Several neurobiological theories of schizophrenia hypothesize that the pathophysiology of the disorder includes abnormal functional interactions between the cortex and thalamus (
1–
3). Thalamocortical networks are organized topographically into parallel pathways linking distinct cortical areas to specific thalamic nuclei (
4,
5). For example, the prefrontal cortex is linked to anterior and dorsomedial areas of the thalamus, whereas motor and somatosensory cortices connect to ventral lateral and ventral posterior-lateral areas of the thalamus, respectively (
4). Consequently, dysfunction of thalamocortical networks may account for the wide array of clinical and cognitive symptoms observed in schizophrenia (
6). Indeed, functional and structural imaging investigations provide broad support for thalamocortical dysfunction (7–
12). However, the topographical arrangement of reciprocal connections between the cortex and thalamus also raises the distinct possibility that thalamocortical networks may be differentially affected in schizophrenia (
13). Determining the anatomical specificity of thalamocortical network dysconnectivity using conventional task-based functional imaging is challenging because it requires using multiple cognitive paradigms that reliably activate distinct cortical areas and their corresponding thalamic targets in healthy subjects as well as in patients with schizophrenia. As a result, the anatomical specificity of thalamocortical functional dysconnectivity in schizophrenia remains largely unknown.
Resting-state functional MRI (fMRI), which examines correlations in intrinsic low-frequency fluctuations in the blood-oxygen-level dependent (BOLD) signal across brain regions, sidesteps many of the limitations associated with conventional task-based functional imaging and has proven useful for mapping brain networks. In a series of elegant studies, Zhang et al. (14, 15) systematically mapped functional connectivity between the cortex and thalamus using resting-state fMRI. Specifically, by parceling the cortex into regions of interest that corresponded to prefrontal, motor, somatosensory, temporal, and parietal/occipital cortices and using them as seeds in a functional connectivity analysis, they were able to demonstrate that activity in each cortical area correlated with distinct, largely nonoverlapping regions of the thalamus. For instance, the prefrontal cortex functionally correlated with the anterior and dorsomedial thalamus, whereas the motor cortex correlated with the ventral lateral thalamus (14, 15). Critically, the patterns of intrinsic functional connectivity revealed using resting-state fMRI correspond closely to anatomical connections based on histology and diffusion tensor imaging of white matter pathways (14–
18). Consequently, resting-state fMRI may be an ideal method for examining the integrity of functional connectivity between the cortex and thalamus in individuals with schizophrenia. To this end, we employed an approach similar to that used by Zhang et al. to examine functional connectivity between the cortex and thalamus in schizophrenia and to determine whether specific thalamocortical networks are differentially affected in the disorder.
Results
Ten healthy subjects and 11 patients had either resting-state scans that did not pass our quality assurance protocol or poor-quality structural scans (i.e., significant motion artifact). Thus, the final data set consisted of 77 healthy comparison subjects and 62 schizophrenia patients (schizoaffective disorder, N=24). Demographic characteristics of patients and healthy subjects are summarized in
Table 1. With the exception of three individuals who were not receiving antipsychotic medication, patients were receiving treatment with one or two atypical antipsychotics (N=42 and N=9, respectively), one atypical and one conventional antipsychotic (N=6), or one conventional antipsychotic (N=2). The groups did not differ on parental education, gender distribution, or ethnicity. As expected, premorbid intellect was lower for the schizophrenia group (t=4.84, df=137, p<0.001). Group differences on age fell short of statistical significance (t=1.92, df=137, p=0.06), with the patient group slightly older than the healthy comparison group. Rather than selectively remove younger healthy subjects from the analysis to exactly equate the two groups on age, which would have reduced statistical power and possibly introduced bias, age was included as a covariate in the between-group imaging analyses. Chi-square analysis indicated that fewer patients were scanned on the 3T-B scanner (healthy subjects, N=36; patients, N=16; χ
2=4.45, df=1, p=0.01). To ensure that data could be combined across scanners, we compared scanners on the five signal features extracted from the quality assurance protocol. None of the features differed between scanners.
