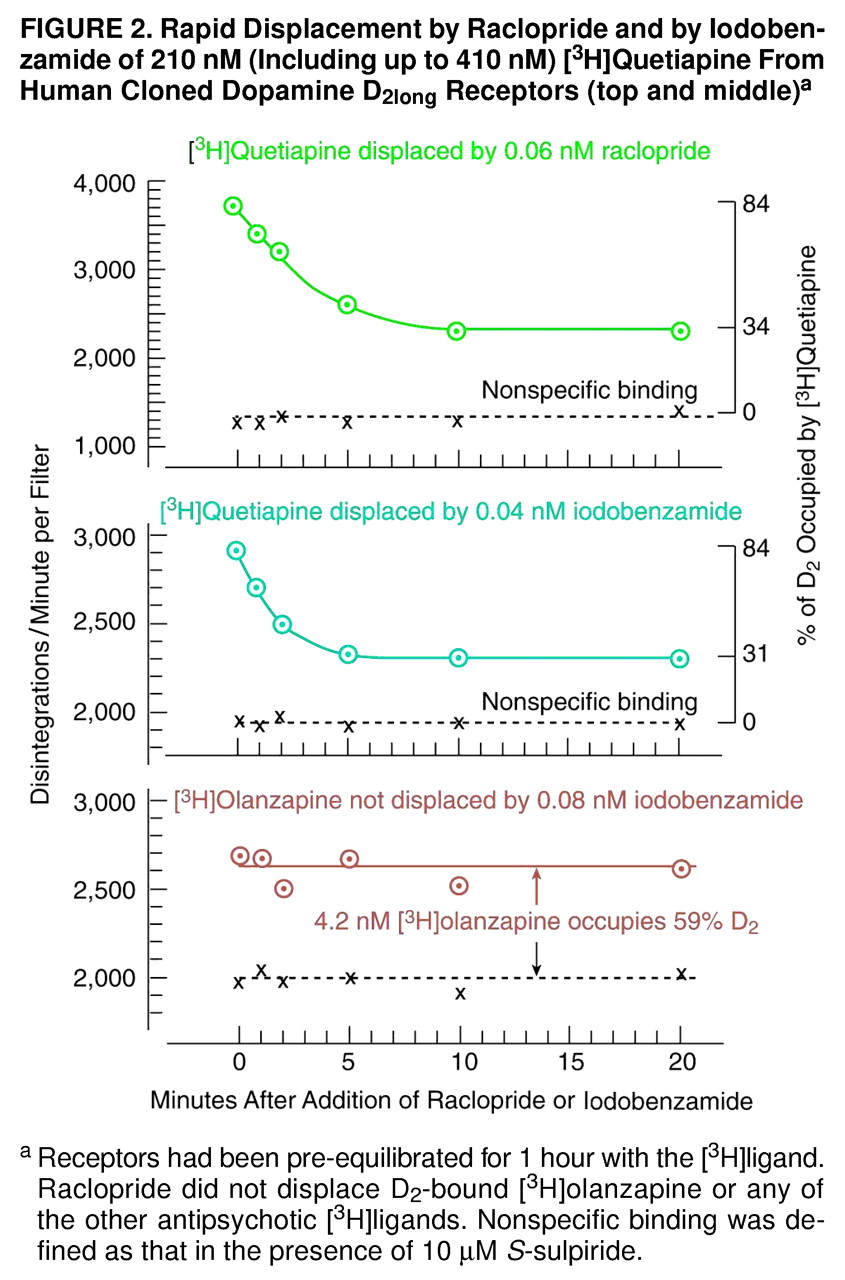
Figure 1 and
Figure 2, the effect of raclopride was long-lasting and persisted beyond 1 hour. (Under clinical conditions, of course, the patient subsequently resumes his or her medication [clozapine] after brain imaging, and any clozapine-displacing action of the injected raclopride dissipates as the raclopride is removed from the body.)
In particular, for example, the peak [
11C]raclopride in striatum corresponds to about 0.005% of the injected radioactivity per gram of striatum (60). Taking the concentration of [
11C]raclopride in the cerebellum (F*, according to the terminology of reference
29) as 0.0015% of the injected radioactivity per gram of striatum (at the time of peak striatal activity) as a rough measure of the “nonspecifically bound” tracer in striatum, one arrives at 0.0035% of the injected radioactivity per gram of striatum for the specifically bound [
11C]raclopride (B*, according to the terminology of reference
29). Hence, as obtained by Volkow et al.
