This work, involving grief caused by a recent separation, adds to the growing body of information about the brain regions involved in sadness. Of importance is that this study lays the groundwork for future brain imaging studies examining the relationship between transient sadness, grief, and clinical depression. Our study specifically examined women grieving a breakup with a romantic lover. Almost all subjects would have met the criteria for depression for a period less than 2 weeks after breakup. Most, however, did not meet the 2-week depression time criteria. At study time, subjects generally were still ruminating about their ex-lover, but most had resolving depressive symptoms. Only one subject met criteria for a major depressive episode at study time.
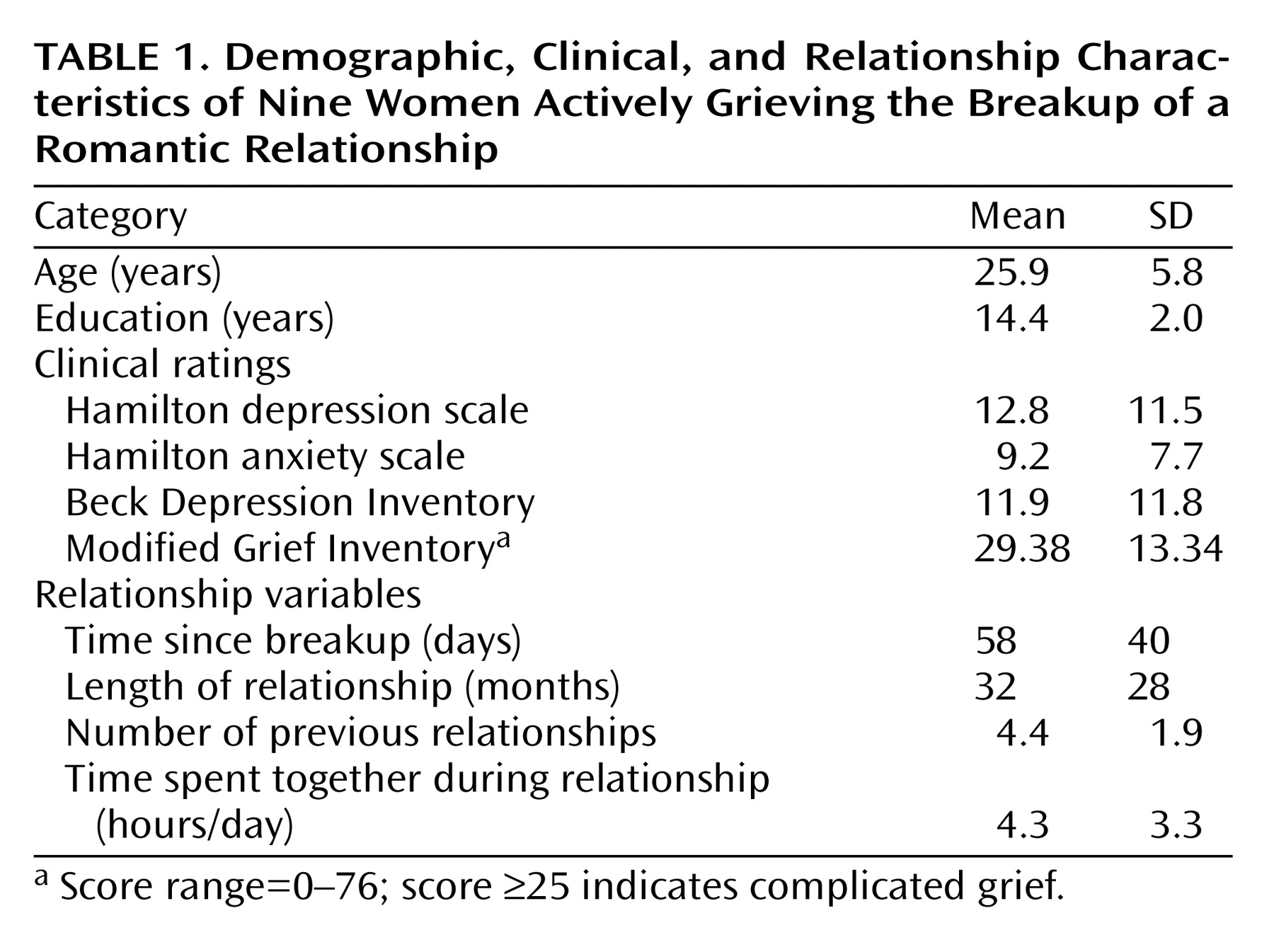
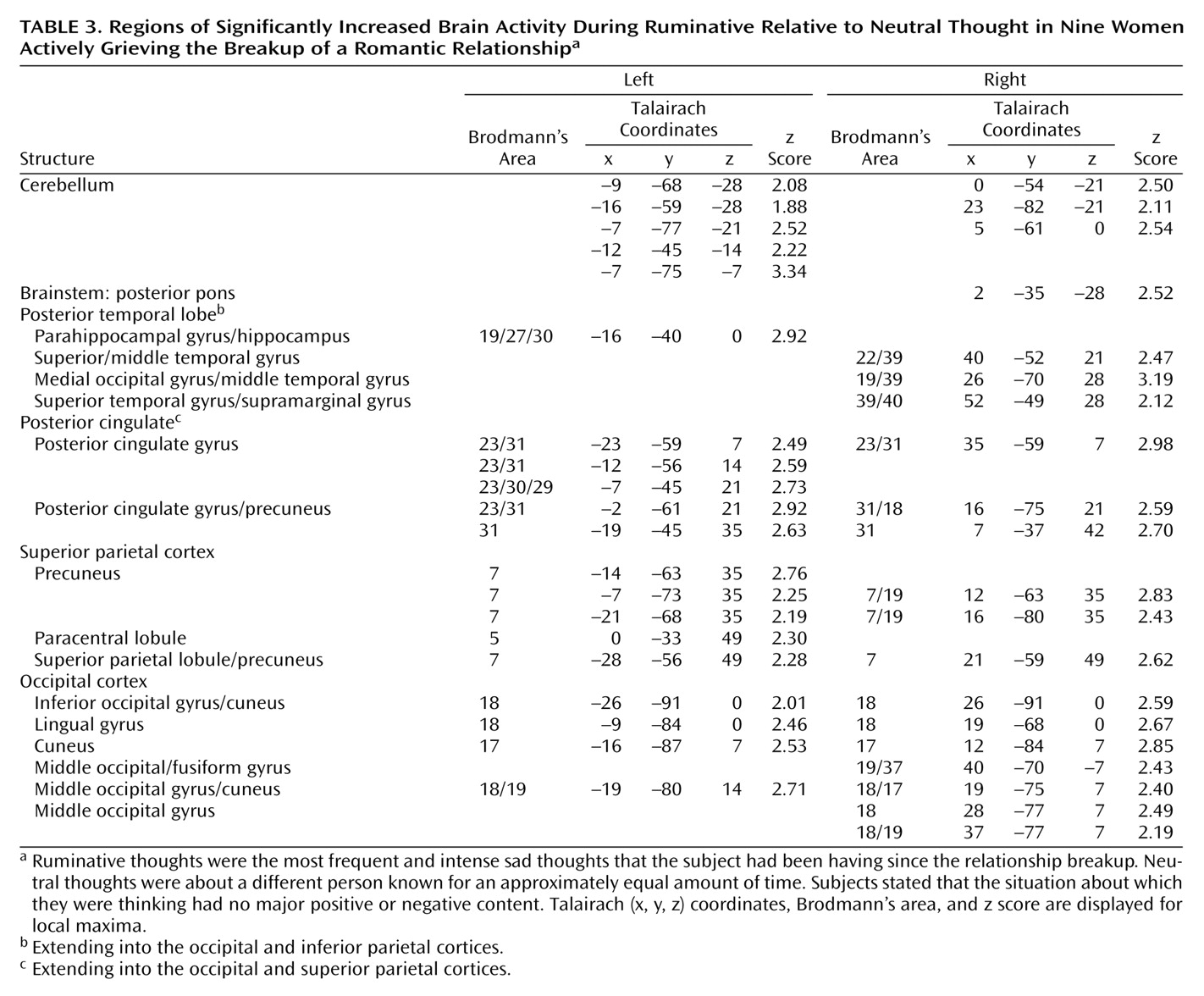
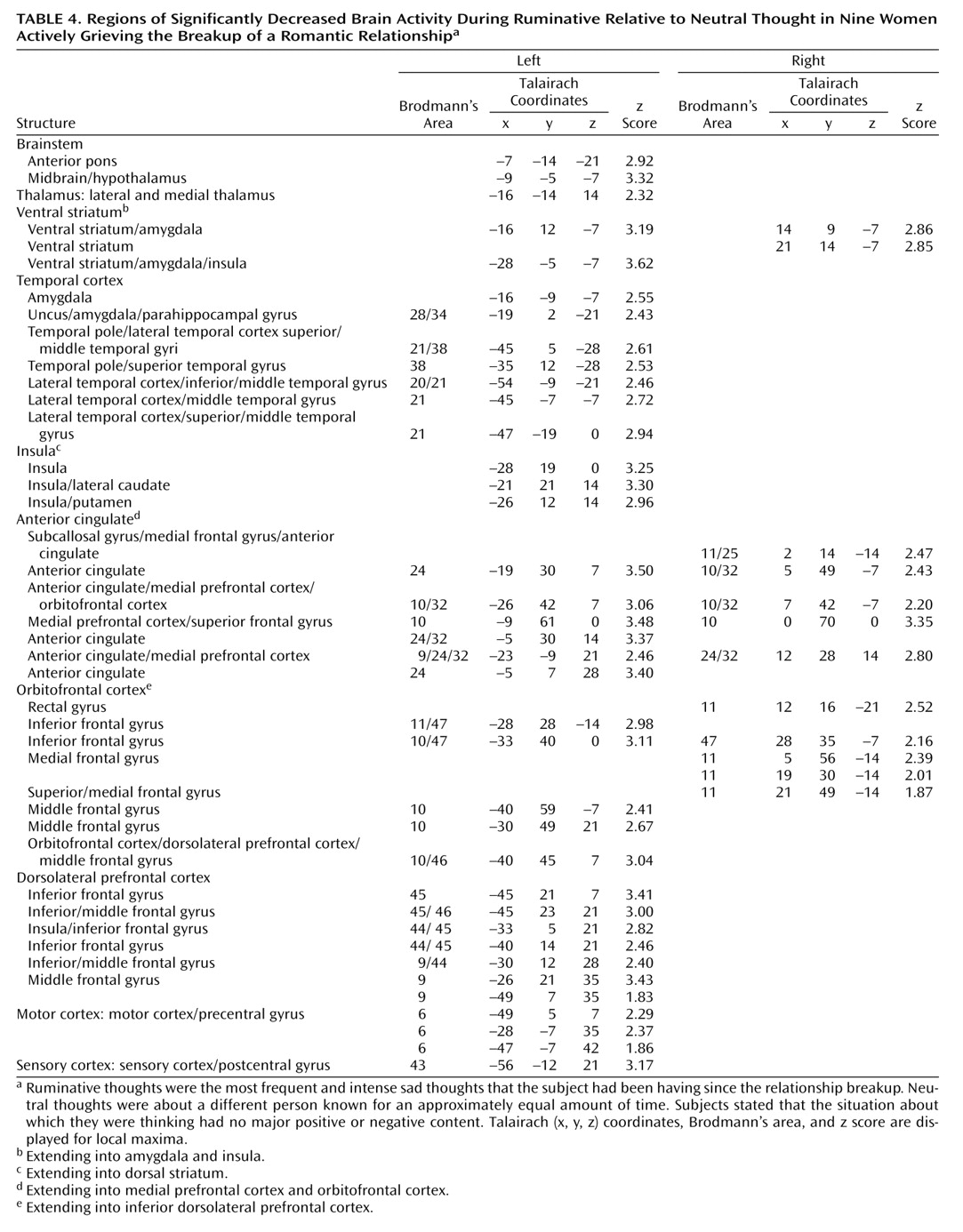
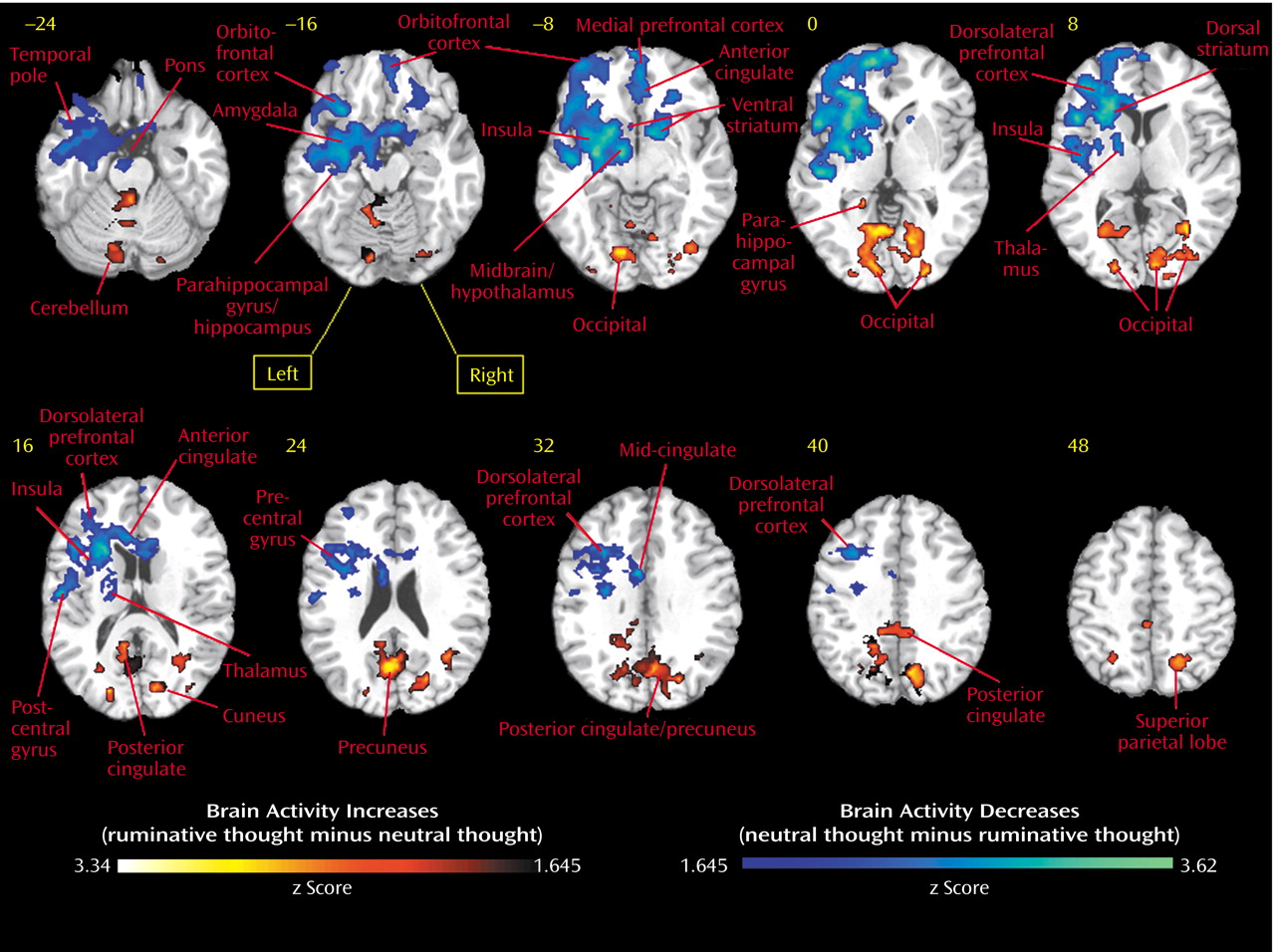
In our study, we found as hypothesized that in premenopausal women who had recently experienced the breakup of a romantic relationship, ruminating about their ex-lover was associated with altered activity in the cerebellum, anterior temporal cortex, insula, anterior cingulate, and prefrontal cortex. Brain activity increases during ruminative relative to neutral thought were generally more prominent in posterior brain regions, including the cerebellum, occipital, and posterior parietal-temporal cortices. Decreases were more prominent on the left and in anterior brain regions, including the anterior brainstem and