Thalamocortical Connectivity
Healthy comparison subjects.
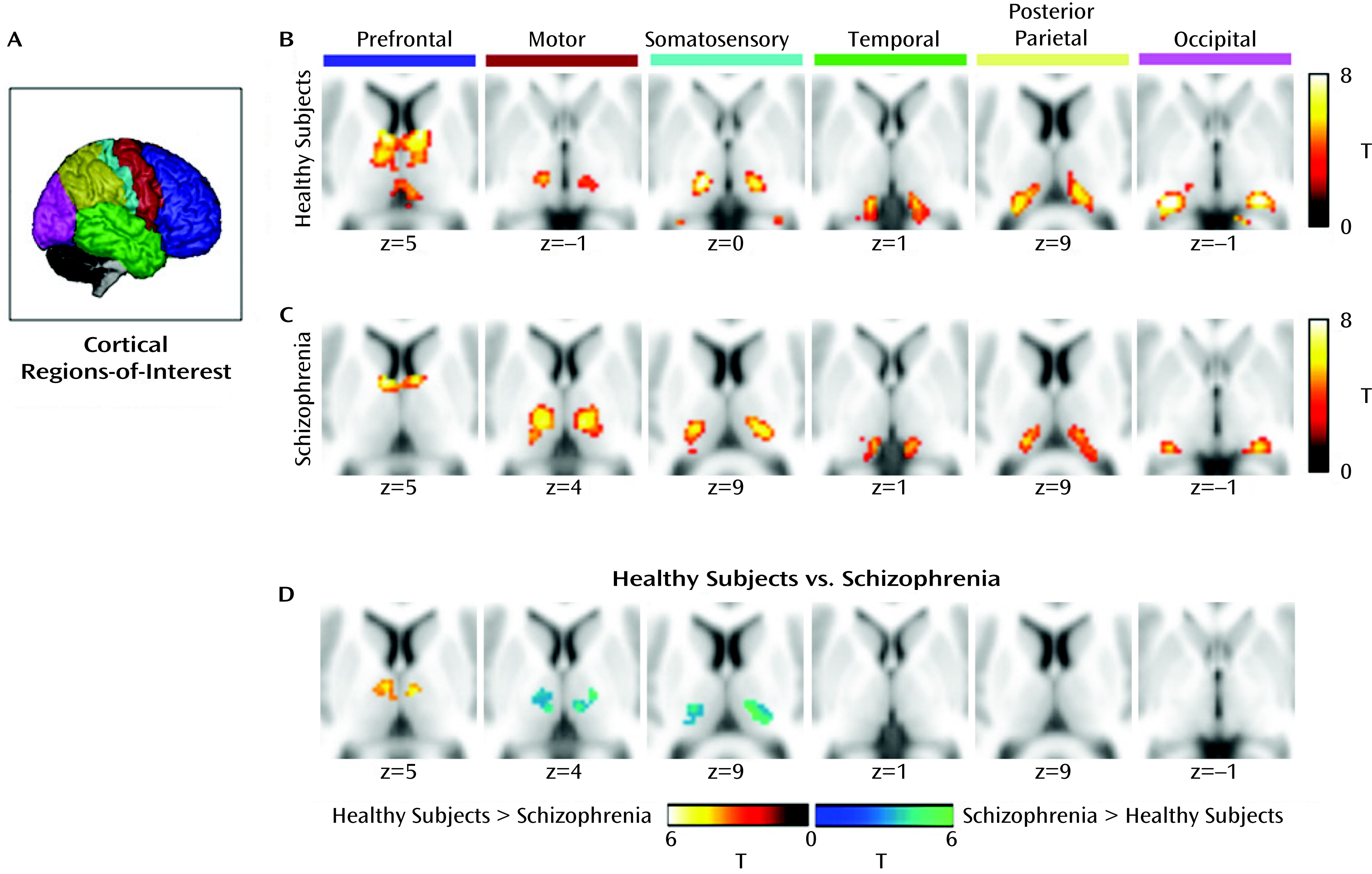
Each cortical region of interest was connected to distinct, largely nonoverlapping regions of the thalamus (
Figure 1). The present results are virtually identical to those reported in previous studies using the same method in healthy adults (14, 15,
23). As expected, the prefrontal cortex was functionally connected to anterior and dorsomedial regions of the thalamus. Motor and somatosensory regions of interest correlated strongly with ventral lateral and ventral posterior-lateral portions of the thalamus, respectively. The temporal lobe and occipital cortex correlated with posterior medial and lateral areas of the thalamus that appeared consistent with the medial geniculate nucleus and the lateral geniculate nucleus, respectively. The posterior parietal cortex was robustly connected with the lateral posterior nucleus and the pulvinar.
