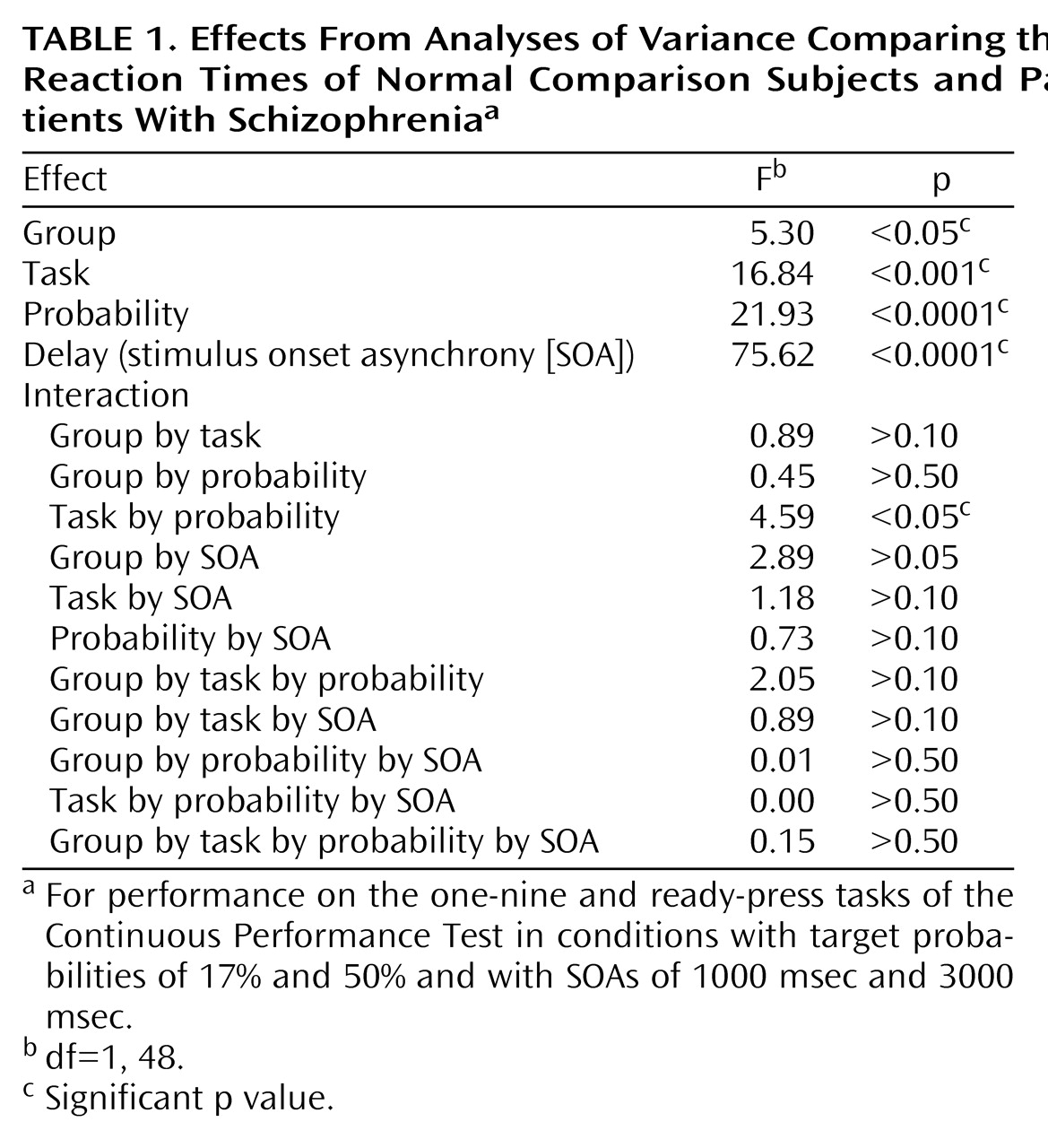
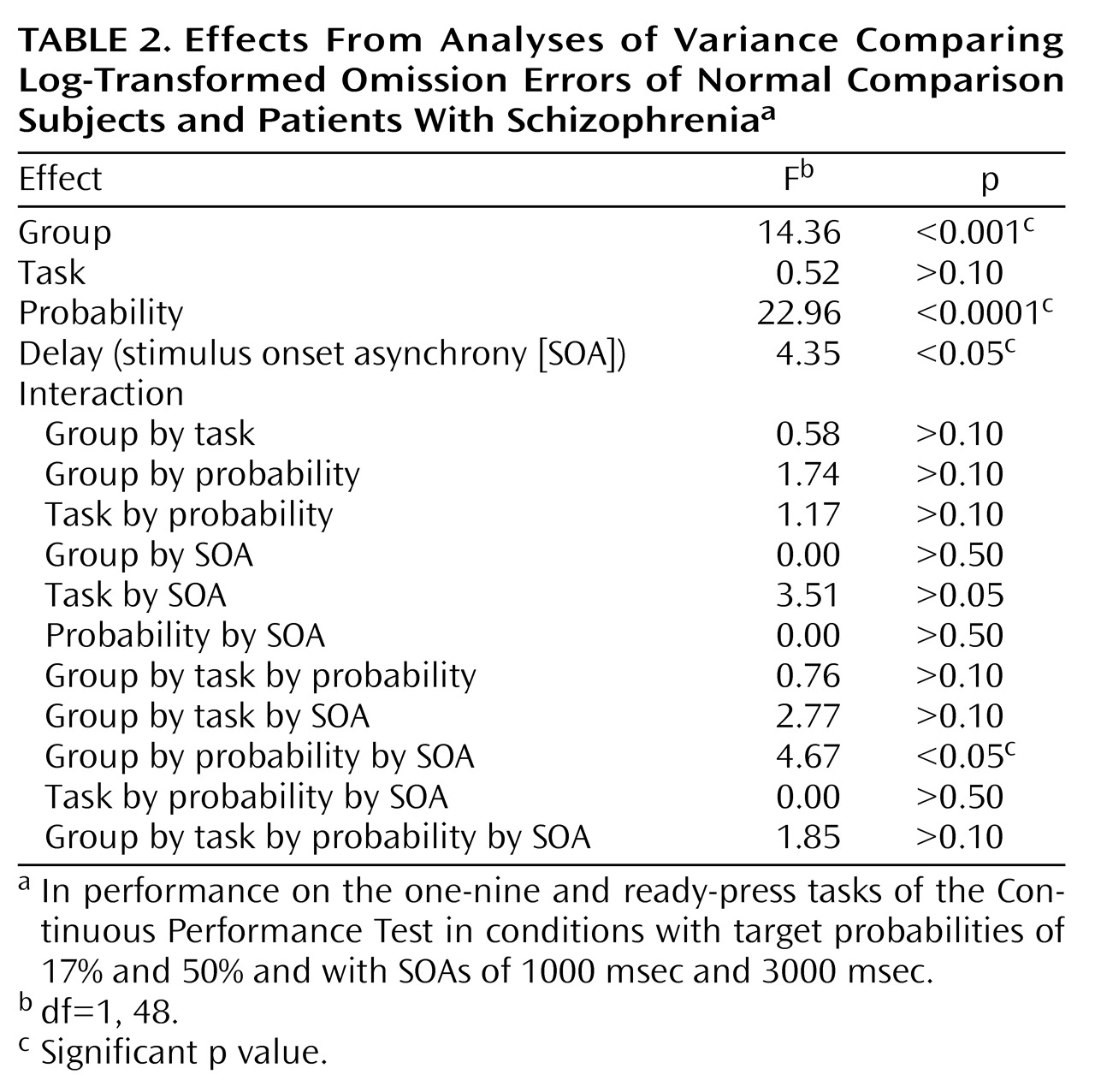
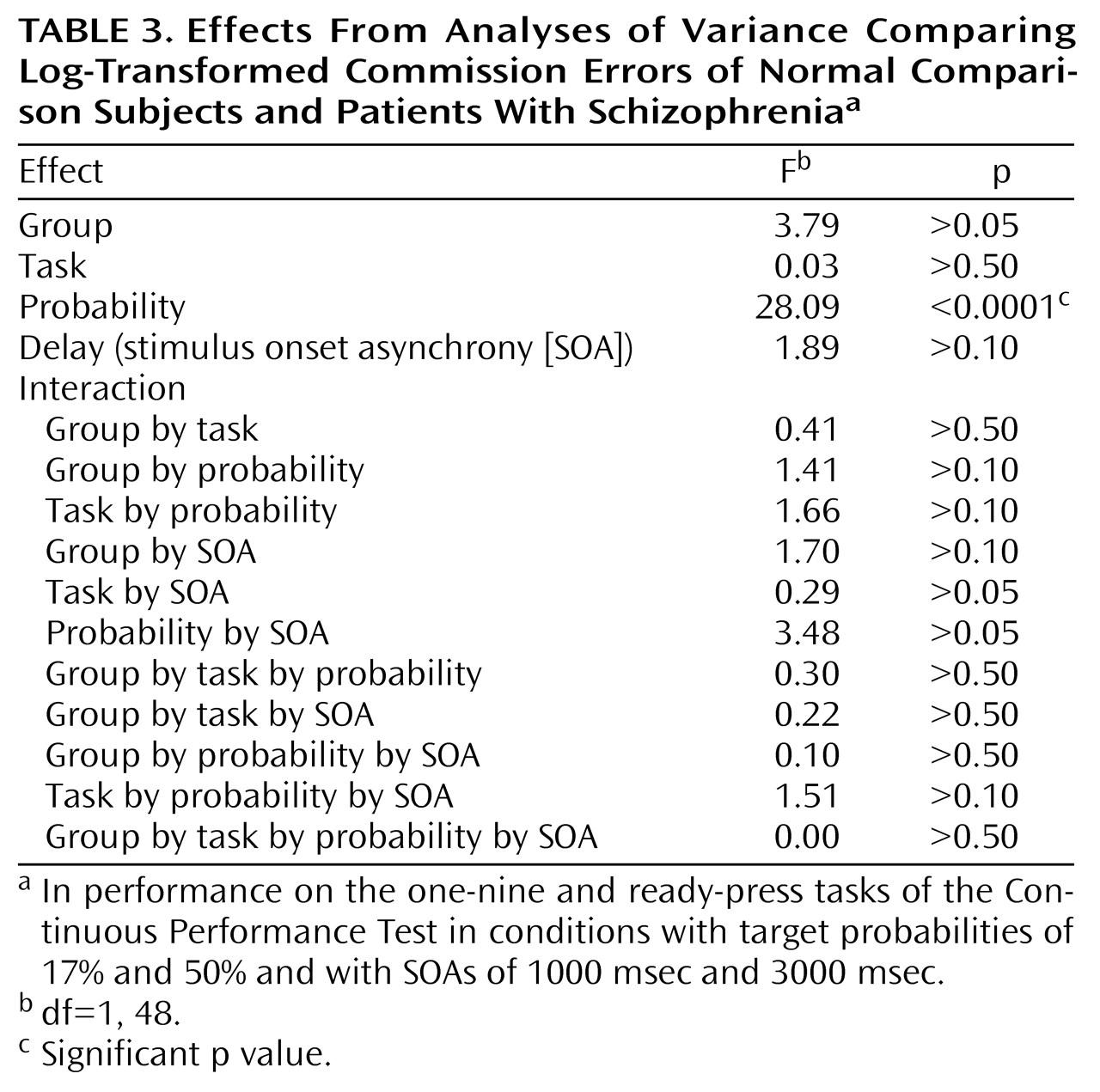
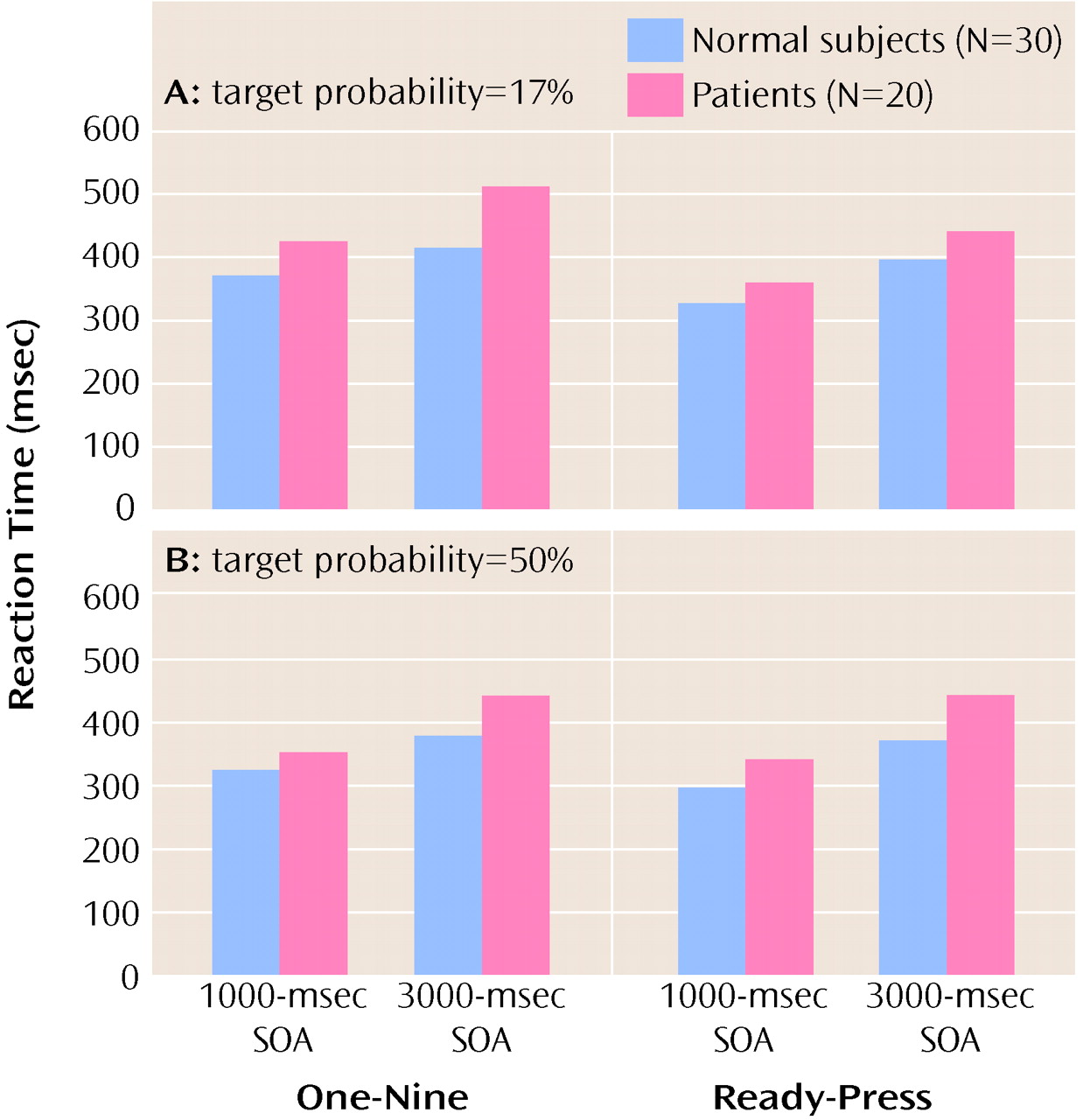
The Continuous Performance Test
(2) has been widely used to measure sustained attentional deficits in schizophrenia. The test measures visual vigilance, which has been used as a principal experimental index of these deficits. In the basic form of this test, subjects monitor a random series of single numbers or letters, which are presented continuously, often at a rate of approximately one per second. Subjects are asked to indicate that they have detected a target event by pressing a response button and to avoid responding to distracting stimuli. The target event may be the appearance of a single stimulus (e.g., the letter “X” appearing in a sequence of letters; this task can be termed Continuous Performance Test–Single) or a stimulus appearing in a particular context (e.g., the letter “X” only when it follows the letter “A;” this task has been termed the Continuous Performance Test–AX)