striatal, thalamic, limbic, temporal, and prefrontal cortices. Specifically, we found brain activity increases during ruminative relative to neutral thought in the cerebellum, posterior brainstem, bilateral lateral temporal cortices, supragenual anterior cingulate/medial prefrontal cortex, right inferomedial dorsolateral prefrontal cortex, and posterior temporoparietal and occipital brain regions. The more anterior region increases—including the lateral temporal cortex, anterior cingulate, and prefrontal cortex—were only present with a more lenient statistical threshold, and all lied adjacent to regions of brain activity decreases during ruminative relative to neutral thought. Activity decreases during ruminative relative to neutral thought were generally more on the left than the right and included the anterior brainstem, thalamus, striatum, medial and lateral temporal cortex, insula, anterior cingulate/medial prefrontal cortex (sub-, pre-, and supragenual regions), orbitofrontal cortex, dorsolateral prefrontal cortex, and primary sensorimotor cortex. In post hoc correlations, we found negative correlations between a subject’s baseline grief ratings and activations during ruminative relative to neutral thought in brain regions similar to where the group showed decreased activation during ruminative relative to neutral thought, except that the orbitofrontal cortex and ventral anterior cingulate/medial prefrontal cortex negative correlations were limited to small portions of the subgenual anterior cingulate and right anterolateral orbitofrontal cortex, respectively.