Schizophrenia patients.
Schizophrenia patients demonstrated a high degree of segregation in the thalamus, and the overall pattern of functional connectivity between the cortex and thalamus corresponded well with previous findings in healthy subjects (
Figure 1). However, there were qualitative differences between patients and healthy subjects. In the patient group, prefrontal cortex connectivity with the thalamus appeared markedly less robust and was restricted largely to the anterior thalamus, whereas the spatial extent of motor and somatosensory connectivity was considerably more extensive. Specifically, the thalamic clusters showing connectivity with these regions of interest were expanded in the dorsal and lateral directions in the patient group.
Group Differences in Thalamocortical Connectivity
Group differences in thalamocortical connectivity are summarized in
Table 2 and depicted graphically in
Figure 1. The patient group demonstrated significantly less connectivity between the prefrontal cortex and left and right dorsomedial thalamus. In contrast, patients exhibited increased thalamic connectivity with the motor and somatosensory cortex. No differences were observed between groups in temporal, posterior parietal, and occipital cortex connectivity with the thalamus.
To ensure that the group differences in connectivity were not a result of antipsychotic medication, structural brain changes in the thalamus, or scanner effects, we extracted the connectivity beta weights from each cluster identified in the between-group analysis and performed several supplementary analyses. To rule out medication effects, we correlated the cluster beta weights with the current antipsychotic dosage in chlorpromazine equivalents. Functional connectivity did not correlate with dosage in any cluster. To rule out the possibility that connectivity differences were a result of structural brain differences in the thalamus, we extracted each participant’s mean gray matter content within each cluster (i.e., average fraction of gray matter within the cluster) from the gray matter segmented images and performed an analysis of covariance (ANCOVA) on the connectivity beta weights, with the mean gray matter content included as a covariate. The group effect was highly significant for each cluster (all p values <0.00005), indicating that the functional connectivity changes observed in the schizophrenia group were not due to differences in the content of gray matter within each cluster. Finally, to ensure that the group effects were not a result of differential allocation of patients and healthy subjects to the two scanners, we performed ANCOVAs on the beta weights extracted from the clusters identified in the between-group analysis with scanner included as a covariate. The group effect remained highly significant for all of the clusters (all p values <0.0005). Additionally, the results of the voxel-wise analysis with scanner included as a covariate were virtually indistinguishable from those of the main analysis (see Figure 2 in the online
data supplement).
We also examined the relationship between the connectivity changes, clinical symptoms (i.e., PANSS positive, negative, and general psychopathology scores), and illness-relevant demographic characteristics (i.e., duration of illness, premorbid IQ). Given the exploratory nature of this analysis, only correlations with alpha levels <0.005 were considered significant. No correlations between functional connectivity and clinical symptoms or illness-relevant demographic characteristics met this threshold.