(60), the ratio B*/F* is 2.3, in general agreement with the average value of 3.55 (SD=0.63) obtained by Farde et al.
(29) for 15 drug-naive patients. Furthermore, as defined previously
(29), receptor occupancy in a drug-treated patient is expressed as “the percent reduction of B*/F* from the value of 3.55.”
Hence, if 36 nmol of [
11C]raclopride are injected, the concentration in the striatum will be about 1 pmol/gram, in agreement with values reported earlier (61). If it is assumed that the concentration of the D
2 receptors in the human striatum is between 20 and 28 pmol/gram
(62–
64), the injected [
11C]raclopride will occupy between 3% and 5% of the total population of D
2 receptors. Thus, for example, an injection of 36 nmol of [
11C]raclopride might result in 1 pmol/gram, or 3%, of the total D
2 population being occupied in a drug-free subject. This control value of 1 pmol/gram, or 3% of total D
2, would be designated as “0% occupancy”
(29). For an individual taking a tightly bound antipsychotic drug such as haloperidol, however, the injection of the 36 nmol of [
11C]raclopride would result in less specific binding of the [
11C]raclopride, with an observed B*/F* value of, say, 1.1 instead of 3.55
(56). This fall in B*/F* would correspond to a D
2 occupation by [
11C]raclopride of 0.3 pmol/gram, or 1% of total D
2. Hence, the drop from 3% to 1% of total D
2 would indicate that haloperidol occupied about 67% of the D
2 population.
However, in the case of a patient taking clozapine (or quetiapine), the injection of 36 nmol of [11C]raclopride (with a specific activity of 500–1,000 Ci/mmol) would displace some of the D2-bound clozapine, resulting in a value for B*/F* that would be slightly higher than expected for the true occupancy by clozapine. For example, if clozapine tightly occupied 67% of the D2 receptors, then one would observe the same data that were just mentioned for haloperidol, namely, an occupation of 1% of total D2 by [11C]raclopride. However, if some of the D2-bound clozapine is displaced by [11C]raclopride, the occupation by [11C]raclopride will not be as low as 1% of the total density of D2 receptors. For example, if such a displacement results in 2% of the total D2 being occupied by [11C]raclopride (compared to the control value of 3% of the total D2 density), the apparent occupancy by clozapine would be seen to be only 33% instead of the expected 67%.
A similar situation obtains with [
123I]IBZM, used in SPECT, where between 1 and 2 nmol are injected. For example, European studies use [
123I]IBZM provided by Cygne B.V. (Eindhoven, The Netherlands)
(20–
22,
65), which prepares [
123I]IBZM by the method of Bobeldijk et al.
(66). This method uses carrier-free Na
123I and
S-benzamide, such that the final specific activity of
S-[
123I]IBZM is the same as that of the carrier-free Na
123I, namely, >3000 Ci/mmol. However, as later noted by Verhoeff et al.