Relationship With Prior Imaging Studies of Sadness and Depression
There have been seven independent samples of healthy individuals studied while they recalled sad memories as compared with neutral memories or rest
(4,
7,
8,
10,
12,
16,
20,
35). All seven examined increases during sadness relative to a control condition, and four also examined decreases. Unlike our study, these were all [
15O]H
2O PET studies, and the sad event picked was generally not an event about which the subjects were actively grieving. In general, these sadness induction studies showed increased subcortical activity, increased or altered paralimbic region activity, and decreased activity in more cortical regions. Namely, consistent findings in healthy individuals undergoing sadness induction included cerebellar increases, brainstem/hypothalamus increases, thalamic increases, striatal increases, insular increases, changed ventrolateral orbitofrontal cortex activity, mostly ventromedial anterior cingulate/medial prefrontal cortex/orbitofrontal cortex increases, changed pregenual anterior cingulate/medial prefrontal cortex activity, changed posterior cingulate activity, mainly dorsolateral prefrontal cortex decreases, lateral temporal cortex decreases, and changed occipital cortex activity.
In our study of actively grieving subjects, we found altered activity in all these brain regions. Consistent with the healthy sadness studies, we found increases in the cerebellum and posterior brainstem, changes in the pregenual and supragenual anterior cingulate/medial prefrontal cortex (increases and decreases supragenually and decreases pregenually), changes (increases) in the posterior cingulate cortex, changes (decreases) in the orbitofrontal cortex, mainly decreases in the dorsolateral prefrontal cortex (extensive decreases on the left but both increases and decreases in a small area of the right inferomedial dorsolateral prefrontal cortex), mainly decreases in the lateral temporal cortex, and changes (increases) in the occipital cortex. In contrast to these studies, we found decreased activity in the anterior brainstem/hypothalamus, thalamus, striatum, anterior lateral paralimbic structures (insula-temporal pole-orbitofrontal cortex), and subgenual/ventral pregenual anterior cingulate/medial prefrontal cortex/medial orbitofrontal cortex. Of interest, we also found decreases in the bilateral medial temporal cortex (left > right) not noted in the sadness studies as a consistent region of change.
While at odds with sadness studies in healthy subjects, our findings of diminished brain activity during ruminative relative to neutral thought in the striatum and anterior cingulate/prefrontal cortex as well as our findings in these regions of decreasing activity during ruminative relative to neutral thought with increasing baseline grief levels are consistent with functional imaging studies of depression, which consistently report mainly decreased anterior cingulate/medial prefrontal cortex activity and decreased dorsolateral prefrontal cortex activity as well as less consistent findings of striatal decreases
(6,
34,
36–39). Our findings, however, are the opposite of consistent depression findings of amygdala, insula, and ventrolateral orbitofrontal cortex increases and less consistent depression findings of thalamic increases
(34,
36–40). Findings of lateral temporal cortex decreases were consistent with healthy sadness studies and some depression studies. In a single [
15O]H
2O PET study of acutely depressed subjects reflecting on sad memories compared with rest
(41), which employed a paradigm similar to our own, the authors also found within-group thalamic decreases as we did but insula and ventrolateral orbitofrontal cortex increases similar to the depression and some healthy sadness induction studies. This sole sadness induction study of depression subjects did not report direct statistical comparisons between depressed and healthy subjects.