(2). Subsequent modifications have been made to increase sensitivity (e.g., through degradation of stimuli or use of two consecutive identical stimuli in which the target event is the second stimulus [Continuous Performance Test, Identical Pairs Version])
(3). Several hypotheses concerned with performance of patients with schizophrenia on the Continuous Performance Test have been proposed. For example, the finding that patients with schizophrenia perform especially poorly on vigilance tasks with high processing loads has led to suggestions that the deficiencies are due to a “reduction in available processing capacity or a temporary disruption of automatic as well as attention-demanding processes”
(4). Patients’ attentional deficits seem to be most reliably elicited on variants of the Continuous Performance Test that have relatively high processing loads (i.e., tests with rapid stimulus presentation, those with stimulus degradation, or those in which memory of the previous stimulus is necessary to make the current decision). Thus the essential problem would seem to be a difficulty with the high moment-to-moment demand on processing capacity. Although the essential components of this deficit remain obscure, Cohen and colleagues
(5,
6) recently operationalized a possible mechanism for this deficit. By adopting a computational approach, they proposed a defective mechanism that would become evident in performance on the Continuous Performance Test when the subject must keep the stimulus information in mind during a delay period between one stimulus and the next. This requirement would tax the already limited available processing capacity, with the result of poor performance in patients with schizophrenia. In the study reported here we wished to explore this issue further by empirically evaluating four possible mechanistic accounts of performance on the Continuous Performance Test in patients with schizophrenia.
Discussion
