Antipsychotic Medications for Low-Income Preschoolers: Long Duration and Psychotropic Medication Polypharmacy
Abstract
Objective:
Methods:
Results:
Conclusions:
HIGHLIGHTS
Methods
Sample
Definitions and Covariates
Statistical Analysis
Results
Descriptive Characteristics
| Total exposure to antipsychotic medication (years) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Overall (N=316) | 0–2 (N=126) | 2–4 (N=104) | 4–6 (N=86) | ||||||
| Variable | N | % | N | % | N | % | N | % | pb |
| Age in 2017 (M±SD years) | 9.6±.6 | 9.6±.7 | 9.7±.6 | 9.5±.7 | .027 | ||||
| Exposure to antipsychotic (M±SD years) | 2.6±1.7 | .8±.6 | 3.1±.6 | 4.6±.4 | <.001 | ||||
| Foster care | 63 | 20 | 23 | 18 | 17 | 16 | 23 | 27 | .169 |
| Nonmetro area | 202 | 64 | 87 | 69 | 67 | 64 | 48 | 56 | .142 |
| Median comorbid psychiatric diagnosesc | 2.5 | 2.0 | 3.0 | 3.0 | <.001 | ||||
| Diagnosis | |||||||||
| Schizophrenia and other psychotic processes | 30 | 10 | 5 | 4 | 13 | 13 | 12 | 14 | .023 |
| Autism spectrum disorders | 137 | 43 | 31 | 25 | 51 | 49 | 55 | 64 | <.001 |
| Other neurodevelopmental disordersd | 208 | 66 | 68 | 54 | 70 | 67 | 70 | 81 | .000 |
| Disruptive behavior disorders (total) | 293 | 93 | 116 | 92 | 101 | 97 | 76 | 88 | .065 |
| Attention-deficit hyperactivity disorder | 288 | 91 | 112 | 89 | 100 | 96 | 76 | 88 | .089 |
| Oppositional defiant disorder | 165 | 52 | 63 | 50 | 54 | 52 | 48 | 56 | .706 |
| Conduct disorder | 173 | 55 | 67 | 53 | 56 | 54 | 50 | 58 | .756 |
| Mood disorders (total) | 203 | 64 | 69 | 55 | 74 | 71 | 60 | 70 | .016 |
| Depressive disorders | 174 | 55 | 54 | 43 | 69 | 66 | 51 | 59 | .001 |
| Bipolar disorder | 88 | 28 | 26 | 21 | 35 | 34 | 27 | 31 | .063 |
| Mood disorder not otherwise specified | 162 | 51 | 48 | 38 | 63 | 61 | 51 | 59 | .001 |
| Disruptive mood dysregulation disorder | 90 | 29 | 21 | 17 | 42 | 40 | 27 | 31 | <.001 |
| Anxiety and trauma-related disorders (total) | 202 | 64 | 78 | 62 | 66 | 63 | 58 | 67 | .707 |
| PTSD | 70 | 22 | 23 | 18 | 26 | 25 | 21 | 24 | .395 |
| Tic disorders | 24 | 8 | 10 | 8 | 6 | 6 | 8 | 9 | .647 |
| Epilepsy and seizure disorders | 70 | 22 | 23 | 18 | 26 | 25 | 21 | 24 | .395 |
| Polypharmacye | |||||||||