(67), to enable the accurate measurement of the specific activity of [
123I]IBZM for both in vitro and in vivo studies, a small amount of carrier iodide is added to the labeling mixture, resulting in a specific activity of the prepared [
123I]IBZM in the range of 50–250 MBq/nmol. Thus, the Cygne B.V. specifications indicate that the [
123I]IBZM has 1 nmol/ml (0.4 µg/ml) of nonradioactive iodobenzamide. Usually, 185 MBq (or 5 mCi) are injected into the patient, and this dose corresponds to 1–2 nmol, depending on the time of injection. After being diluted in the bloodstream, and allowing for the fact that 4.4%–12.3% of the [
123I]IBZM is free in the plasma water
(68,
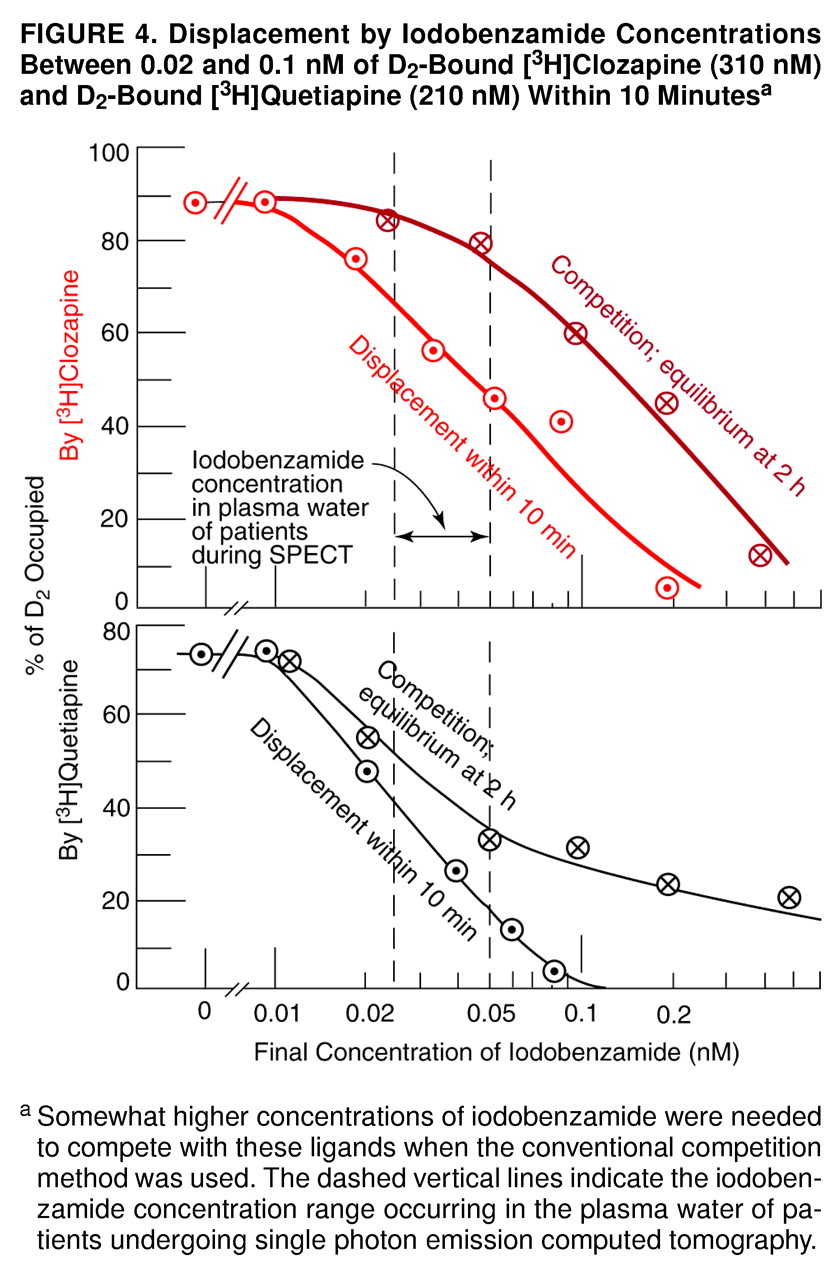
69), the final molarity of free iodobenzamide in the plasma water of a human volunteer is of the order of 0.02 nM–0.05 nM. This concentration range would be expected to displace some of the D
2-bound clozapine or quetiapine from the human volunteer’s striatum in a matter of minutes, as indicated in
Figure 4.
