Experiencing cancer is a stressful life event. Even after the successful treatment of cancer, patients face many long-term problems, such as fear of recurrence
(1,
2). Previous studies have reported that 1%–54% of cancer patients experience a major depressive episode
(3,
4), but major depressive episodes after cancer diagnosis have generally been assumed to be reactive depression, with a short duration
(2–
4). Although a past history of a major depressive episode before cancer diagnosis has been reported to be a risk factor for a major depressive episode after a cancer diagnosis, patients who experience their first major depressive episode after receiving a cancer diagnosis are frequently encountered in clinical oncology settings
(1,
3,
4). Little is known, however, about the neurobiological basis of first depressive episodes after cancer diagnosis in cancer patients.
Studies using high-resolution magnetic resonance imaging (MRI) have reported an 8%–15% smaller hippocampal gray matter volume in female patients with a history of recurrent major depressive episodes than in matched comparison subjects
(5,
6), and other studies have found 11%–19% smaller hippocampal volumes in a group of male and female patients with recurrent major depressive episodes
(7,
8). Yet another study reported the alteration of temporal gray matter, including that of the hippocampus, in patients with chronic treatment-resistant major depressive episodes
(9,
10). However, several investigators have not found any reductions in hippocampal volume in depressed patients
(11–
13), including some reports that assessed the combined volumes of the amygdala-hippocampal complex as a single measurement
(14–
17). Most of the studies that found a reduced hippocampal volume were performed in older patients
(5,
6,
18), and many of the reports described patients with recurrent
(5–
8) and/or treatment-resistant major depressive episodes
(9,
10). Although one study that reported a smaller hippocampal volume did not find a significant association between the number of episodes and the volume
(18), it has been reported that the hippocampal volume of patients with recurrent major depressive episodes is inversely correlated with the cumulative duration of the depressive episodes
(5,
6,
19) and with the number of years that have passed since illness onset
(8). In addition, prolonged stressor exposure and excessive glucocorticoid treatment have been reported to damage the hippocampal neurons of animals
(20). Thus, the small hippocampal volume observed in patients with recurrent major depressive episodes may be a consequence of prolonged and severe recurrent major depressive episodes
(21), but no direct evidence of causality was obtained in a longitudinal study.
The hippocampus has been found to play a role in the negative-feedback regulation of the hypothalamic-pituitary-adrenal (HPA) axis
(22), which responds to stress. A smaller hippocampus has been reported to alter the response of the HPA axis to stress in animals
(23), and dysregulation of the HPA axis has been reported in patients with a major depressive episode
(24). These findings suggest that a smaller hippocampus may play a role in the development of a major depressive episode after a stressful life event; furthermore, early-life stressors, which are a risk factor for a major depressive episode
(25), have been associated with a smaller hippocampus
(26). Thus, a smaller hippocampus may predispose an individual to the development of a major depressive episode after a stressful life event. To our knowledge, two cross-sectional studies have demonstrated no significant association between hippo-campal volume and the first major depressive episode in a group of men and women
(8,
27). However, one of the two studies reported a smaller hippocampus in a male subgroup, and the possibility that a smaller hippocampus may predispose an individual to the development of a major depressive episode could not be discounted.
To our knowledge, no previous study has investigated whether an association exists between hippocampal volume and a first major depressive episode after a cancer diagnosis in cancer patients. Although some of the factors associated with major depressive episodes after cancer diagnosis are thought to be the same as those in depressed patients without cancer
(28), first major depressive episodes after cancer diagnosis also exhibit unique characteristics, such as the impact of having a life-threatening illness and a relatively late onset. Thus, first major depressive episodes after cancer diagnosis may have a different etiology and pathophysiology from those of chronic major depressive episodes, including the recurrent, treatment-resistant, or early-onset episodes examined in previous studies on the relation between hippocampal volume and depressive episodes. The purpose of this study was to compare the hippocampal volumes of cancer patients who experienced their first major depressive episode after receiving their cancer diagnosis and those of cancer patients who had no history of such episodes at any time in their life. A secondary purpose was to investigate whether an association exists between delayed recall function as a surrogate marker of hippocampal function and a depressive episode after cancer diagnosis.
Method
Subjects
This study was approved by the institutional review board and the ethics committee of the National Cancer Center in Tokyo. The study was performed after obtaining the patients’ written informed consent.