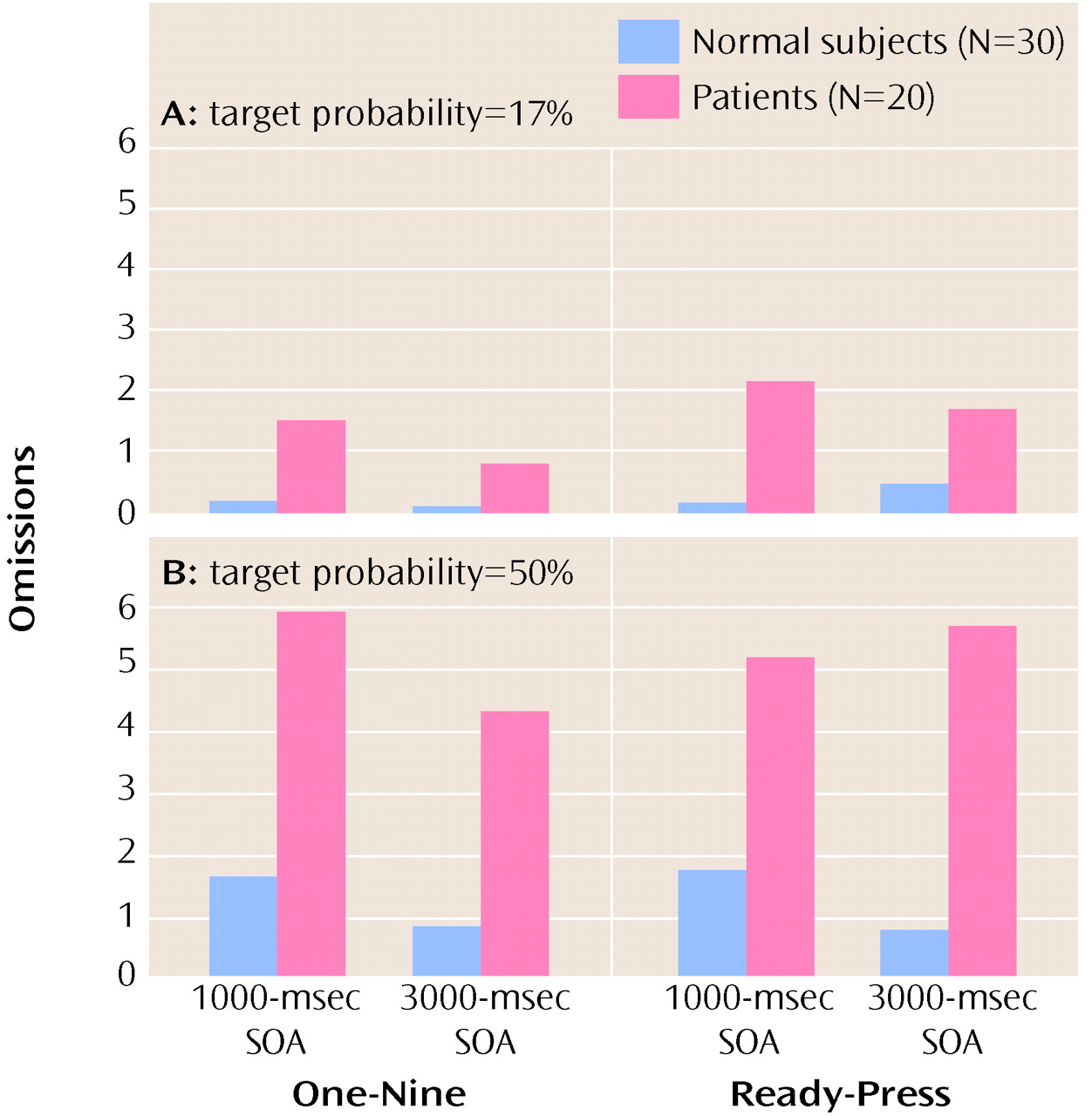
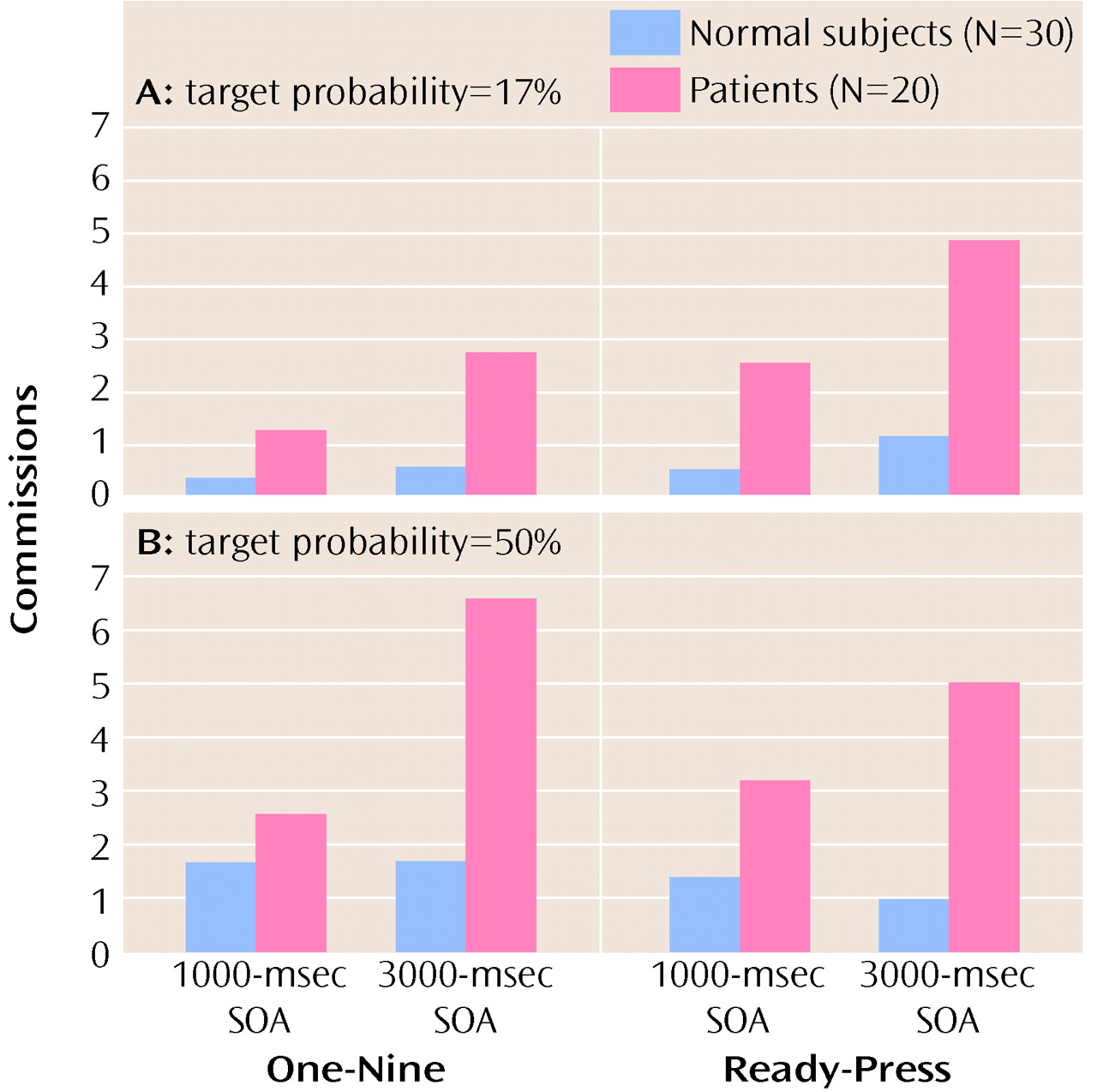
In this study we tested four possible mechanistic explanations of impaired performance on the Continuous Performance Test in patients with schizophrenia, yet we found no support for any of these hypothesized factors. First, we manipulated the amount of stimulus-response mapping. This procedure resulted in superior reaction times on the “ready-press” task compared with the “one-nine” task (i.e., the manipulation worked). However, it did not produce a disproportionate deficit in performance in patients with schizophrenia. Second, we increased the target probability to bias subjects to make responses. We expected that patients with schizophrenia would have disproportionate problems in inhibiting responses. Again, the manipulation worked in the sense that a difference in performance between the two tasks was found. However, despite previous research indicating that patients with schizophrenia have such inhibition problems, we found