In sum, we found activity increases during ruminative relative to neutral thought in the cerebellum, posterior brainstem, lateral temporal cortices, supragenual anterior cingulate/medial prefrontal cortex, right inferomedial dorsolateral prefrontal cortex, and posterior temporal-parietal and occipital brain regions. The more anterior region increases—including the lateral temporal cortices, anterior cingulate, and prefrontal cortex—were all surrounded by regions of more prominent decreases. Activity decreases during ruminative relative to neutral thought were more extensive on the left and located in the anterior brainstem, thalamus, striatum, medial and lateral temporal cortex, insula, anterior cingulate, and frontal cortex. All of these brain regions have been found to be altered in sadness induction studies in healthy individuals. With the exception of the cerebellum and occipital and parietal cortices, all of the regions have also been found to be altered with at least some consistency in depression.
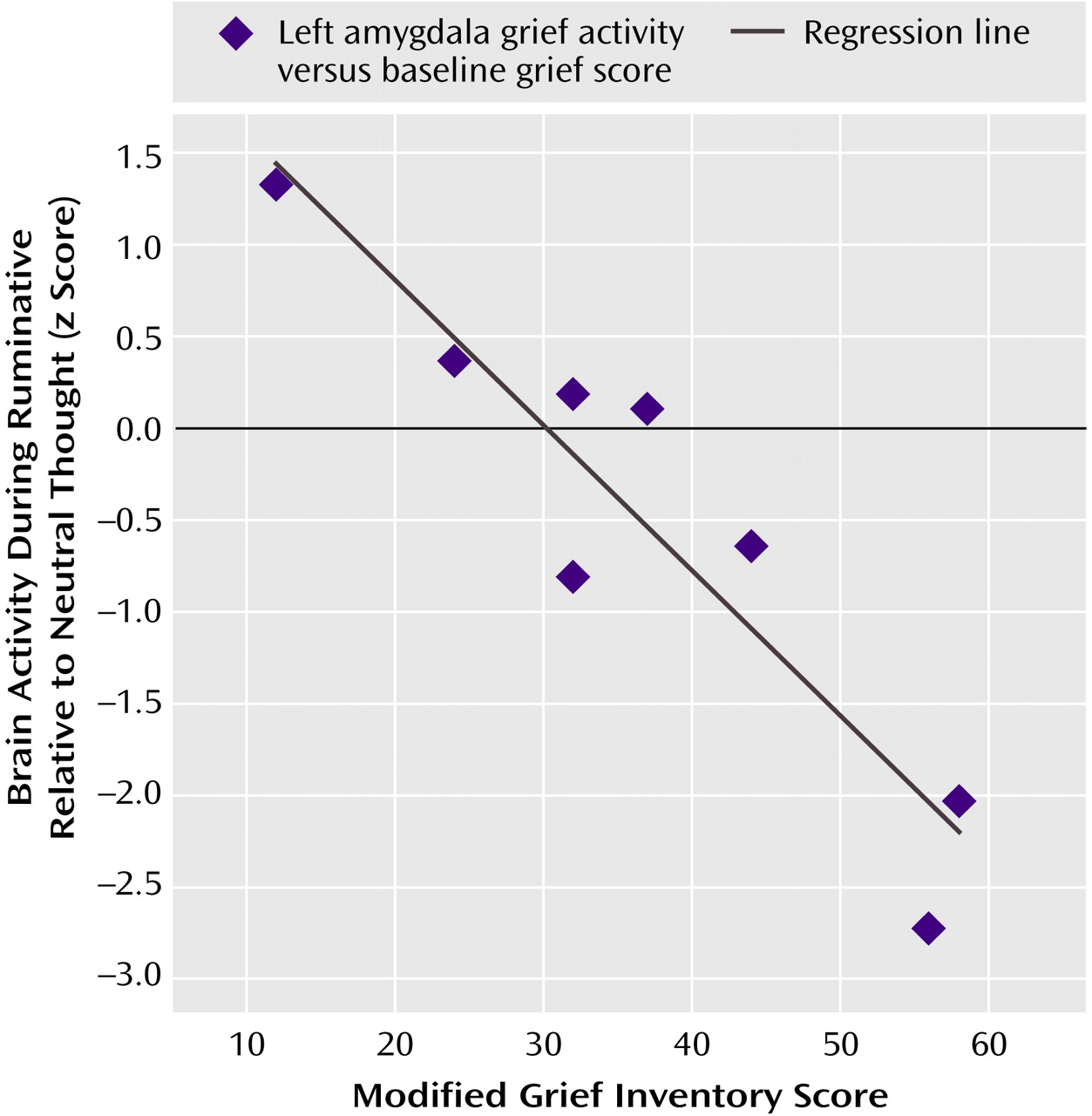
Our most robust finding was of prominent activity decreases during ruminative relative to neutral thought in the subcortex, medial and lateral temporal cortices, insula, anterior cingulate, and frontal cortex. These regions of group decreases also showed diminishing activity during ruminative relative to neutral thought with increasing level of a person’s grief, except for limited findings in the ventral anterior cingulate/prefrontal cortex. Our findings of changed (primarily diminished) anterior cingulate, frontal, and lateral temporal activity in our grieving subjects are consistent with findings in healthy subjects undergoing sadness induction and depression subjects in general. Our finding of decreased insula activity is at odds with consistent findings in healthy sadness induction studies and depression. Our finding of decreased medial temporal cortex (limbic) activity (including the amygdala) is at odds with consistent depression findings and is not a consistent finding in healthy sadness induction studies. Potential reasons for our discrepant findings of medial temporal lobe decreases are as follows. First, our study was not one of pure sadness, instead involving a mixed state of sadness, anger, and anxiety. Also, decreases in the medial temporal region in our actively grieving subjects could be specific to the experience of actively thinking about a negative experience that is on one’s mind constantly, which is different from grief remembered from long ago. Next, perhaps subjects with higher grieving scores have hyperactive amygdala activity during the neutral baseline condition and with further increases in emotion such as ruminating about a lost relationship, they actually get decreases in amygdala activity. A justification for this is that mild to moderate doses of noradrenaline, which increase stress, placed in the amygdala improves fear conditioning in rats, but high doses do not
(42). This may be analogous to the finding that low to moderate stress increases cerebral blood flow (cortically and sometimes subcortically) and performance, whereas high stress levels decrease cerebral blood flow (cortically and sometimes subcortically) and performance