The subjects were recruited between February 1998 and April 1999 during follow-up visits to the Division of Breast Surgery, National Cancer Center Hospital East. We selected all patients in the records for breast cancer surgery who had survived more than 3 years since surgery so that the interview itself would not be distressing
(29,
30). The aim of this protocol study was to investigate the associations between brain morphology and major depressive episodes and distressing cancer-related recollections in cancer survivors. Previously, we reported a significant association between distressing cancer-related recollections and hippocampal volume
(31) in cancer survivors; this previous study population partially overlapped with that of the present study. The inclusion criteria were 1) female gender, 2) age between 18 and 55 years, and 3) no history of major depressive episode before cancer. Only women were selected to minimize sex-based brain differences. Exclusion criteria were 1) double cancer or clear evidence of residual or recurrent cancer during regular medical checkups conducted by an oncologist (S.I.), 2) a history of any neurological disorder or traumatic brain injury accompanied by periods of unconsciousness, 3) a history of substance abuse or dependence, 4) a family history of early dementia among first- or second-degree relatives, 5) any physical symptoms that interfered with daily life, as assessed by performance status defined by the Eastern Cooperative Oncology Groups
(32), 6) psychotropic medication within the previous month, and 7) cognitive impairment defined as a score of less than 24 on the Mini-Mental State Examination
(33,
34).
Of the 394 patients who had survived more than 3 years after surgery, 187 patients met the criteria, and 137 could be contacted at the clinic. Fifty-three of the 137 refused to participate in the study (26 were too busy to participate, 10 were not interested, seven refused to undergo the MRI examination, three lived too far away to come to the hospital, one was averse to the hospital, and six refused for unknown reasons). Eighty-four subjects were interviewed with a semistructured interview, including the Structured Clinical Interview for DSM-IV Axis I Disorders, clinician version
(35), that was conducted by a trained psychiatrist (T.N.). After the interview, nine subjects who had a history of a major depressive episode before the cancer were excluded from the analyses. Seven subjects were excluded because of an MRI acquisition error. The remaining 68 study participants were currently not being treated for breast cancer except by adjuvant tamoxifen therapy (N=29). There were no significant differences in age, number of days after surgery, or pathological stage between the 68 subjects who participated in the study and the 100 eligible subjects who did not.
Whether the subjects had a history of major depressive episode, but not its intensity, was determined by semistructured interview. Two psychiatrists both assessed the same nine subjects, and the kappa value for history of major depressive episodes after cancer diagnosis was 1.0.
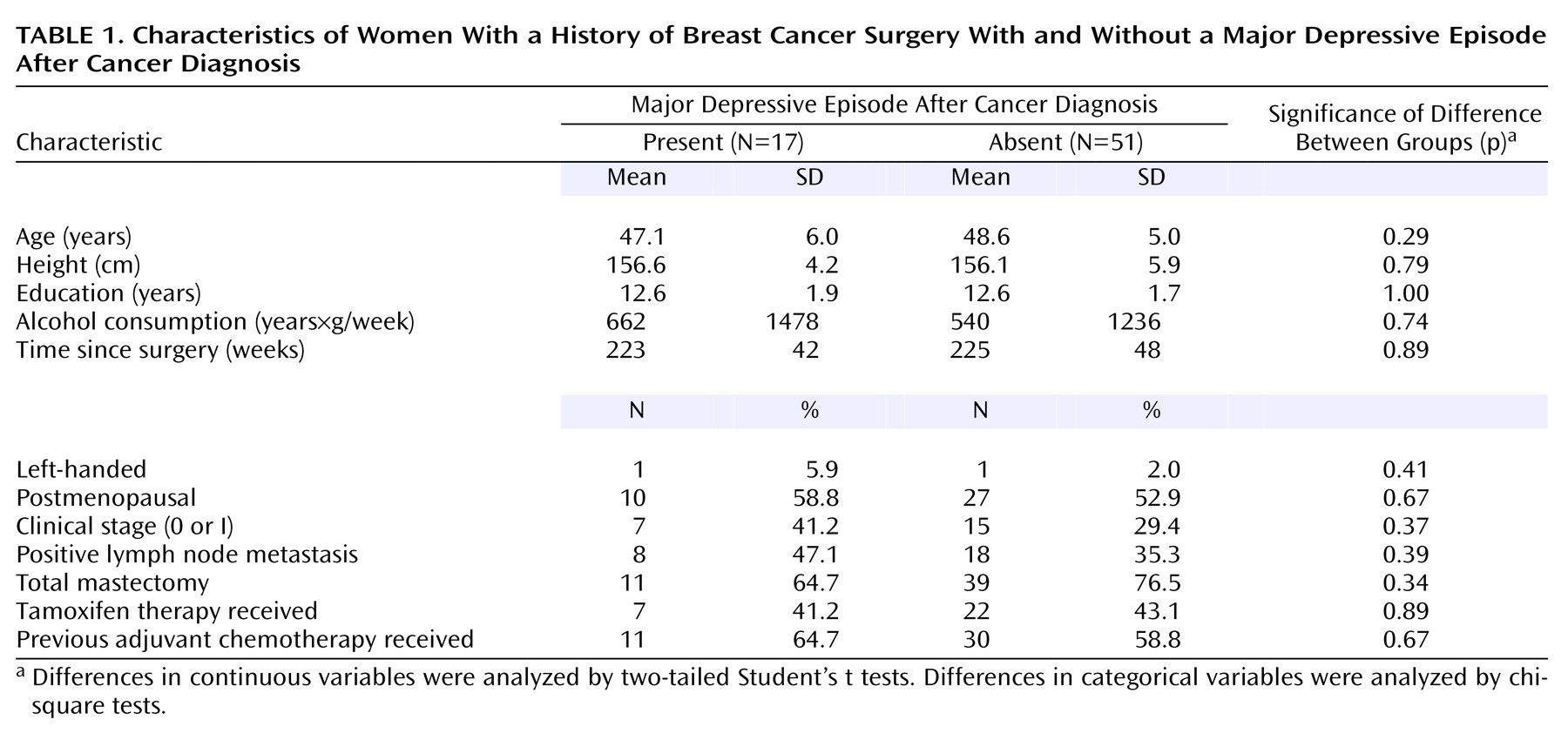