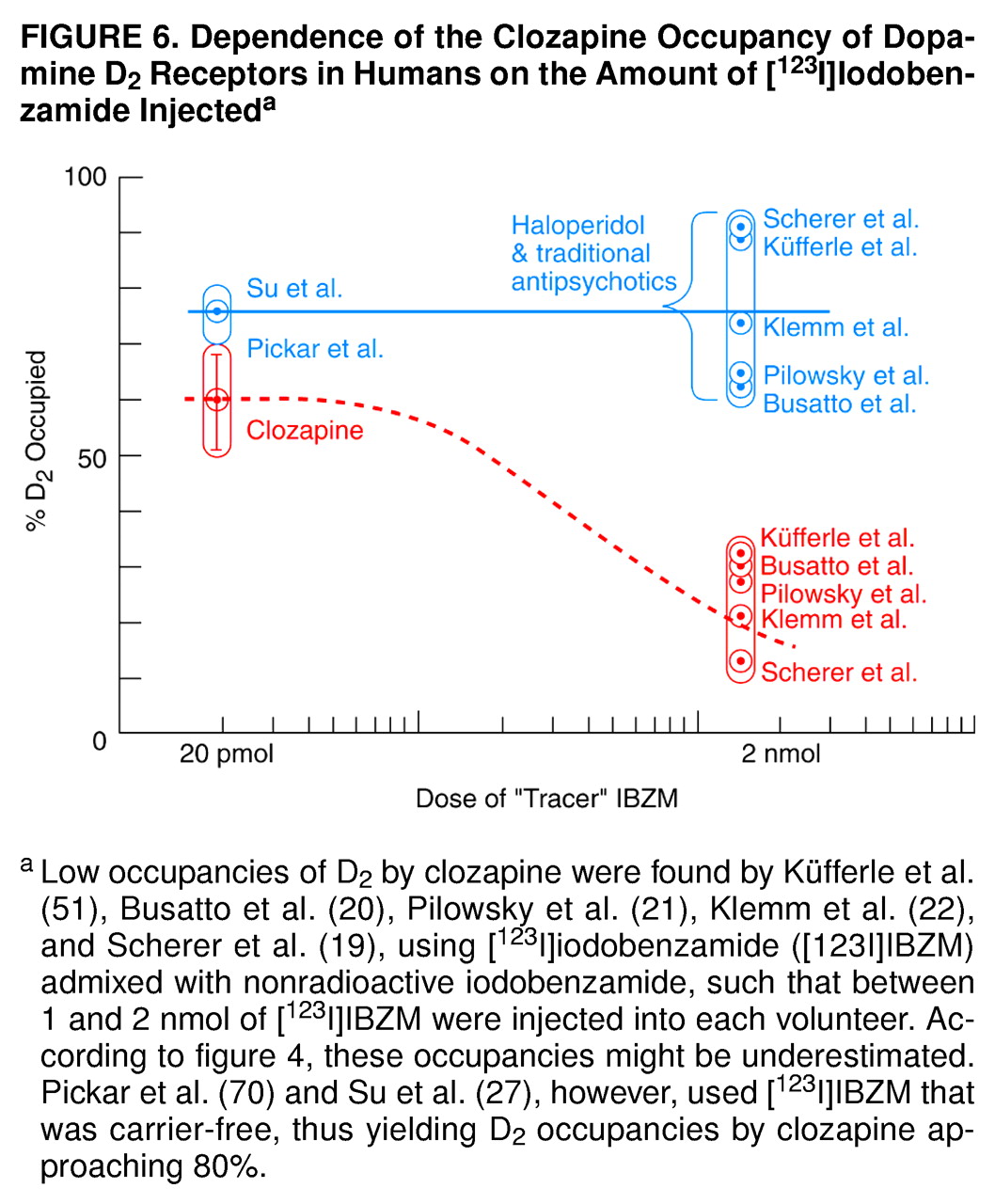
Thus, the data in
Figure 4 may account for the low occupancies of 12%–32% of D
2 by clozapine, as monitored by the [
123I]IBZM used in the European studies
(19–
22,
51). In other words, according to the data in
Figure 4, the D
2 occupancies by clozapine in these studies may be underestimated. In the United States, however, the [
123I]IBZM used by Pickar et al.
(70) was carrier-free, yielding D
2 occupancies by clozapine approaching 80%. Hence, the clozapine occupancy of D
2 depends on the amount of [
123I]IBZM injected, as illustrated in the summary in
Figure 6.