Discussion
Dysfunction of thalamocortical networks has been implicated in the pathophysiology of schizophrenia (2,
3). We used resting-state fMRI to determine the anatomical specificity of thalamocortical network dysfunction in schizophrenia (15). We found that functional connectivity between the prefrontal cortex and dorsomedial/anterior thalamus was reduced in schizophrenia. In contrast, thalamic functional connectivity with motor and somatosensory cortical areas was markedly increased. These results indicate that functional networks linking the cortex to the thalamus are abnormal in schizophrenia and that the changes are characterized by both hypo- and hyperconnectivity.
The combination of decreased prefrontal-thalamic and increased thalamic connectivity with motor and somatosensory cortical regions is the most striking aspect of our findings, which, at first glance, appear inconsistent with the general notion that neural connectivity is reduced overall in schizophrenia (e.g., reference
25). However, when interpreted from a developmental perspective, our results provide compelling support for the neurodevelopmental model of schizophrenia. Using the same method, Fair et al. (
23) found marked differences in thalamocortical functional connectivity between children, adolescents, and adults. Specifically, prefrontal-thalamic connectivity is largely absent in children and adolescents, suggesting that this network develops abruptly during the transition from adolescence to adulthood. Motor and somatosensory connectivity, on the other hand, appears to follow an inverted U-curve, which is maximal in adolescence compared with childhood and adulthood. The changes observed in schizophrenia may result from abnormal late brain maturation during the transition from adolescence to adulthood, which derails the normal development of prefrontal-thalamic connectivity and refinement of somatomotor-thalamic connectivity. The functional consequences of these changes remain to be characterized; however, reduced structural connectivity between the prefrontal cortex and thalamus has been linked to working memory impairment and prefrontal brain activity in schizophrenia (
26). It is possible that a similar relationship may be observed for resting-state functional connectivity.
While the combination of decreased prefrontal and increased somatomotor connectivity with the thalamus is consistent with atypical brain maturation in schizophrenia, the absence of group differences in temporal-thalamic connectivity potentially argues against a neurodevelopmental basis for thalamocortical dysconnectivity. In contrast to prefrontal-thalamic connectivity, temporal cortex connectivity with the thalamus decreases with age in typically developing individuals (
23). Therefore, schizophrenia patients might be expected to demonstrate increased temporal-thalamic connectivity if thalamocortical network dysfunction is indeed associated with atypical brain maturation. However, the precise timing of developmental changes in thalamocortical functional connectivity is poorly understood. The limited available evidence, which comes from a single cross-sectional study, suggests that much of the reduction in temporal-thalamic connectivity occurs between childhood and adolescence (
23). Consequently, it is possible that the developmental disruption in schizophrenia occurs after temporal-thalamic connectivity has fully matured but before prefrontal and somatomotor thalamic networks have reached adult levels. A better understanding of the normal developmental trajectories of thalamocortical connectivity and investigation of thalamocortical connectivity in first-episode schizophrenia are required to test this hypothesis. The lack of group differences in temporal-thalamic, as well as occipital-thalamic connectivity, is also interesting given strong evidence of sensory processing deficits in schizophrenia (
27). It is possible that connectivity during tasks, rather than in the resting state, may be associated with sensory processing dysfunction. Alternatively, auditory and visual sensory processing deficits in schizophrenia may be related to dysfunction at the level of cortico-cortical interactions, rather than thalamocortical connectivity.
Our findings also raise the possibility that thalamocortical dysconnectivity results from selective pathology of one or more nuclei of the thalamus or their corresponding cortical targets. A reduced number of neurons in the mediodorsal thalamus has been reported by several investigators, although there are also reports of normal numbers of neurons (for a review, see reference
13). Similarly, an array of neuronal and molecular changes have also been observed in dorsolateral prefrontal cortex circuitry (for a review, see reference
28). Thus, thalamocortical dysconnectivity in schizophrenia may result from selective pathology of specific thalamic nuclei or their corresponding cortical targets. Resting-state connectivity networks are conserved across species, suggesting that animal models will be particularly useful in elucidating the effects of focal neuronal, molecular, and genetic manipulations on large-scale brain networks (
29–
33).