Of the 68 study participants, 17 (25%) met the criteria for major depressive episode after cancer diagnosis. The mean duration of the episode was 11.9 weeks (median=4.0, SD=14.2, range=2–50), and the mean number of major depressive episodes after the diagnosis was 1.1 (median=1.0, SD=0.3, range=1–2). Only two (12%) of the 17 subjects had two episodes after cancer diagnosis; the other 15 (88%) had just one episode. None of the subjects had experienced a major depressive episode with psychotic features, and none of the subjects had a history of any other axis I psychiatric disorder. The 17 subjects had been in remission for a mean of 215 weeks (median=229, SD=66, range=66–334) since the episode. None of the subjects met the criteria for a current major depressive episode at the time of the investigation. Of the 51 subjects who had never experienced a major depressive episode, one subject had a history of posttraumatic stress disorder. As shown in
Table 1, statistical analyses indicated no significant differences in the backgrounds of the groups that did or did not experience a major depressive episode after cancer diagnosis. None of the subjects in the present study had taken any psychotropic medication for at least one month before the MRI examination. Furthermore, none of the subjects had ever taken antidepressants or mood stabilizers. Nine subjects (53%) in the first major depressive episode group had a history of taking anxiolytics or hypnotics (administered for 4 weeks or longer in two subjects and for less than 4 weeks in seven subjects in the past), and 15 subjects (29%) in the group that did not experience a depressive episode had a history of taking anxiolytics or hypnotics (administered for 4 weeks or longer in three subjects and for less than 4 weeks in 12 subjects in the past).
MRI Acquisition and Volumetric Measurements
The details concerning MRI acquisition and hippocampal volume measurements were described in our previous study
(31). Briefly, the images were generated on a 1.5-T MRI unit (Signa scanner, GE Medical Systems, Milwaukee) with three-dimensional spoiled gradient-recalled acquisition of 1.5-mm on contiguous sections perpendicular to the anterior-posterior commissure plane under the following conditions: field of view=230 mm, matrix=256×256 pixels, TR=25 msec, TE=5 msec, flip angle=45°. The images were analyzed by ANALYZE-PC software (Biomedical Imaging Resource, Mayo Foundation, Rochester, Minn.). The boundaries of the hippocampus were similar to those reported previously
(31) and were traced manually with a pen-tablet pointing device. Measurements were mainly made in the coronal orientation, and other perspectives were used to aid anatomic localization. A sagittal view was used to confirm the anterior end of the hippocampal head. The details concerning amygdala volume measurements were described in previous studies
(36). The semiautomatic volumetric procedures for measuring whole brain volume and intracranial volume were similar to those reported previously
(31,
37). Raters were blind to the subjects’ characteristics. The intraclass correlation coefficients for intrarater reliability based on 30 subjects and interrater reliability based on 10 subjects were 0.97 and 0.91, respectively, for the hippocampus and 0.94 and 0.82, respectively, for the amygdala. The coefficients based on 10 subjects were 0.99 and 0.99, respectively, for whole brain volume and 0.99 and 0.99, respectively, for intracranial volume.