no evidence of this in our study. Third, we manipulated the length of time that subjects had to hold a previous stimulus in working memory. Again, although performance on the two tasks (SOA=1000 msec and SOA=3000 msec) was different, the patients with schizophrenia in our study did not show the expected (and previously found) decrement in performance as the SOA increased. Finally, we manipulated the amount of available motor planning resources available by adding a dual task component to the test (having subjects tap concurrently). Although, this manipulation worsened performance marginally, again there was not a group difference. To summarize, none of the four manipulations we employed produced the predicted differences in the performance of patients with schizophrenia on the Continuous Performance Test. However, it is important to keep in mind that patients’ performance on the test tasks (accuracy or reaction time) was always worse than comparison subjects’ performance (see below).
We suggest three possible reasons why we did not find differential changes in performance on the Continuous Performance Tests in the patients with schizophrenia. First, the findings may simply have resulted from a cohort effect, i.e., the patients in our study were not representative of the population of patients with schizophrenia (see, for example, reference
6, unpublished 1998 manuscript of Elvevåg). However, we argue that this probably was not the case, because the patients in our study had performed badly on other frequently used Continuous Performance Test tasks, such as the 1–9 (digit) task in the Gordon Systems (GDS-Model II)
(16). Furthermore, our sample size was adequate (patient N=20) and the diagnosis was reliably established by SCID (DSM-IV). Most important, the patients in our study by no means performed normally; their performance was consistently and significantly less accurate and slower than that of comparison subjects. Thus, we contend that previous findings may not be as reliably replicable as we expected.
Second, is it possible that we failed to operationalize the variables appropriately, even though each variable produced a significant main effect in terms of reaction time (“ready-press” only) and omission or commission errors. Manipulating stimulus-response mapping affected performance on the Continuous Performance Test but did not affect performance differently in the two groups. Regarding SOA, it is possible that if we had made the interval longer, to make the task disproportionately more difficult for patients, a difference between the groups would have emerged. However, previous research has shown that the performance of patients with schizophrenia suffers disproportionately even at a delay period of 2000 msec (e.g., unpublished 1998 manuscript of Elvevåg). Alternatively, it may be that the target probability was not high enough. In other variants of the Continuous Performance Test it is possible to set the cue-target ratio at 80%. A study by Servan-Schreiber et al.