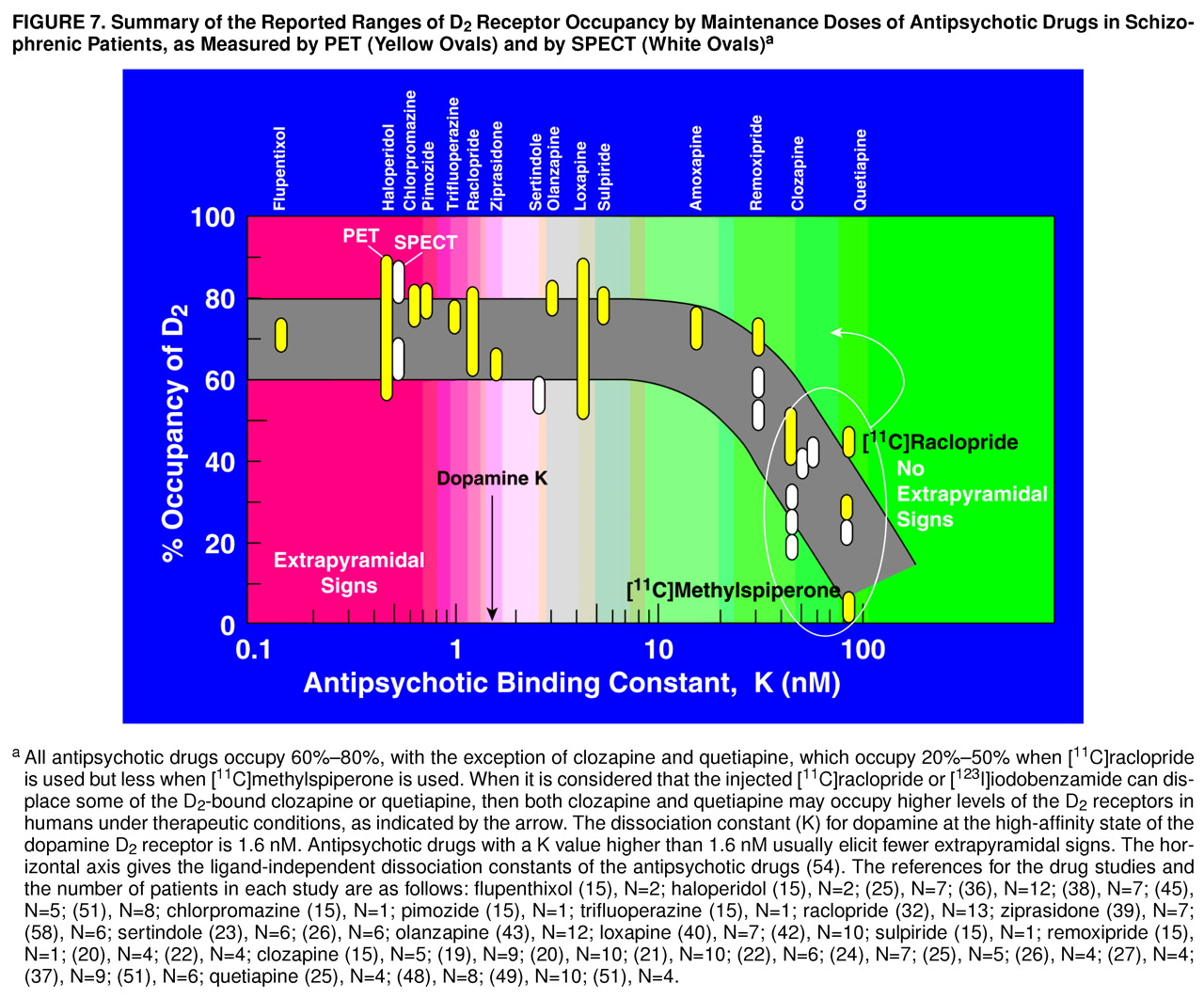
Considering that iodobenzamide, as well as raclopride, may displace some of the D
2-bound clozapine or quetiapine in the human brain striatum, then clozapine or quetiapine may occupy more dopamine D
2 receptors in humans than are currently estimated. This is illustrated in
Figure 7, which summarizes the observations that all antipsychotic drugs occupy 60%–80% of D
2 receptors, except for clozapine and quetiapine. However, if allowance is made for some displacement of the D
2-bound clozapine or quetiapine by the injected radioligand, then the D
2 occupancies by clozapine and quetiapine may rise. This conclusion is the same as that derived previously by using the antipsychotic molarities in plasma water and using the ligand-independent dissociation constants of the antipsychotic drugs
(54).