Memory Measures
We used the delayed recall index and percent retention (delayed recall score/immediate recall score×100) from the Wechsler Memory Scale—Revised
(38,
39) as surrogate markers of hippocampal function. We also obtained the Wechsler Memory Scale—Revised indexes of attention/concentration, immediate visual memory, and immediate verbal memory for each participant.
Data Analyses
In previous studies
(5–
7), patients’ hippocampal volume differed from that of comparison subjects by 8%–19%, and based on our pilot study, we estimated that the difference and standard deviation would be 8% and 9%, respectively. Assuming a lifetime prevalence of major depressive episode after cancer diagnosis in cancer survivors of 20%, 63 subjects were needed (13 cancer survivors with a major depressive episode and 50 without) for a two-tailed alpha of 0.05 and a beta of 0.20. The groups with and without a major depressive episode after cancer diagnosis in this study consisted of 17 and 51 subjects, respectively.
Hippocampal volume, amygdala volume, and whole brain volume were analyzed by one-way analysis of variance (ANOVA) and analysis of covariance (ANCOVA), with age, alcohol consumption, handedness, and intracranial volume used to control for differences between the two groups. Repeated-measures ANCOVA with side as the repeated-measures (within-group) factor and age, alcohol consumption, handedness, and intracranial volume as covariates was used to compare left and right hippocampal volume in the subjects with and without a major depressive episode after cancer diagnosis. In a subanalysis to minimize the influence of age, we also performed a paired t test to compare regional brain volumes between the 17 subjects with a first major depressive episode and a one-to-one age- and education-matched subject group from among the 51 subjects without a major depressive episode.
The comparisons of the subjects with and without a major depressive episode after cancer diagnosis also included analysis of the delayed recall index, percentage retention, attention/concentration index, immediate visual index, and immediate verbal memory index by ANOVA and by ANCOVA, with years of education and alcohol consumption as covariates.
As an additional analysis to investigate the possibility of a smaller hippocampus already being present in the subjects who had a major depressive episode after cancer diagnosis, we analyzed the difference in the number of subjects who had a major depressive episode after cancer diagnosis between those with a small hippocampus and those with a large hippocampus with the chi-square test. We used median hippocampal volumes on the left and right to divide the subjects into those with low and high left and right hippocampal volumes.
We also examined associations between medical factors, such as type of mastectomy, interval since cancer surgery, clinical stage, history of adjuvant chemotherapy and tamoxifen therapy, and hippocampal volume with the two-tailed Student’s t test and Pearson’s correlation test to exclude associations between medical factors and hippocampal volume because all subjects were cancer survivors and no healthy comparison subjects were examined. In addition, we stratified all the subjects into four groups according to their age and surveyed the means and standard deviations of the hippocampal volumes in each group to examine whether a nonlinear association exists between age and hippocampal volume, since a nonlinear association between age and brain volume has been previously reported
(40).
The alpha level of significance in most statistical analyses was p<0.05. Since multiple comparisons were made in the analyses of memory function, the p value was set at 0.01. All analyses were performed by using SPSS software version 11 (SPSS, Inc., Chicago).