(43–
53). Further, amygdala and medial temporal lobe activity is often noted to be altered rather than increased in anxiety disorders such as panic disorder and post-traumatic stress disorder (PTSD) (J.P. Lorberbaum, unpublished literature review). For example, diminished medial temporal lobe activity has been noted in PTSD subjects internally reflecting on their traumatic event
(54). Next, in some fMRI studies, amygdala activity has been noted to initially briefly increase for roughly 10–15 seconds then decrease below baseline over the course of a single epoch. Amygdala activity has even shown increases in earlier epochs but habituation to below baseline in later ones
(55). Our epochs were 60 seconds and repeated 4.5 times. Last, our sadness induction involved internally generated emotions with eyes closed. With visually (externally) induced sadness, medial temporal lobe activation is common, especially in the amygdala. The phenomenon of predominant amygdala activation by visual mood induction was already statistically substantiated by the meta-analysis of Phan and colleagues
(56). In 50% of the visual induction studies they had reviewed, the amygdala turned out to have increased activity, regardless of the targeted mood. However, in only 7% of internally recall-driven studies were amygdala increases found.
Limitations
Our study had several limitations that should be kept in mind while interpreting its results. 1) Variability: potential causes of variability include the small sample size, varying range of time from breakup, lack of control for menstrual phase, and inattention to cigarette smoking as well as the wide inter-subject differences in distraction, ease of performing the sadness task, and stage of grief. 2) Reversibility of emotion: during fMRI, subjects may not have been able to completely recover from their sad, ruminative state in time for the neutral state. Even though subjects rated themselves less sad during the neutral condition, this may have been true in part due to a response bias, since conditions were categorized as “sad” and “neutral.” 3) Mixed emotions, not pure sadness: during ruminative thought, subjects felt significantly more sad, anxious, and angry than during neutral thought, albeit at a lesser level for the latter two emotions. Thus, the induced emotion was not pure sadness. However, we were more interested in the neural correlates of grief itself than in pure sadness. Grieving about a breakup might therefore be considered to be a mixed emotional state, which may limit comparability with studies examining sadness. 4) Quality of the control task: we tried to match neutral and sad thoughts for the mental techniques used to produce these emotions (i.e., mental imagery and imagining of spoken words) and familiarity of the person in the neutral event. However, one might assume that, compared with the neutral thought state, the ruminative thought content was more important, familiar, and more frequently thought about. Therefore, the occipital activation during sadness might be explainable as a stronger activation of primary and secondary visual areas from more vivid imagery.