| 2 medication classes | 79 | 25 | 50 | 40 | 22 | 21 | 7 | 8 | .004 |
| 3 medication classes | 107 | 34 | 34 | 27 | 42 | 40 | 31 | 36 | <.001 |
| ≥4 medication classes | 97 | 31 | 11 | 9 | 39 | 38 | 47 | 55 | <.001 |
| Prescribed by a psychiatrist | 266 | 84 | 99 | 79 | 91 | 88 | 76 | 88 | .083 |
| Prescribed by a primary care provider | 300 | 95 | 118 | 94 | 97 | 93 | 85 | 99 | .153 |
| Visit with a primary care provider | 297 | 94 | 118 | 94 | 97 | 93 | 82 | 95 | .818 |
| Emergency department visit | 277 | 88 | 109 | 87 | 88 | 85 | 80 | 93 | .189 |
| Inpatient hospitalizationf | 107 | 34 | 27 | 21 | 44 | 42 | 36 | 42 | .001 |
| Metabolic screeningg | 131 | 42 | 32 | 25 | 52 | 50 | 47 | 55 | <.001 |
| Lipid screening | 150 | 48 | 42 | 33 | 57 | 55 | 51 | 59 | <.001 |
| Blood glucose screening | 236 | 75 | 81 | 64 | 83 | 80 | 72 | 84 | .002 |
| Metabolic dysfunctionh | 28 | 9 | 5 | 4 | 11 | 11 | 12 | 14 | .032 |
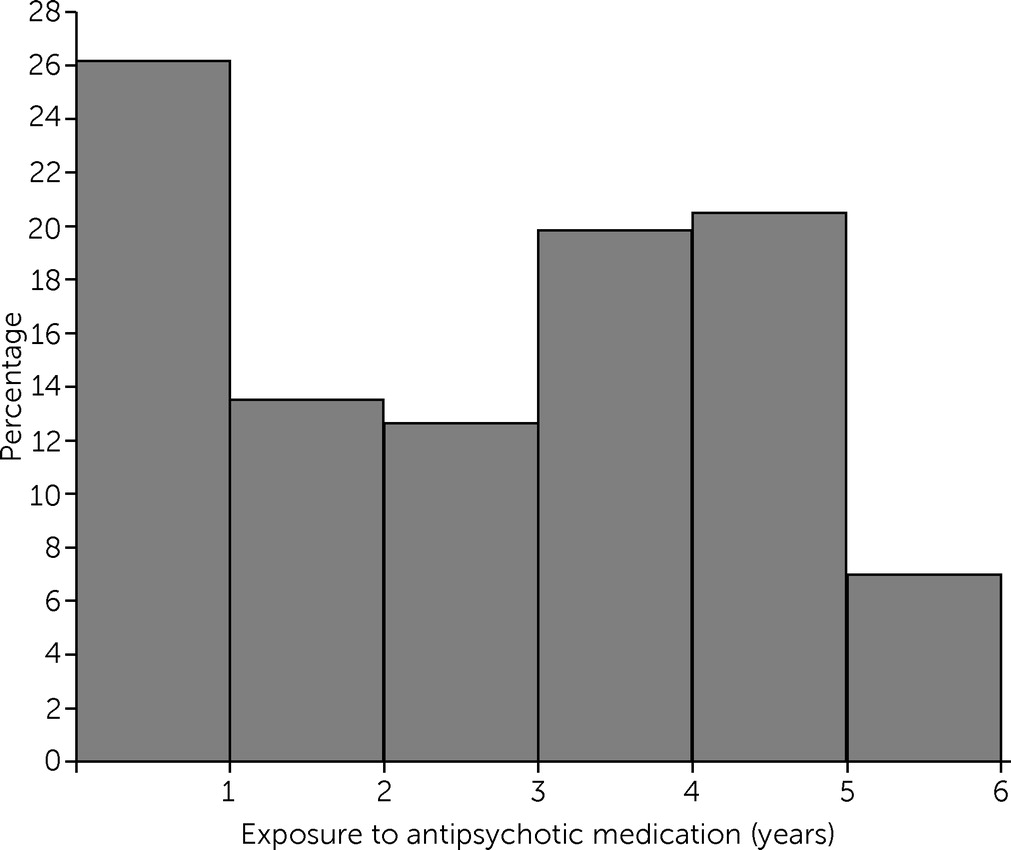
Diagnoses
Safety and Monitoring
Types of Medications
| Medication | N | % |
|---|---|---|
| Antipsychotics | 316 | 100 |
| First generation | 17 | 5 |
| Second generation | 315 | >99 |
| Risperidone | 294 | 93 |
| Aripiprazole | 100 | 32 |
| Quetiapine | 85 | 27 |
| Stimulants | 275 | 87 |
| Amphetamine based | 229 | 72 |
| Methylphenidate | 194 | 61 |
| Dexmethylphenidate | 95 | 30 |
| Alpha-agonists | 286 | 91 |
| Clonidine | 230 | 73 |
| Guanfacine | 212 | 67 |
| Antidepressants | 200 | 63 |
| Trazodone | 95 | 30 |
| Sertraline | 67 | 21 |
| Mirtazapine | 53 | 17 |
| Fluoxetine | 50 | 16 |
| Anticonvulsants and mood stabilizers | 157 | 50 |
| Valproic acid | 76 | 24 |
| Oxcarbazepine | 59 | 19 |
| Lamotrigine | 55 | 17 |
| Lithium | 13 | 4 |
| Benzodiazepines, anxiolytics, and hypnotics | 115 | 36 |
| Hydroxyzine | 83 | 26 |
Predictors of Duration of Antipsychotic Medication
| Variable | IRRa | 95% CI | p |
|---|---|---|---|
| Male (reference: female) | 1.25 | 1.04–1.49 | .017* |
| Nonmetro residence (reference: metro residence) | .95 | .82–1.11 | .536 |
| Prescribed by a psychiatrist | .91 | .78–1.06 | .230 |
| Child in foster care in 2012 | 1.25 | 1.00–1.56 | .046* |
| Schizophrenia and other psychotic processes | 1.04 | .68–1.59 | .855 |