Results
Hippocampal Volume
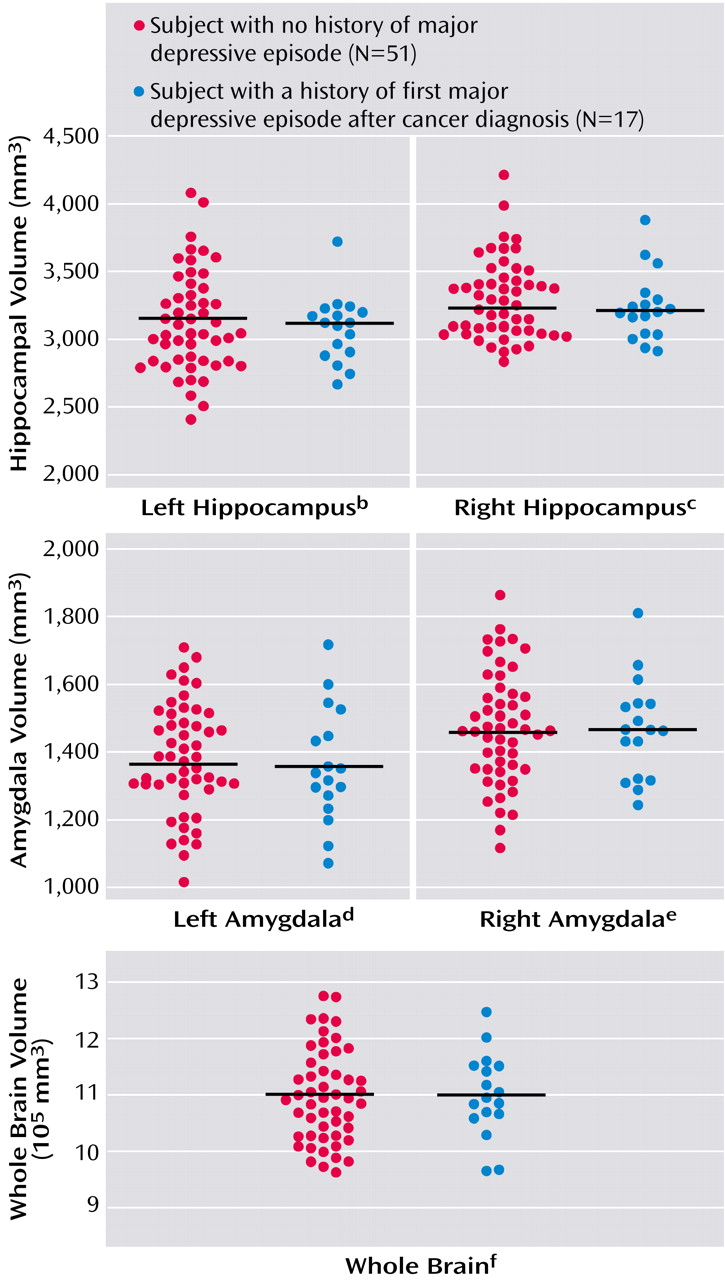
The mean left and right hippocampal volumes of the subjects who experienced a major depressive episode after their cancer diagnosis (N=17) and of the subjects who never experienced a major depressive episode (N=51) are shown in
Figure 1. No statistically significant differences in the volumes of the left or right hippocampus were seen between the two groups based on the results of ANOVAs, and ANCOVAs also did not produce any significant differences (left: F=0.66, df=1, 62, p=0.42; right: F=1.17, df=1, 62, p=0.28). A repeated-measures ANCOVA showed no significant interaction between side and the first major depressive episode after cancer diagnosis (F=0.01, df=1, 62, p=0.92) and no significant main effect of side (F=1.08, df=1, 62, p=0.30). A subanalysis using a paired t test between the 17 cancer survivors who experienced a first major depressive episode after cancer diagnosis and their age- and education-matched comparison subjects revealed no significant difference in the left or right hippocampal volume (left hippocampus: t=0.32, df=16, p=0.75; right hippocampus: t=0.51, df=16, p=0.62).
As an additional analysis, we used the median left hippocampal volume (3,096.6 mm3) to divide the subjects into those with low and high left hippocampal volume. Eight of the subjects with a small left hippocampus (N=34) had a major depressive episode after cancer diagnosis, as opposed to nine of the subjects with a large left hippocampus (N=34), but the difference was not significant (χ2=0.08, df=1, p=0.78). The median value for the right side (3,276.0 mm3) was then used as the dividing line to separate subjects with a low and a high right hippocampal volume. Ten of the subjects with a small right hippocampus (N=34) had a major depressive episode after cancer diagnosis versus seven of the subjects with a large right hippocampus (N=34), but this difference also was not significant (χ2=0.71, df=1, p=0.40).