(6) found that unmedicated patients with schizophrenia have a disproportionate problem overriding their overlearned response tendencies at long delay periods, compared to normal subjects and patients with schizophrenia who have received medication. In the Continuous Performance Test task used in this study, it was not possible to increase the target probability much higher than 50% without producing an obvious and predictable pattern of stimuli. It is possible that this procedure did not build up a response tendency as strong as the one that would have resulted from a task that required subjects to respond to every stimulus rather than just to the stimuli that called for “yes” responses. What is more probable is that building up response bias is not a particularly reliable way to elicit errors in patients with schizophrenia. Even Servan-Schreiber et al.
(6), who proposed this parameter, failed to find that patients who were receiving medication were especially susceptible to errors on overlearned pairings at long SOAs (see also unpublished 1998 manuscript of Elvevåg for similar findings). Moreover, it is very important to note that Servan-Schreiber et al. employed psychiatric inpatients (with depression) with unreported medicated status as comparison subjects. This being said, their general finding that all subjects performed better in the short SOA condition was not replicated in our study. However, we do not disagree with the conclusion of Servan-Schreiber et al. that delayed response deficits are an important component of cognitive impairments in schizophrenia. Certainly these deficits have been reliably observed in a variety of paradigms (e.g., references
17–
22). Rather, we posit that in the “classic” Continuous Performance Test (relative to low target probability and short SOA) different factors lie behind poor performance.
Third and finally, the dual task component did not interfere with performance on the Continuous Performance Test as much as we expected and, most important, did not do so differentially. Perhaps this result occurred because much of the processing of Continuous Performance Test tasks takes place in cortical association areas, not in the motor system. If so, it is possible that we needed to employ a more complex concurrent task, such as arithmetic problem solving.
We believe that our data suggest that patients’ impairments on the Continuous Performance Test are due to encoding problems. If this is so, a shorter, rather than a longer, SOA might have a differential impact on the performance of patients with schizophrenia. Certainly this explanation is consistent with the high sensitivity shown on versions of the Continuous Performance Test in which the stimuli are degraded (e.g., reference
23) or in which the stimulus duration is very brief (e.g., reference
24). The significant interaction effect of group, target probability (low), and SOA (short) on omission errors is consistent with the idea of an encoding deficit in patients with schizophrenia. For example, if patients were slow in constructing a representation, a short SOA would more likely lead to interference, as a second stimulus would exact a cost on ongoing processes. Such a situation would result in patients’ making relatively more omission errors, but patients thus might have a more conservative response bias in environments in which much information is not salient (e.g., the low target probability condition). It is noteworthy that studies using EEG
(25) and pharmacological interventions in monkeys
(26) have shown that “imprecision” in encoding can account for much of the variance in simple attention and working memory tasks. It is interesting to note that the anterior cingulate, thought to play a key role in attention, also appears to become preferentially active under difficult encoding conditions
(27).
The study reported here produced unexpected negative results. Although group differences were large because patients made more errors and were slower, experimental parameters designed to manipulate response readiness, stimulus-response mapping, and working memory demands had little differential impact on patients. Thus, simple notions about Continuous Performance Test failure do not appear to account for the performance of patients with schizophrenia, at least in this study group. However, the results may still be heuristic because they indicate that in certain situations or conditions (of low target probability and short SOA), patients appear to have difficulty encoding (i.e., constructing a representation) and making rapid subsequent decisions of whether to respond.
The Continuous Performance Test provides clinicians and researchers with a valuable tool to index one of the key cognitive deficits characteristic of schizophrenia. The usefulness of this task is not in question. What our study does question is the explanatory power of certain contemporary mechanistic accounts of performance on the Continuous Performance Test, most notably our own. Perhaps Continuous Performance Test omissions represent a frank failure of patients’ ability to encode stimuli, due either to network shutdown or increases in background noise.