| Autism spectrum disorders | 1.26 | 1.03–1.54 | .022* |
| Other neurodevelopmental disordersb | 1.14 | .90–1.45 | .272 |
| Disruptive behavior disorders | |||
| Attention-deficit hyperactivity disorder | .98 | .80–1.20 | .440 |
| Oppositional defiant disorder | 1.08 | .89–1.32 | .096 |
| Conduct disorder | .83 | .67–1.03 | .827 |
| Mood disorders | |||
| Depressive disorders | 2.12 | .90–4.97 | .084 |
| Bipolar disorder | 1.24 | .94–1.64 | .126 |
| Mood disorder not otherwise specified | .50 | .21–1.16 | .108 |
| Disruptive mood dysregulation disorder | .38 | .15–.98 | .044* |
| Anxiety and trauma-related disorders | |||
| PTSD | 1.18 | .86–1.63 | .302 |
| Tic disorders | .84 | .40–1.77 | .644 |
| Epilepsy and seizure disorders | 1.01 | .77–1.31 | .956 |
| At least one inpatient code in 2012 | 1.16 | .89–1.50 | .265 |
| At least one emergency department visit in 2012 | 1.01 | .87–1.17 | .904 |
| N of mental disorders in 2012 | 1.00 | .90–1.10 | .967 |
Discussion
Conclusions
Footnote
Supplementary Material
- Download
- 95.30 KB
References
Information & Authors
Information
Published In
History
Keywords
Authors
Funding Information
Metrics & Citations
Metrics
Citations
Export Citations
If you have the appropriate software installed, you can download article citation data to the citation manager of your choice. Simply select your manager software from the list below and click Download.
For more information or tips please see 'Downloading to a citation manager' in the Help menu.
View Options
View options
PDF/EPUB
View PDF/EPUBLogin options
Already a subscriber? Access your subscription through your login credentials or your institution for full access to this article.
Personal login Institutional Login Open Athens loginNot a subscriber?
PsychiatryOnline subscription options offer access to the DSM-5-TR® library, books, journals, CME, and patient resources. This all-in-one virtual library provides psychiatrists and mental health professionals with key resources for diagnosis, treatment, research, and professional development.
Need more help? PsychiatryOnline Customer Service may be reached by emailing [email protected] or by calling 800-368-5777 (in the U.S.) or 703-907-7322 (outside the U.S.).