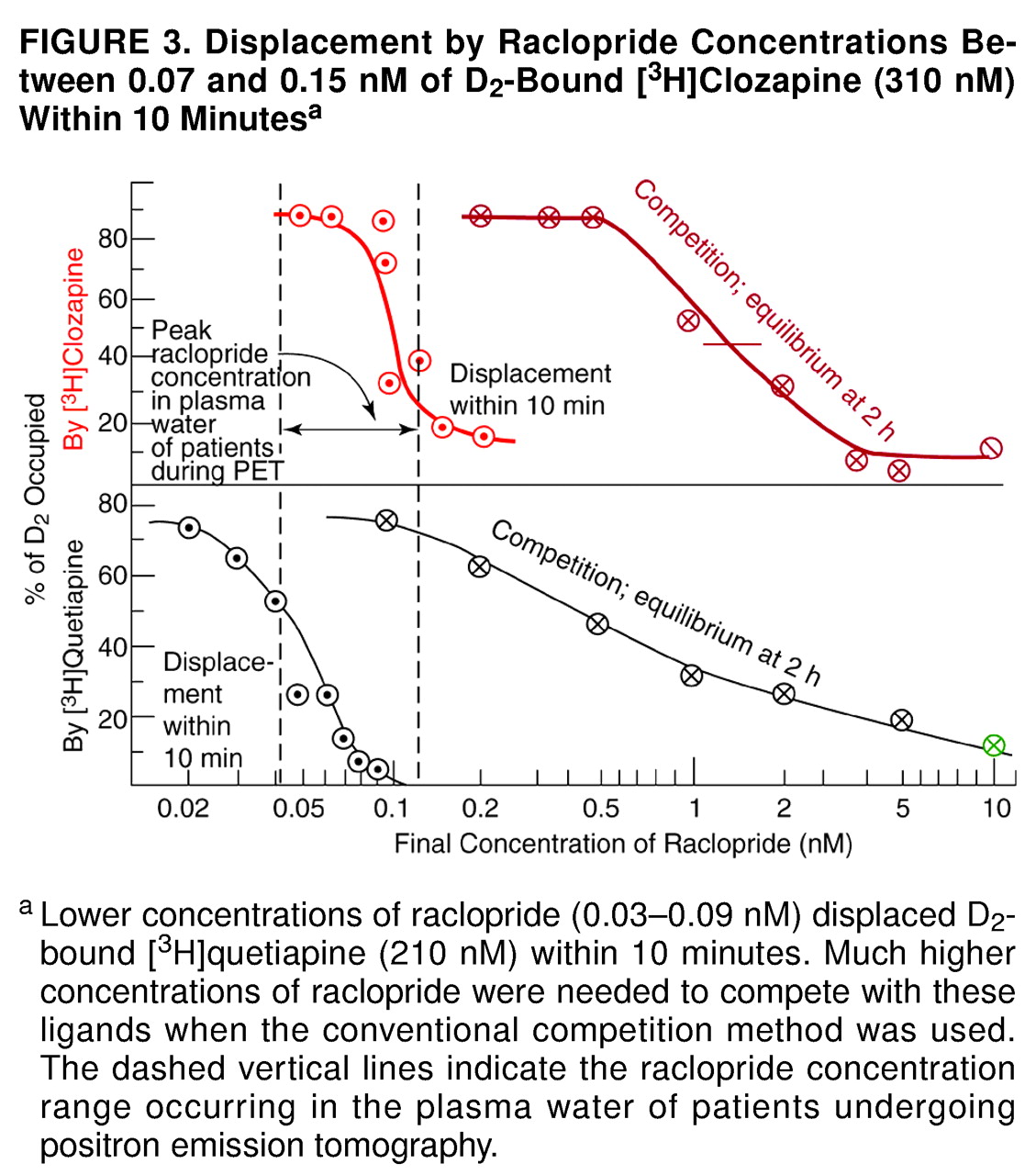
A particularly surprising result of this study was the observation that preexposure of the cloned D
2 receptors to clozapine caused the receptors to be more readily displaced by raclopride. As shown in
Figure 3, when the radioactive clozapine and the nonradioactive raclopride were added simultaneously (as is done in the traditional competition-type method), the K value (dissociation constant) of raclopride was about 1 nM, in agreement with previous data in vitro
(53,
54) as well as in vivo in human positron tomography. (Although the raclopride K value in the striatum of human volunteers is between 7.7 and 9.1 nmol/gram of striatum [
64,
71], these values convert to 1.5–1.8 nM, given that the tissue/buffer partition coefficient for raclopride is 5 [nmol/gram]/[nmol/ml] [
53,
72–
74), and L. Farde, personal communication].) However, when the cloned D
2 receptors were preexposed to clozapine for 1 hour, raclopride displaced 50% of bound radioactive clozapine at 0.1 nM (
Figure 3). When the preexposure was shortened to 10 minutes, the results were the same as the results when the competition method was used (data not shown). In other words, the prolonged exposure of the cloned D
2 receptors to clozapine for 1 hour increased the D
2 affinity for raclopride. Although the molecular basis of this effect is not known, G protein-linked receptors can interact and cooperate to exist as dimers or multimers with multiple states of affinity for an antagonist, as found for muscarinic receptors
(75,
76) and dopamine D
2 receptors
(77). It is possible, therefore, that preexposure of D
2 receptors to clozapine may alter the affinity state of D
2 for raclopride, but such speculation must be directly tested in future experiments.