Memory Functioning
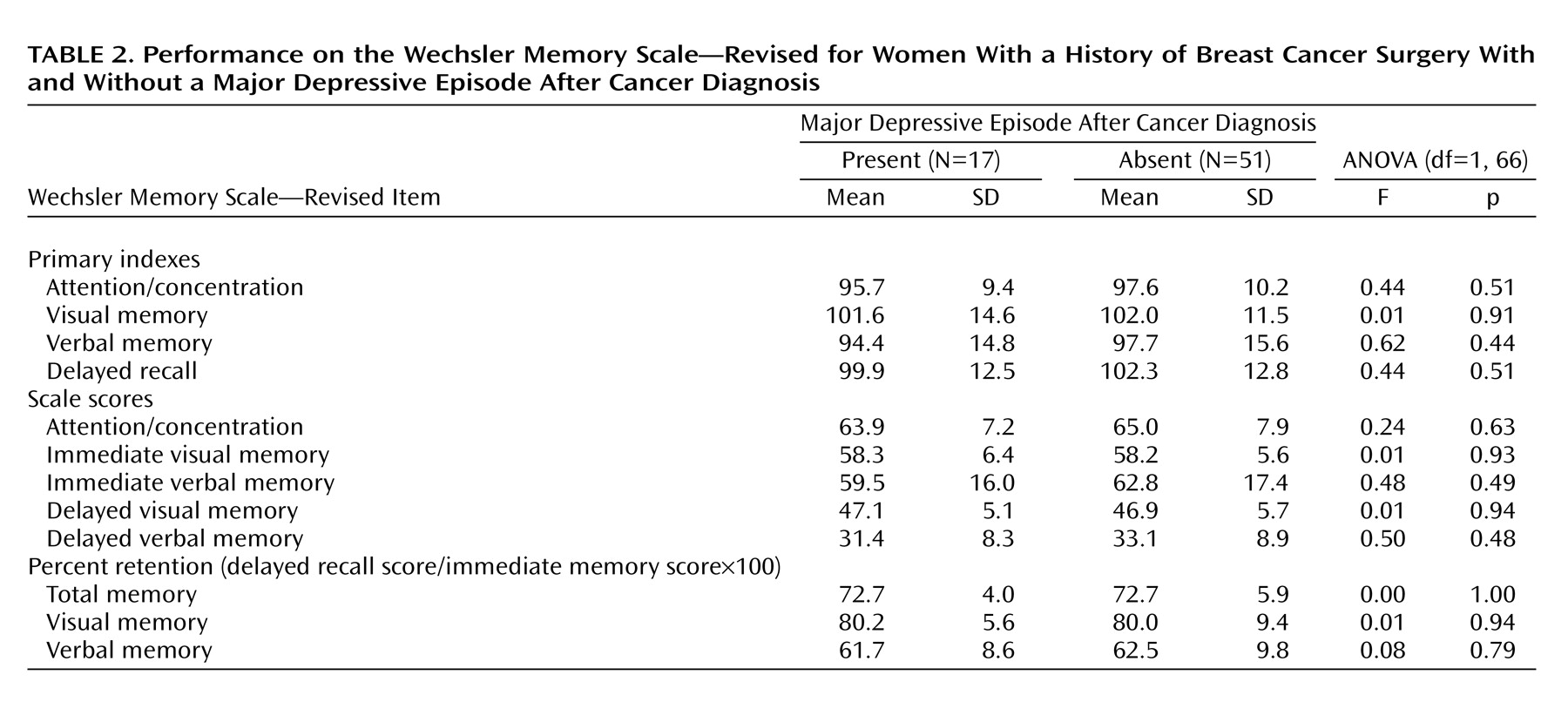
The memory measures of subjects who experienced a major depressive episode after cancer diagnosis and of those who did not are shown in
Table 2. No significant differences in the delayed recall index, the percent retention of visual and verbal memory scores, attention/concentration index, immediate visual memory index, or immediate verbal memory index were observed between the two groups. ANCOVAs showed no significant differences in the delayed recall index (F=0.56, df=1, 64, p=0.46); the percent retention of total (F=0.00, df=1, 63, p=0.98), visual (F=0.03, df=1, 63, p=0.85), or verbal memory scores (F=0.20, df=1, 63, p=0.66); or the attention/concentration index (F=0.46, df=1, 64, p=0.50), immediate visual memory index (F=0.03, df=1, 64, p=0.87), or immediate verbal memory index (F=0.72, df=1, 64, p=0.40) between the two groups.