Our results are also informed by considering the physiology of BOLD functional connectivity. There is considerable overlap between functional and structural connectivity in the thalamus, suggesting that thalamocortical functional connectivity reflects direct anatomical connections (14). Results from a recent diffusion tensor imaging investigation demonstrating reduced connectivity between the thalamus and lateral prefrontal cortex and increased connectivity between the somatosensory cortex and thalamus in schizophrenia provide a potential anatomical basis for our findings (
26). However, it is clear from the broader functional connectivity literature that brain regions that are not directly anatomically connected can still demonstrate robust functional connectivity, indicating that resting-state connectivity networks reflect extended, polysynaptic networks (e.g., reference
34). This interpretation is supported by findings from a recent combined resting-state fMRI/electrocorticography investigation, which found that functional connectivity, particularly positive correlations, predicted electrically evoked brain responses (
35). Combined, these findings confirm a neural basis for low-frequency BOLD functional connectivity but raise the possibility that abnormal thalamocortical connectivity in schizophrenia may reflect alterations in direct or indirect pathways linking the thalamus and cortex. Future work combining functional and structural connectivity will help clarify the nature of thalamocortical dysconnectivity in schizophrenia.
There are several limitations of our investigation that merit consideration. First, schizophrenia patients were receiving antipsychotic medication. Although we did not find any evidence that medication was related to functional connectivity abnormalities observed in patients, it is possible that antipsychotic treatment effects on connectivity may not be dose-dependent. Second, low-frequency BOLD functional connectivity varies to some extent across cognitive states, levels of consciousness (i.e., awake versus light sleep), and even eyes-open versus eyes-closed conditions (
36–
39). Since we instructed participants to keep their eyes closed during scanning, we cannot rule out the possibility that some may have fallen asleep during scanning and that group differences in arousal may have contributed to our results. Studies examining thalamocortical connectivity across cognitive states and levels of arousal are required to determine whether the alterations observed in schizophrenia patients transcend cognitive states and arousal levels. Moreover, resting-state connectivity is modified by recent experiences, raising the possibility that the abnormalities we observed in thalamocortical connectivity are secondary to negative life experiences associated with illness chronicity, such as long-term reduction in social interaction and cognitive engagement, rather than a result of the pathophysiology of schizophrenia (
40). Third, while we argue that the changes are at least partially consistent with neurodevelopmental hypotheses of schizophrenia, it is premature to exclude a neurodegenerative explanation for our findings. A combination of decreased network connectivity with compensatory increases in other networks has been observed in degenerative illnesses, such as Alzheimer’s disease (
41). Replication of our findings in nonmedicated/minimally treated first-episode or early-phase schizophrenia patients will strengthen the case for a neurodevelopmental basis for thalamocortical dysconnectivity. Finally, although we applied a well-established method to examine thalamocortical functional connectivity, there are nonetheless limitations of the technique. The use of large cortical areas as seeds, while useful for functionally segregating the thalamus, does not allow for a more fine-grained analysis at the cortical level. Interestingly, using a voxel located in the mediodorsal thalamus as the seed for functional connectivity analysis, Welsh et al. (
12) found that mediodorsal thalamic connectivity with the left and right caudate and anterior cingulate gyrus was reduced in schizophrenia. Follow-up investigation using the thalamic clusters identified in our study as seeds in an independent cohort of patients, or an independent-components analysis, may help further refine the anatomical specificity of thalamocortical dysconnectivity.
In conclusion, we observed that altered resting-state functional connectivity between the thalamus and cortex is altered in schizophrenia. The alterations are characterized by decreased prefrontal-thalamic connectivity and increased thalamic connectivity with motor and somatomotor cortex. Combined, our results implicate abnormal late-brain maturation in the neuropathology of schizophrenia. Future studies combining functional connectivity with assessment of phenotypes more closely related to thalamocortical networks than to complex clinical symptoms may help elucidate the functional consequences of thalamocortical dysconnectivity in schizophrenia.