The present data (figures 1–5) are consistent with the time course of D
2 occupancy measured clinically. For example, whereas the D
2 occupancy by haloperidol in patients is long-lasting
(78), the D
2 occupancy by clozapine or by quetiapine in patients continues only for a matter of hours
(7,
50).
Although the data from this study show that clozapine and quetiapine are readily released from D
2, these data do not reveal the time course of release of these antipsychotic drugs from D
2 under ordinary clinical conditions (i.e., in the absence of brain imaging experiments). However, the data in
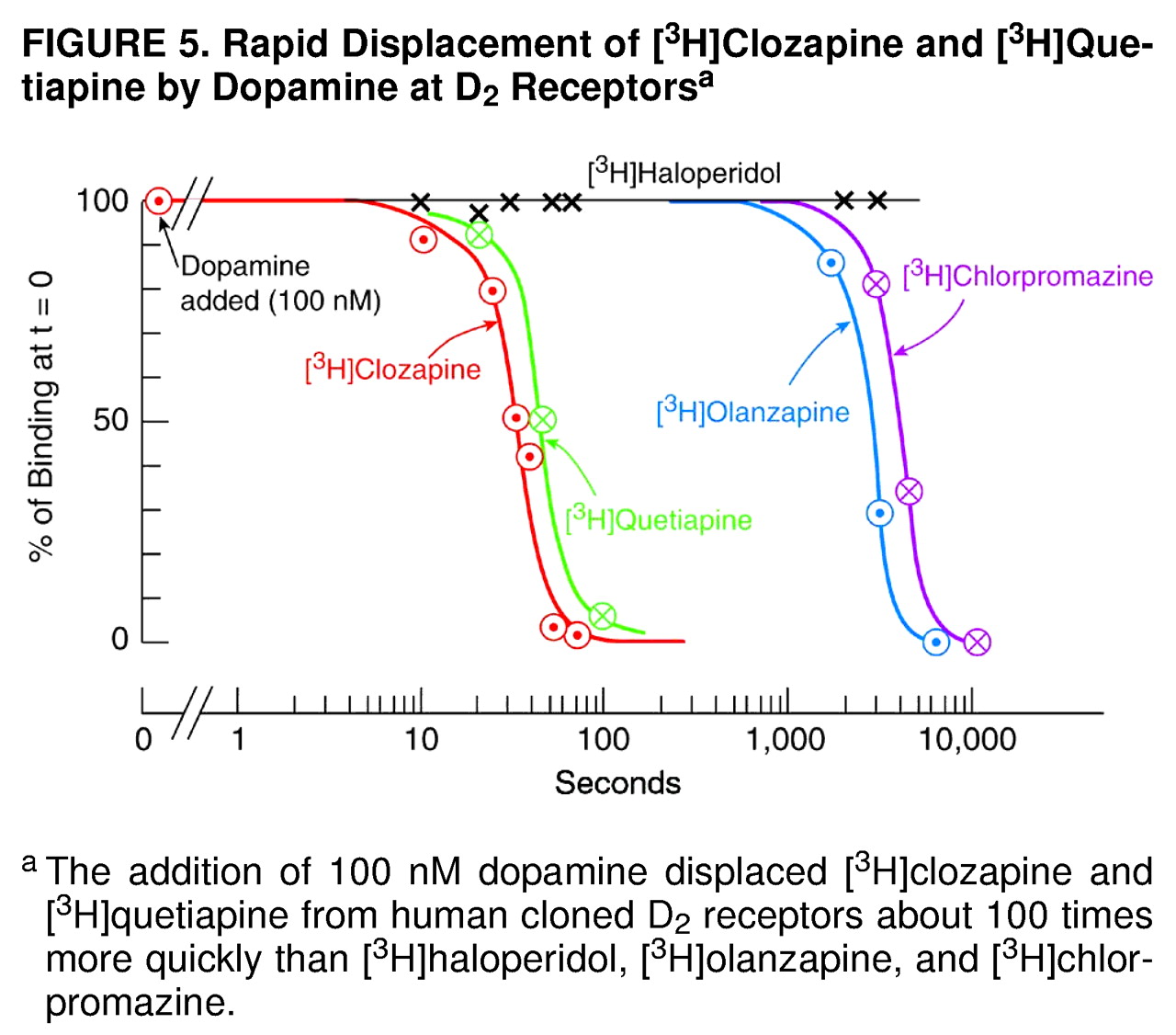
Figure 5, showing the rapid displacement of clozapine from D
2 by 100 nM dopamine (which is the average level of dopamine in the synaptic space
[79]) is consistent with the hypothesis that these antipsychotic drugs are displaced from the patient’s D
2 receptors by endogenous dopamine (reviewed in reference 54), thereby contributing to the early reemergence of psychotic symptoms
(1–
11).
Additional data (not shown) revealed that an extremely high concentration of dopamine (30 µM) displaced 50% of [
3H]quetiapine in 4 seconds, 50% of [
3H]clozapine in 9 seconds, and 50% of [
3H]haloperidol in 7 minutes; the result with [
3H]haloperidol is in excellent agreement with the previously known value of 7.5 minutes for the time needed for 50% dissociation of haloperidol from the D
2 receptor
(80). Lower concentrations of dopamine displace [
3H]clozapine more slowly, as just mentioned. The physicochemical factors determining the “loose binding” properties of quetiapine and clozapine are not known, although, presumably, the oil/water (or membrane/buffer) partition coefficients of these drugs are important.
Remoxipride has a K value of 30 nM, close to the values of 44 nM for clozapine and 78 nM for quetiapine (
Figure 7), yet it occupies about 70% of D
2 (56). As summarized in