Hippocampal Volume and Medical Factors
As additional analyses, we examined possible associations between medical factors and hippocampal volume. No significant correlation between hippocampal volume and the interval since cancer surgery was seen (r=0.116, N=68, p=0.35), and no significant difference in the hippocampal volumes between subjects with a clinical stage of 0 or I (N=22) and those with a clinical stage of II or III (N=46) (t=0.39, df=66, p=0.70), between subjects with (N=41) and without (N=27) a history of adjuvant chemotherapy (t=0.63, df=66, p=0.53), between subjects who had (N=29) and who had not (N=39) received tamoxifen therapy (t=0.59, df=66, p=0.56), or between subjects who had undergone a total mastectomy (N=50) and those who had undergone a partial mastectomy (N=18) (t=0.93, df=66, p=0.36) were seen. Although a previous study reported a nonlinear association between age and brain volume, no obvious differences in the mean hippocampal volumes of the four age groups were observed in the present study. A linear regression analysis also revealed no significant association between age and hippocampal volume (data not shown).
Amygdala Volumes
The mean left amygdala volume was 1,357 mm3 (SD=169) in the subjects with a major depressive episode and 1,380 mm3 (SD=162) in the subjects with no depressive episodes. The mean right amygdala volume was 1,464 mm3 (SD=147) in the depressive episode group and 1,468 mm3 (SD=166) in the group without depressive episodes. No significant differences in the left or right amygdala volumes between subjects with and without a first major depressive episode after cancer diagnosis were revealed by an ANOVA (left: F=0.24, df=1, 66, p=0.63; right: F=0.01, df=1, 66, p=0.92) or an ANCOVA (left: F=1.12, df=1, 62, p=0.29; right: F=0.28, df=1, 62, p=0.60). A subanalysis using a paired t test between the 17 cancer survivors with a first major depressive episode after cancer diagnosis and their age- and education-matched comparison subjects revealed no significant differences in the left or right amygdala volumes (left amygdala: t=0.32, df=16, p=0.75; right amygdala: t=0.51, df=16, p=0.62).
Discussion
To our knowledge, this is the first study to investigate whether an association exists between hippocampal volume and a first major depressive episode after cancer diagnosis in cancer survivors. The results showed no significant differences in the left or right hippocampal volumes of cancer survivors who experienced a first major depressive episode after cancer diagnosis and those who had no history of major depressive episodes. The major depressive episodes after cancer diagnosis were of relatively short duration and were nonpsychotic, and almost all of the cancer survivors with a major depressive episode experienced only one episode. No significant differences in sociodemographic or medical factors were seen between the two groups in the present study. These results suggest the absence of a significant association between hippocampal volume and a first major depressive episode after cancer diagnosis in cancer survivors.
In a previous study investigating the possible association between hippocampal volume and a first major depressive episode, the mean duration of the depressive episode was 37 weeks—that is, three times longer than the duration of the depressive episode seen in the present study
(27). No significant associations between the total or gray hippocampal volume and the first major depressive episode among male and female subjects as a whole or among the female subgroup were seen; however, a significantly smaller hippocampus was observed only among the male subgroup that experienced a first major depressive episode
(27). In another recent study, no significant association between gray hippocampal volume and the first major depressive episode among a combined group of male and female patients was seen
(8). The results of the present study, indicating that a significant association between hippocampal volume and the occurrence of a first major depressive episode of short duration does not exist among female cancer survivors, are consistent with the findings of these previous studies
(8,
27). Thus, the hippocampal volume does not appear to be significantly associated with the occurrence of a first major depressive episode of short duration among female patients.
Sheline and colleagues
(5,
6,
19) demonstrated a smaller hippocampus in patients with recurrent major depressive episodes, but the cumulative duration of the depressive episodes in their study (mean duration: 151 weeks, SD=147) was longer than that in the present study. They also demonstrated an inverse correlation between the hippocampal volume and the cumulative duration of the recurrent episodes
(5,
6,
19). No significant reduction in the mean hippocampal volume of the first major depressive episode group may have been observed in the present study because the duration of the first episode was shorter (mean duration=11.6 weeks). Moreover, since the durations of the episodes in the present study were short, with a narrow range (SD=14.2 weeks), a significant correlation between the duration of the depressive episodes and the hippocampal volume—if such a correlation did indeed exist—may have been undetectable.