Figure 7, however, it appears that the region between 30 and 40 nM for the antipsychotic dissociation constant is a transition region, above which displacement of the antipsychotic drug appears to occur, but below which displacement of the antipsychotic does not appear to occur. Melperone, however, has a dissociation constant of about 50 nM
(54) yet occupies 70% of D
2 receptors (56). One of the problems in analyzing the action of melperone is that it has an active metabolite, FG5155
(81), which is also an antagonist at D
2 receptors (unpublished data), complicating the interpretation of the pharmacology involved.
The present data, indicating that [
3H]clozapine binds loosely to the D
2 receptor, are also consistent with the observation that clozapine occupies more D
2 receptors in the cerebral cortex of patients than in the striatum
(82). That is, the higher output of endogenous dopamine in the striatum readily displaces more D
2-bound clozapine in the striatum, compared with the lower output of dopamine in the cerebral cortex
(54).
The summary in
Figure 7 omits the important drug risperidone. The risperidone dose range of 2–6 mg/day occupies between 60% and 80% of D
2 receptors, with “mild” parkinsonism occurring in patients taking 6 mg/day and having D
2 occupancies higher than 75%
(35,
37,
44,
83,
84). Such extrapyramidal signs, however, are generally low or negligible in patients taking 2–4 mg/day of risperidone and having D
2 occupancies between 60% and 75%
(35,
37,
44,
83,
84).
The observations in the present study (figures 1–4) also illustrate the principle that D
2 occupancy by an antipsychotic drug depends on the radioligand used
(53,
54). Other studies have also shown that a radioligand should have a lower affinity than the drug for which a site’s occupancy is being probed
(85–
88). PET receptor occupancy studies are relevant to clinical realities, but technical aspects must be recognized before results can be usefully interpreted.
In conclusion, with respect to the apparently low D
2 occupancy of clozapine, it may not be that “less is better” for the patient
(89), but rather that “loose is better.”