The first major depressive episodes in the cancer patients in this study were uniquely characterized by the existence of an obvious stressor (the diagnosis of a life-threatening illness) and a relatively late onset. Previous studies reporting a smaller hippocampus in patients with depressive episodes did not describe the preceding physical illness or the stressor encountered before the onset of the episode
(5–
10,
18). Differences in the natures of the stressors may be related to the differences in the results of the previous studies and ours. In addition, the first major depressive episodes after cancer diagnosis in the present study remitted without the use of antidepressants, had a relatively short duration, had no psychiatric comorbidities, and had a relatively late onset. On the other hand, the depressive episodes in the previous studies describing a reduced hippocampal volume were chronic, recurrent
(6–
8,
19), treatment resistant
(9,
10), and/or had, in part, an early onset
(8). Patients experiencing such episodes often exhibited psychiatric comorbidities, suggesting a possible genetic predisposition toward depression, compared to the subjects in the present study
(41). Thus, the results of the present study suggest that nonchronic, late-onset, nonrefractory first major depressive episodes with no psychiatric comorbidities after cancer diagnosis may not be associated with a smaller hippocampus.
When the subjects in the present study were divided into two groups according to the median hippocampal volume value, no significant difference was found between the proportion of subjects with the first major depressive episode in those with a larger hippocampus and those with a smaller hippocampus. We speculate that a smaller hippocampus might not be a cause of susceptibility to a first major depressive episode after cancer diagnosis, although further study regarding this point is needed.
The results of the present study showed no significant differences in memory function, including delayed recall function, between the cancer survivors who experienced a first major depressive episode after cancer diagnosis and those who had never experienced a major depressive episode. In previous studies, a decrease in memory function was not prominent among patients who experienced a first major depressive episode
(42,
43). However, a recent report by MacQueen et al.
(8,
44) demonstrated a significant alteration of memory function in patients who had experienced a first major depressive episode when memory function was examined with a task that has been recognized as a method of examining hippocampus-dependent memory. Although no significant difference in delayed memory function was found between subjects who had or had not experienced a depressive episode in the present study, more specific measurements may demonstrate an alteration in hippocampal function in cancer survivors who experience a first major depressive episode after cancer diagnosis.
The amygdala has been implicated in major depressive episodes
(45). However, the results of previous studies measuring amygdala volume in patients with major depressive episodes have been inconsistent
(46–
48). A previous study reported a significantly larger amygdala volume, as measured with MRI, in patients who had experienced a first major depressive episode
(47). The difference between the results of this previous study and ours may be attributed to differences in the age and sex of the subjects, the duration of the depressive episodes, the depressed state (current versus past episode), and the methods used to measure amygdala volume. Further study is needed to determine whether amygdala volume is related to the occurrence of a first major depressive episode.
The present study had the following limitations:
1.
Because of its cross-sectional design, it could not confirm any causality between hippocampal volume and a first major depressive episode after cancer diagnosis
2.
Since the results were obtained from only one institution, the subjects were not representative of all cancer survivors
3.
All of the subjects were cancer survivors, and no healthy comparison subjects who had not experienced cancer were examined. Although the present study did not show any significant associations between hippocampal volume and medical factors, we could not exclude the possibility of the cancer experience itself having unknown effects on hippocampal volume or the presence of cancer-associated factors diluting the difference between the two groups. In the present study, some of the subjects had received tamoxifen. Previous studies have shown that tamoxifen produces mood-stabilizing effects through the inhibition of protein kinase C
(49,
50)4.
In the present study, we did not examine the presence of important depression-associated factors, such as a family history of major depressive episodes
(51)5.
Because the number of subjects in this study was determined so that an 8% difference in hippocampal volume could be detected between the two groups, we were unable to detect differences smaller than 8%
6.
Although the present study showed no significant differences in total hippocampal volume, since we did not separately measure the volume of the gray or white matter of the hippocampus, we could not exclude the possibility of a significant difference in gray or white matter volumes
In conclusion, no significant differences in left or right hippocampal volumes were found between female cancer survivors who experienced a nonchronic first major depressive episode of relatively short duration and their respective comparison subjects, who had never experienced a major depressive episode. However, a longitudinal study involving healthy comparison subjects and a functional neuroimaging study are needed to elucidate possible associations between major depressive episodes after cancer diagnosis and hippocampal volume.